ddPCR Developed for BCR-ABL1 Fusion Transcript in B-Lymphoblastic Leukemia
|
By LabMedica International staff writers Posted on 12 Jan 2022 |
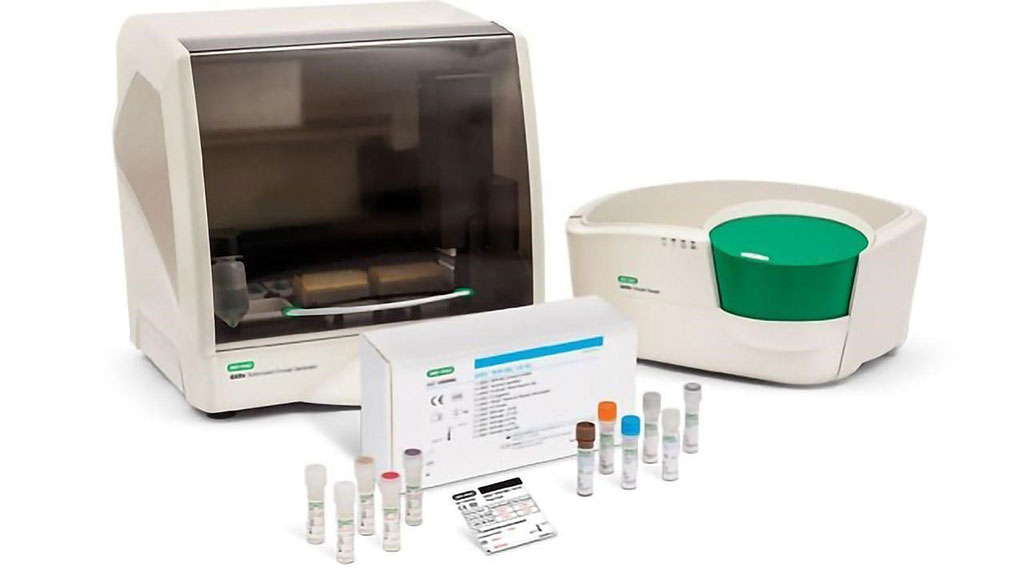
Image: QX200 AutoDG ddPCR System and QXDx BCR-ABL %IS Kit (Photo courtesy of Bio-Rad)
Droplet digital polymerase chain reaction (ddPCR) is a novel polymerase chain reaction (PCR) technique reliant on massive sample partitioning to generate individual reaction chambers (droplets) in which individual amplification targets are detected.
Standardized measurements of BCR-ABL1 p210 translocation by RT-qPCR assays for chronic myelogenous leukemia have proved challenging because of various sources of error including processing of specimens, reference gene selection, the reverse transcriptase reaction, and quantification of transcripts.
Medical Scientists at the University of Minnesota (Minneapolis, MN, USA) obtained clinical samples from specimens from patients who had been previously tested for BCR-ABL1 p190 transcript in the molecular diagnostics laboratory. Samples consisted of both bone marrow and peripheral blood. All samples were stored at −80 °C. The scientists’ aim was to compare results of ddPCR and RT-qPCR BCR-ABL1 fusion transcript measurements of patient samples and determine if either method is superior.
RNA was isolated with the Maxwell RSC simplyRNA Blood Kit and Maxwell RCS instrument (Promega, Madison, WI, USA). The clinical RT-qPCR assay was performed using the TaqMan Reverse Transcription and TaqMan Universal PCR Master Mix (Applied Biosystems, Waltham, MA, USA). Droplet digital PCR was performed using the One-Step RT-ddPCR Advanced Kit for Probes and for automated studies, the QX200 AutoDG system (Bio-Rad Laboratories, Hercules, CA, USA). The PCR was performed in a Bio-Rad C1000 Touch thermal cycler.
The investigators reported that droplet digital polymerase chain reaction was able to detect the BCR-ABL1 p190 transcript to 0.001% (1:10−5) with a calculated limit of detection and limit of quantitation of 4.1 and 5.3 transcripts, respectively. When tested on patient samples, ddPCR was able to identify 20% more positives than a laboratory-developed 2-step RT-qPCR assay. Of the 101 samples measured, 65 samples with a previously positive p190 transcript and sufficient RNA for analysis were further analyzed. Thirty-five of 65 samples (54%) were measured as positive by both methods, and the BCR-ABL1:ABL1 ratio was compared. The majority of samples showed concordant results between the two methods, but several samples demonstrated discordant quantification.
The authors concluded that droplet digital polymerase chain reaction demonstrated increased detection of BCR-ABL1 compared with RT-qPCR. Improved detection of BCR-ABL1 p190 and the potential for improved standardization across multiple laboratories makes ddPCR a suitable method for disease monitoring in patients with acute B-lymphoblastic leukemia. The study was published in the January, 2022 issue of the journal Archives of Pathology and Laboratory Medicine.
Related Links:
University of Minnesota
Promega
Applied Biosystems
Bio-Rad Laboratories
Standardized measurements of BCR-ABL1 p210 translocation by RT-qPCR assays for chronic myelogenous leukemia have proved challenging because of various sources of error including processing of specimens, reference gene selection, the reverse transcriptase reaction, and quantification of transcripts.
Medical Scientists at the University of Minnesota (Minneapolis, MN, USA) obtained clinical samples from specimens from patients who had been previously tested for BCR-ABL1 p190 transcript in the molecular diagnostics laboratory. Samples consisted of both bone marrow and peripheral blood. All samples were stored at −80 °C. The scientists’ aim was to compare results of ddPCR and RT-qPCR BCR-ABL1 fusion transcript measurements of patient samples and determine if either method is superior.
RNA was isolated with the Maxwell RSC simplyRNA Blood Kit and Maxwell RCS instrument (Promega, Madison, WI, USA). The clinical RT-qPCR assay was performed using the TaqMan Reverse Transcription and TaqMan Universal PCR Master Mix (Applied Biosystems, Waltham, MA, USA). Droplet digital PCR was performed using the One-Step RT-ddPCR Advanced Kit for Probes and for automated studies, the QX200 AutoDG system (Bio-Rad Laboratories, Hercules, CA, USA). The PCR was performed in a Bio-Rad C1000 Touch thermal cycler.
The investigators reported that droplet digital polymerase chain reaction was able to detect the BCR-ABL1 p190 transcript to 0.001% (1:10−5) with a calculated limit of detection and limit of quantitation of 4.1 and 5.3 transcripts, respectively. When tested on patient samples, ddPCR was able to identify 20% more positives than a laboratory-developed 2-step RT-qPCR assay. Of the 101 samples measured, 65 samples with a previously positive p190 transcript and sufficient RNA for analysis were further analyzed. Thirty-five of 65 samples (54%) were measured as positive by both methods, and the BCR-ABL1:ABL1 ratio was compared. The majority of samples showed concordant results between the two methods, but several samples demonstrated discordant quantification.
The authors concluded that droplet digital polymerase chain reaction demonstrated increased detection of BCR-ABL1 compared with RT-qPCR. Improved detection of BCR-ABL1 p190 and the potential for improved standardization across multiple laboratories makes ddPCR a suitable method for disease monitoring in patients with acute B-lymphoblastic leukemia. The study was published in the January, 2022 issue of the journal Archives of Pathology and Laboratory Medicine.
Related Links:
University of Minnesota
Promega
Applied Biosystems
Bio-Rad Laboratories
Latest Molecular Diagnostics News
- cfDNA Testing Reduces Pregnancy Risks
- Non-Invasive Biosensor Facilitates Early Kidney Disease Detection
- New High-Sensitivity Cardiac Troponin Test Quickly Rules Out Heart Attack
- Gene Technology Outperforms Standard Newborn Screening Tests in Pioneering Study
- Maternal Blood Test Identifies Congenital Heart Diseases in Fetus
- Enhanced Ultra-Sensitive Protocol Detects Parkinson’s Disease Proteins in Extracellular Vesicles in Blood
- New RNA Molecules Can Help Predict Bowel Cancer Recurrence
- Respiratory Panel to Help Clinicians Make Precise Treatment Decisions in Outpatient Settings
- Integrating Cardiovascular Risk Biomarkers Aids in Detection of ‘Inflammaging’
- Genetic Signature in Newborns Predicts Neonatal Sepsis Before Symptoms Appear
- Integrating Multiple Protein Markers Predicts Health Outcomes in Chronic Kidney Disease Patients
- Rapid Finger Prick Blood Test to Detect Active Syphilis at Point of Care
- Urine Tests Could Reveal Early Signs of Cancer and Other Diseases
- AI-Powered Smart PCR System to Revolutionize Clinical Diagnostics
- Simple Blood Test Identifies Women in Labor at Risk for Preeclampsia
- Point-Of-Care Paper-Based Test Could Diagnose Cancer at Bedside
Channels
Clinical Chemistry
view channel.jpg)
POC Saliva Testing Device Predicts Heart Failure in 15 Minutes
Heart failure is a serious condition where the heart muscle is unable to pump sufficient oxygen-rich blood throughout the body. It ranks as a major cause of death globally and is particularly fatal for... Read more
Screening Tool Detects Multiple Health Conditions from Single Blood Drop
Infrared spectroscopy, a method using infrared light to study the molecular composition of substances, has been a foundational tool in chemistry for decades, functioning similarly to a molecular fingerprinting... Read more
Integrated Chemistry and Immunoassay Analyzer with Extensive Assay Menu Offers Flexibility, Scalability and Data Commutability
As global healthcare systems increasingly shift towards networked laboratory operational models to enhance efficiency and patient access, there is a greater need for innovative solutions tailored to the... Read moreMolecular Diagnostics
view channel
cfDNA Testing Reduces Pregnancy Risks
The highly anticipated emergence of "precision medicine" promises customized technologies that can benefit individuals while potentially lowering healthcare costs. Now, new research suggests that pregnancy... Read more
Non-Invasive Biosensor Facilitates Early Kidney Disease Detection
Traditionally, kidney function has been assessed by measuring blood creatinine levels, which reflect muscle breakdown. Elevated creatinine levels may indicate that the kidneys are not effectively filtering waste.... Read moreImmunology
view channel.jpg)
Advanced Imaging Method Maps Immune Cell Connections to Predict Cancer Patients Survival
A growing tumor is influenced not only by the tumor cells themselves but also by the surrounding tissue, which alters its biology. Immune cells communicate by transferring vital signaling proteins to their... Read more
Computational Tool Predicts Immunotherapy Outcomes for Metastatic Breast Cancer Patients
Immunotherapy aims to enhance the body’s immune response to target cancer cells, but not all patients experience a positive reaction to such treatments. Identifying which patients will benefit from immunotherapy... Read more
Biomarker Could Predict Immunotherapy Response in Liver Cancer
Until recently, patients diagnosed with hepatocellular carcinoma had limited treatment options, with existing therapies extending life by only a few months. Immunotherapy has emerged as a new alternative... Read more
Epigenetic Test Could Determine Efficacy of New Immunotherapy Treatments Against Multiple Myeloma
Multiple myeloma is a blood cancer that primarily affects individuals over the age of sixty, and its occurrence rises as the population ages. In this disease, the bone marrow—the spongy tissue inside bones... Read moreMicrobiology
view channel
High-Accuracy Bedside Test to Diagnose Periprosthetic Joint Infection in Five Minutes
Periprosthetic joint infection (PJI) represents a significant global issue that is worsening as the number of joint replacements increases due to aging populations. In the United States alone, the anticipated... Read more_1.jpg)
Innovative Diagnostic Approach for Bacterial Infections to Enable Faster and Effective Treatment
For patients with bacterial infections, timely treatment with the appropriate antibiotics significantly improves their chances of recovery. Current methods for identifying which antibiotics will be effective... Read more
Non-Invasive Stool Test to Diagnose Endometriosis and Help Reduce Disease Progression
Endometriosis, a painful condition impacting nearly 200 million women globally, occurs when tissue similar to the lining of the uterus grows outside its usual location, such as on the intestines or the... Read more
Automated Positive Blood Culture Sample Preparation Platform Designed to Fight Against Sepsis and AMR
Delayed administration of antibiotics to patients with bloodstream infections significantly increases the risk of morbidity and mortality. For optimal therapeutic outcomes, it is crucial to rapidly identify... Read morePathology
view channel
AI Tool Uses Imaging Data to Detect Less Frequent GI Diseases
Artificial intelligence (AI) is already being utilized in various medical fields, demonstrating significant potential in aiding doctors in diagnosing diseases through imaging data. However, training AI... Read moreAI-Based Method Shows Promise for Pathological Diagnosis of Hereditary Kidney Diseases
Alport syndrome is a hereditary kidney disorder characterized by kidney dysfunction, sensorineural hearing loss, and ocular abnormalities. Early in the disease, patients experience hematuria, which is... Read moreTechnology
view channel
New Noninvasive Methods Detect Lead Exposure Faster, Easier and More Accurately at POC
Exposure to lead can negatively affect health in multiple ways, leading to damage in the brain and central nervous system, delays in development and growth, learning and behavioral issues, problems with... Read more
Noninvasive Test Detects Malaria Without Blood Sample
Malaria remains a significant global health issue, with approximately 250 million cases and over 600,000 deaths reported annually. Nearly half of the world's population is at risk for malaria infection,... Read moreIndustry
view channel
Microbiologics Acquires Diagnostic Quality Controls Manufacturer SensID
Microbiologics (St. Cloud, MN, USA), a biotechnology company specializing in infectious disease reference materials and contract research services, has acquired SensID (Rostock, Germany), a manufacturer... Read more
















_1.jpg)