Blood Test Predicts Prostate Cancer in Chemotherapy Recipients
|
By LabMedica International staff writers Posted on 05 Jan 2011 |
A simple blood test can help predict whether men taking a drug to shrink their prostate will progress to an aggressive form of prostate cancer.
The presence of prostate specific antigen (PSA) in the blood is a screening test for prostate cancer and although sometimes when elevated can indicate cancer, where none is present and miss life threatening tumors.
Investigators at Washington University School of Medicine (St. Louis, MO, USA), studied 8,231 men ages 50-75 who were randomly assigned to receive a placebo or a daily 0.5 mg dose of the prostate shrinking drug of dutasteride. The men had elevated PSA levels of 2.5 ng/mL to 10 ng/mL, but no evidence of cancer on biopsies performed within six months of enrolling in the trial. The scientists were testing the performance of the PSA test as a marker for prostate cancer, particularly for aggressive cancer. Among men taking dutasteride, the investigators found that any subsequent rise in PSA levels over the course of the study was more likely to be linked to aggressive, high-grade tumors (Gleason score 7-10), compared to rising PSA levels in men on a placebo. The Gleason scoring system measures tumor aggressiveness based on biopsy results and can range from 2-10, with 10 being the most aggressive.
The data showed that over four years, the PSA levels increased in 72% of men taking a placebo and only 29% of men taking dutasteride,. However, there was no significant difference in high-grade tumors between the two groups. Men taking dutasteride were almost twice as likely to have aggressive prostate cancer if their PSA levels rose, compared to men whose PSA levels went up while taking a placebo. In men with any increase in PSA, aggressive, high-grade tumors were diagnosed in 13.2 % of those on dutasteride and 7.7% of those taking a placebo.
Even a slight rise in PSA levels was a more accurate predictor of aggressive tumors. Among men whose PSA levels increased one point or less 10.3% of those taking dutasteride had aggressive cancer, compared with 5.4% taking a placebo. That trend also held for larger increases in PSA levels. Among men whose PSA levels rose two points or more, nearly 20.9% of those taking dutasteride had aggressive cancer, compared with 9.8% taking a placebo. In contrast, PSA levels tended to decrease or remain stable in men taking dutasteride who had either low-grade tumors or no cancer at all.
Gerald Andriole, MD, chief of urological surgery at the school of medicine, said, "Men who are taking dutasteride can be confident that the drug does not weaken the ability of PSA to find cancer if it develops. Rather, the drug enhances the ability to find cancer if PSA levels are rising." The study was published online on November 12, 2010, in the Journal of Urology.
Related Links:
Washington University School of Medicine
The presence of prostate specific antigen (PSA) in the blood is a screening test for prostate cancer and although sometimes when elevated can indicate cancer, where none is present and miss life threatening tumors.
Investigators at Washington University School of Medicine (St. Louis, MO, USA), studied 8,231 men ages 50-75 who were randomly assigned to receive a placebo or a daily 0.5 mg dose of the prostate shrinking drug of dutasteride. The men had elevated PSA levels of 2.5 ng/mL to 10 ng/mL, but no evidence of cancer on biopsies performed within six months of enrolling in the trial. The scientists were testing the performance of the PSA test as a marker for prostate cancer, particularly for aggressive cancer. Among men taking dutasteride, the investigators found that any subsequent rise in PSA levels over the course of the study was more likely to be linked to aggressive, high-grade tumors (Gleason score 7-10), compared to rising PSA levels in men on a placebo. The Gleason scoring system measures tumor aggressiveness based on biopsy results and can range from 2-10, with 10 being the most aggressive.
The data showed that over four years, the PSA levels increased in 72% of men taking a placebo and only 29% of men taking dutasteride,. However, there was no significant difference in high-grade tumors between the two groups. Men taking dutasteride were almost twice as likely to have aggressive prostate cancer if their PSA levels rose, compared to men whose PSA levels went up while taking a placebo. In men with any increase in PSA, aggressive, high-grade tumors were diagnosed in 13.2 % of those on dutasteride and 7.7% of those taking a placebo.
Even a slight rise in PSA levels was a more accurate predictor of aggressive tumors. Among men whose PSA levels increased one point or less 10.3% of those taking dutasteride had aggressive cancer, compared with 5.4% taking a placebo. That trend also held for larger increases in PSA levels. Among men whose PSA levels rose two points or more, nearly 20.9% of those taking dutasteride had aggressive cancer, compared with 9.8% taking a placebo. In contrast, PSA levels tended to decrease or remain stable in men taking dutasteride who had either low-grade tumors or no cancer at all.
Gerald Andriole, MD, chief of urological surgery at the school of medicine, said, "Men who are taking dutasteride can be confident that the drug does not weaken the ability of PSA to find cancer if it develops. Rather, the drug enhances the ability to find cancer if PSA levels are rising." The study was published online on November 12, 2010, in the Journal of Urology.
Related Links:
Washington University School of Medicine
Latest Clinical Chem. News
- Noninvasive Urine Test May Support Earlier Diagnosis of Psychiatric Disorders
- At-Home Blood and Cognitive Tests Support Dementia Risk Stratification
- Ultrasensitive Test Detects Key Biomarker of Frontotemporal Dementia Subtype
- Routine Blood Tests Years Before Pregnancy Could Identify Preeclampsia Risk
- Blood Test Detects Testicular Cancer Missed by Standard Markers
- Routine Blood Tests Identify Biomarkers Linked to PTSD
- Proteomic Data Underscore Need for Age-Specific Pediatric Reference Ranges
- Routine Blood Count Ratio Linked to Future Alzheimer’s and Dementia Risk
- Label-Free Microfluidic Device Enriches Tumor Cells and Clusters from Pleural Effusions
- Rapid Biosensor Detects Pancreatic Cancer Biomarker for Early Detection
- Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
- Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
- Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
- AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
- Next Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
- Blood Metabolite Test Detects Early Cognitive Decline
Channels
Molecular Diagnostics
view channel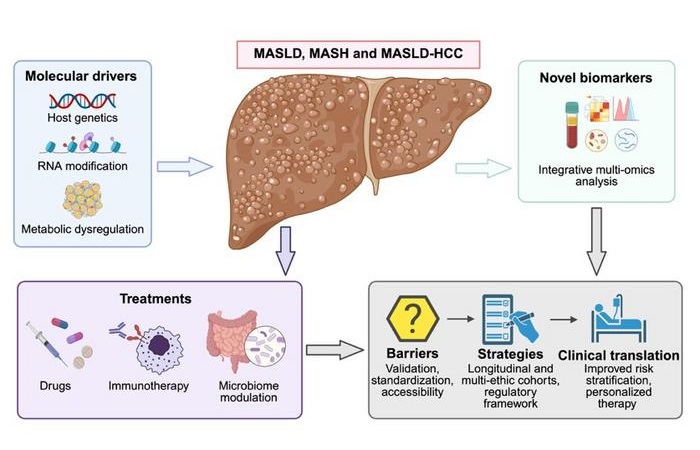
Emerging Biomarkers Advance Early Detection of MASLD and Liver Cancer Risk
Metabolic dysfunction-associated steatotic liver disease (MASLD) affects about 30% of people worldwide and can advance to metabolic dysfunction-associated steatohepatitis (MASH), fibrosis, cirrhosis, and... Read more
Patented Isothermal Amplification Chemistry Advances Decentralized Testing
Molecular diagnostics offer high sensitivity for pathogen detection but typically rely on thermal cycling and specialized instruments, limiting their use outside centralized laboratories.... Read more
Finger-Prick Blood Test Aids Early Tuberculosis Detection and Risk Stratification
Household contacts of people with tuberculosis face an estimated 2% risk of developing disease, yet most are asymptomatic at the time of screening. Early-stage cases are often missed because symptom checks... Read more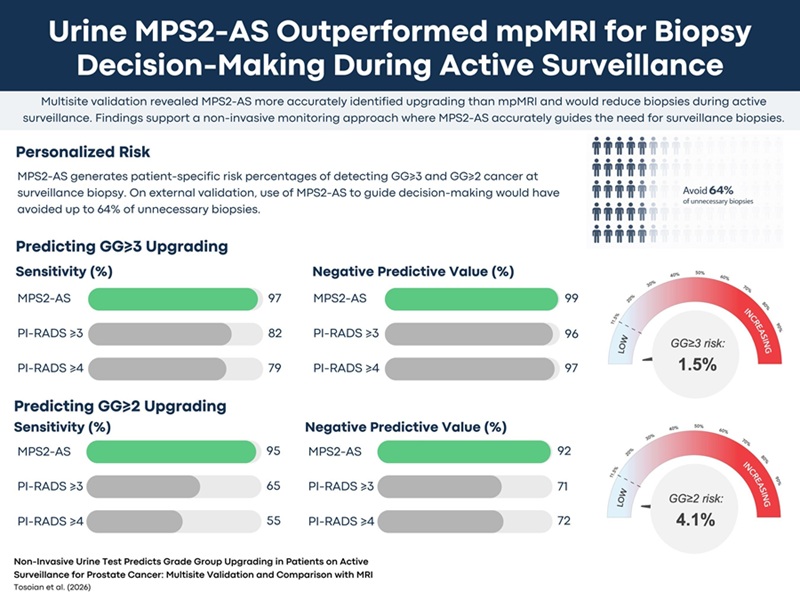
Urine Test Beats MRI in Identifying Prostate Cancer Upgrading During Active Surveillance
Active surveillance is common for men with low-risk prostate cancer, yet deciding when to repeat biopsy remains challenging. Prostate-specific antigen (PSA) testing and magnetic resonance imaging (MRI)... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read more
Point-of-Care Tests Could Expand Access to Mpox Diagnosis
Mpox outbreaks in non-endemic regions have underscored the need for rapid, accessible diagnostics to limit transmission. Polymerase chain reaction (PCR) remains the clinical reference, yet it depends on... Read moreMicrobiology
view channel
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read more
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read more
Tumor-on-a-Chip Platform Models Pancreatic Cancer Treatment Response
Pancreatic cancer remains one of the hardest malignancies to treat because tumors are embedded within a dense microenvironment that shapes growth and therapy response. Standard laboratory models often... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more





















