Elevated PlGF Levels Found with Acute Fatty Liver of Pregnancy
|
By LabMedica International staff writers Posted on 22 Jul 2021 |

Image: The ROCHE Cobas 6000 Chemistry Analyzer (Photo courtesy of Roche Diagnostics)
Acute fatty liver of pregnancy (AFLP) is a severe liver disorder unique to pregnancy, typically occurring after 30 weeks’ gestation. Although uncommon, with an estimated global incidence of 1 in 7,000 to 15,000 pregnancies, AFLP is a life-threatening disease for both mother and child.
During preeclampsia, poor placentation triggers the excessive release of the soluble vascular endothelial growth factor receptors (VEGFR); also known as soluble Fms-like tyrosine kinase-1 (sFlt-1), which binds its free circulating ligands VEGF and placental growth factor (PlGF). The ensuing angiogenic imbalance is thought to contribute significantly to the clinical manifestations of this disorder.
Physicians at the Erasmus MC University Hospital (Rotterdam, the Netherlands) and their colleagues utilized human serum samples from a database of women with singleton pregnancies who had a clinical diagnosis of AFLP between 2005 and 2020. The team compared matched women with AFLP to 12 gestational age (GA) matched women with either no preeclampsia, confirmed preeclampsia, or HELLP syndrome, given that the values of sFlt-1, free, and total PlGF alter with advancing gestation.
Measurement of sFlt-1, Free PlGF, and Total PlGF was carried out. For the thermal dissociation of all sFlt-1-PlGF complexes, serum samples were placed in a heating block at 70 °C for 10 minutes. Measurements of sFlt-1 and PlGF before and after heating were performed using the automated Elecsys immunoassay from Roche Diagnostics (Cobas 6000, e-module; Rotterdam, the Netherlands).
The scientist found that median levels of free PlGF were significantly lower in 13 women with preeclampsia (117pg/mL) or in 12 women with hemolysis elevated liver enzymes and low platelet count syndrome (59 pg/mL) compared with 11 women without preeclampsia (349 pg/mL). In contrast, median total PlGF did not differ between women with no preeclampsia, preeclampsia, and hemolysis elevated liver enzymes and low platelet count syndrome (354 pg/mL versus 435 pg/mL versus 344 pg/mL) respectively, whereas it was markedly elevated in AFLP compared with all groups (2,054 pg/mL). Furthermore, in AFLP, both sFlt-1 and total PlGF declined rapidly post-delivery, with significantly higher in 12 pre-delivery total PlGF (median, 2,054 pg/mL) than 14 postpartum levels (median, 163 pg/mL), suggesting that in AFLP, PlGF is largely placenta-derived.
The authors concluded that their findings indicated that like sFlt-1, PlGF production is significantly upregulated in AFLP, mainly originating from the placenta. Importantly, total PlGF can now be easily calculated from already available free PlGF and sFlt-1 levels, allowing subsequent evaluation of other groups in whom PlGF is altered. The study was published on June 28, 2021in the journal Hypertension.
Related Links:
Erasmus MC University Hospital
Roche Diagnostics
During preeclampsia, poor placentation triggers the excessive release of the soluble vascular endothelial growth factor receptors (VEGFR); also known as soluble Fms-like tyrosine kinase-1 (sFlt-1), which binds its free circulating ligands VEGF and placental growth factor (PlGF). The ensuing angiogenic imbalance is thought to contribute significantly to the clinical manifestations of this disorder.
Physicians at the Erasmus MC University Hospital (Rotterdam, the Netherlands) and their colleagues utilized human serum samples from a database of women with singleton pregnancies who had a clinical diagnosis of AFLP between 2005 and 2020. The team compared matched women with AFLP to 12 gestational age (GA) matched women with either no preeclampsia, confirmed preeclampsia, or HELLP syndrome, given that the values of sFlt-1, free, and total PlGF alter with advancing gestation.
Measurement of sFlt-1, Free PlGF, and Total PlGF was carried out. For the thermal dissociation of all sFlt-1-PlGF complexes, serum samples were placed in a heating block at 70 °C for 10 minutes. Measurements of sFlt-1 and PlGF before and after heating were performed using the automated Elecsys immunoassay from Roche Diagnostics (Cobas 6000, e-module; Rotterdam, the Netherlands).
The scientist found that median levels of free PlGF were significantly lower in 13 women with preeclampsia (117pg/mL) or in 12 women with hemolysis elevated liver enzymes and low platelet count syndrome (59 pg/mL) compared with 11 women without preeclampsia (349 pg/mL). In contrast, median total PlGF did not differ between women with no preeclampsia, preeclampsia, and hemolysis elevated liver enzymes and low platelet count syndrome (354 pg/mL versus 435 pg/mL versus 344 pg/mL) respectively, whereas it was markedly elevated in AFLP compared with all groups (2,054 pg/mL). Furthermore, in AFLP, both sFlt-1 and total PlGF declined rapidly post-delivery, with significantly higher in 12 pre-delivery total PlGF (median, 2,054 pg/mL) than 14 postpartum levels (median, 163 pg/mL), suggesting that in AFLP, PlGF is largely placenta-derived.
The authors concluded that their findings indicated that like sFlt-1, PlGF production is significantly upregulated in AFLP, mainly originating from the placenta. Importantly, total PlGF can now be easily calculated from already available free PlGF and sFlt-1 levels, allowing subsequent evaluation of other groups in whom PlGF is altered. The study was published on June 28, 2021in the journal Hypertension.
Related Links:
Erasmus MC University Hospital
Roche Diagnostics
Latest Clinical Chem. News
- 3D Printed Point-Of-Care Mass Spectrometer Outperforms State-Of-The-Art Models
- POC Biomedical Test Spins Water Droplet Using Sound Waves for Cancer Detection
- Highly Reliable Cell-Based Assay Enables Accurate Diagnosis of Endocrine Diseases
- New Blood Testing Method Detects Potent Opioids in Under Three Minutes
- Wireless Hepatitis B Test Kit Completes Screening and Data Collection in One Step
- Pain-Free, Low-Cost, Sensitive, Radiation-Free Device Detects Breast Cancer in Urine
- Spit Test Detects Breast Cancer in Five Seconds
- Electrochemical Sensors with Next-Generation Coating Advances Precision Diagnostics at POC
- First-Of-Its-Kind Handheld Device Accurately Detects Fentanyl in Urine within Seconds
- New Fluorescent Sensor Array Lights up Alzheimer’s-Related Proteins for Earlier Detection
- Automated Mass Spectrometry-Based Clinical Analyzer Could Transform Lab Testing
- Highly Sensitive pH Sensor to Aid Detection of Cancers and Vector-Borne Viruses
- Non-Invasive Sensor Monitors Changes in Saliva Compositions to Rapidly Diagnose Diabetes
- Breakthrough Immunoassays to Aid in Risk Assessment of Preeclampsia
- Urine Test for Monitoring Changes in Kidney Health Markers Can Predict New-Onset Heart Failure
- AACC Releases Comprehensive Diabetes Testing Guidelines
Channels
Molecular Diagnostics
view channel
Urine Test to Revolutionize Lyme Disease Testing
Lyme disease is the most common animal-to-human transmitted disease in the United States, with around 476,000 people diagnosed and treated annually, and its incidence has been increasing.... Read more
Simple Blood Test Could Enable First Quantitative Assessments for Future Cerebrovascular Disease
Cerebral small vessel disease is a common cause of stroke and cognitive decline, particularly in the elderly. Presently, assessing the risk for cerebral vascular diseases involves using a mix of diagnostic... Read more
New Genetic Testing Procedure Combined With Ultrasound Detects High Cardiovascular Risk
A key interest area in cardiovascular research today is the impact of clonal hematopoiesis on cardiovascular diseases. Clonal hematopoiesis results from mutations in hematopoietic stem cells and may lead... Read moreHematology
view channel
Next Generation Instrument Screens for Hemoglobin Disorders in Newborns
Hemoglobinopathies, the most widespread inherited conditions globally, affect about 7% of the population as carriers, with 2.7% of newborns being born with these conditions. The spectrum of clinical manifestations... Read more
First 4-in-1 Nucleic Acid Test for Arbovirus Screening to Reduce Risk of Transfusion-Transmitted Infections
Arboviruses represent an emerging global health threat, exacerbated by climate change and increased international travel that is facilitating their spread across new regions. Chikungunya, dengue, West... Read more
POC Finger-Prick Blood Test Determines Risk of Neutropenic Sepsis in Patients Undergoing Chemotherapy
Neutropenia, a decrease in neutrophils (a type of white blood cell crucial for fighting infections), is a frequent side effect of certain cancer treatments. This condition elevates the risk of infections,... Read more
First Affordable and Rapid Test for Beta Thalassemia Demonstrates 99% Diagnostic Accuracy
Hemoglobin disorders rank as some of the most prevalent monogenic diseases globally. Among various hemoglobin disorders, beta thalassemia, a hereditary blood disorder, affects about 1.5% of the world's... Read moreImmunology
view channel
Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
Transplanted organs constantly face the risk of being rejected by the recipient's immune system which differentiates self from non-self using T cells and B cells. T cells are commonly associated with acute... Read more
AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
Current strategies for matching cancer patients with specific treatments often depend on bulk sequencing of tumor DNA and RNA, which provides an average profile from all cells within a tumor sample.... Read more
Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
Cancer treatment typically adheres to a standard of care—established, statistically validated regimens that are effective for the majority of patients. However, the disease’s inherent variability means... Read moreMicrobiology
view channelEnhanced Rapid Syndromic Molecular Diagnostic Solution Detects Broad Range of Infectious Diseases
GenMark Diagnostics (Carlsbad, CA, USA), a member of the Roche Group (Basel, Switzerland), has rebranded its ePlex® system as the cobas eplex system. This rebranding under the globally renowned cobas name... Read more
Clinical Decision Support Software a Game-Changer in Antimicrobial Resistance Battle
Antimicrobial resistance (AMR) is a serious global public health concern that claims millions of lives every year. It primarily results from the inappropriate and excessive use of antibiotics, which reduces... Read more
New CE-Marked Hepatitis Assays to Help Diagnose Infections Earlier
According to the World Health Organization (WHO), an estimated 354 million individuals globally are afflicted with chronic hepatitis B or C. These viruses are the leading causes of liver cirrhosis, liver... Read more
1 Hour, Direct-From-Blood Multiplex PCR Test Identifies 95% of Sepsis-Causing Pathogens
Sepsis contributes to one in every three hospital deaths in the US, and globally, septic shock carries a mortality rate of 30-40%. Diagnosing sepsis early is challenging due to its non-specific symptoms... Read morePathology
view channel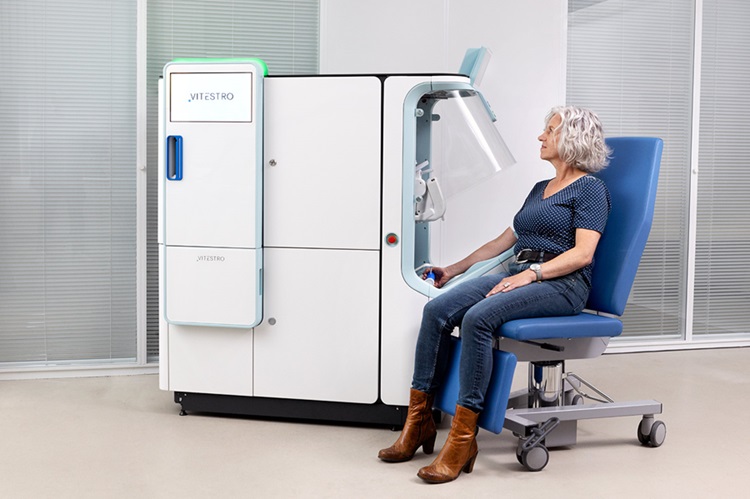
Robotic Blood Drawing Device to Revolutionize Sample Collection for Diagnostic Testing
Blood drawing is performed billions of times each year worldwide, playing a critical role in diagnostic procedures. Despite its importance, clinical laboratories are dealing with significant staff shortages,... Read more.jpg)
Use of DICOM Images for Pathology Diagnostics Marks Significant Step towards Standardization
Digital pathology is rapidly becoming a key aspect of modern healthcare, transforming the practice of pathology as laboratories worldwide adopt this advanced technology. Digital pathology systems allow... Read more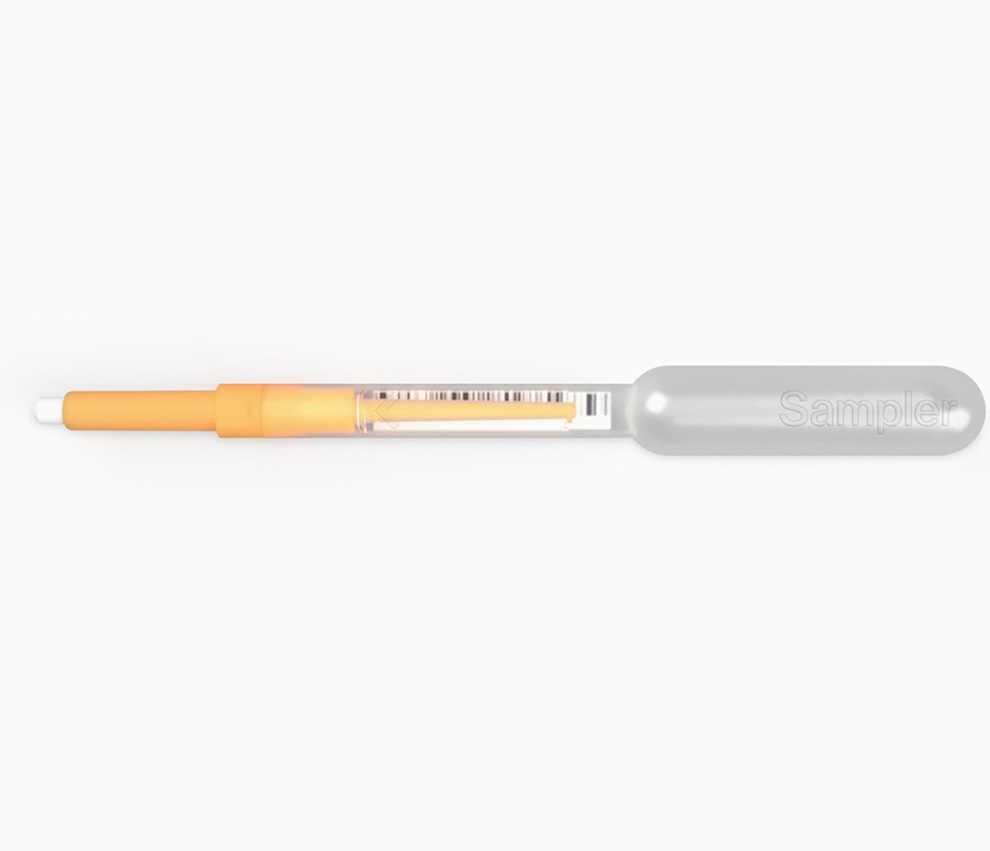
First of Its Kind Universal Tool to Revolutionize Sample Collection for Diagnostic Tests
The COVID pandemic has dramatically reshaped the perception of diagnostics. Post the pandemic, a groundbreaking device that combines sample collection and processing into a single, easy-to-use disposable... Read moreTechnology
view channel
New Diagnostic System Achieves PCR Testing Accuracy
While PCR tests are the gold standard of accuracy for virology testing, they come with limitations such as complexity, the need for skilled lab operators, and longer result times. They also require complex... Read more
DNA Biosensor Enables Early Diagnosis of Cervical Cancer
Molybdenum disulfide (MoS2), recognized for its potential to form two-dimensional nanosheets like graphene, is a material that's increasingly catching the eye of the scientific community.... Read more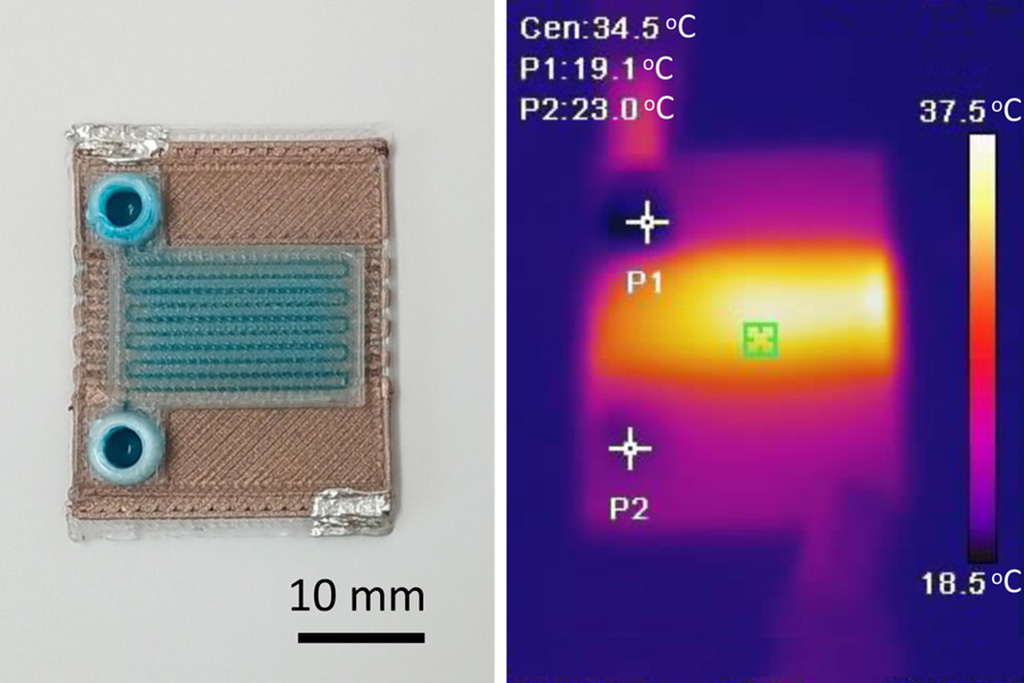
Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
Microfluidics, which are miniature devices that control the flow of liquids and facilitate chemical reactions, play a key role in disease detection from small samples of blood or other fluids.... Read more
Breakthrough in Diagnostic Technology Could Make On-The-Spot Testing Widely Accessible
Home testing gained significant importance during the COVID-19 pandemic, yet the availability of rapid tests is limited, and most of them can only drive one liquid across the strip, leading to continued... Read moreIndustry
view channel_1.jpg)
Thermo Fisher and Bio-Techne Enter Into Strategic Distribution Agreement for Europe
Thermo Fisher Scientific (Waltham, MA USA) has entered into a strategic distribution agreement with Bio-Techne Corporation (Minneapolis, MN, USA), resulting in a significant collaboration between two industry... Read more
ECCMID Congress Name Changes to ESCMID Global
Over the last few years, the European Society of Clinical Microbiology and Infectious Diseases (ESCMID, Basel, Switzerland) has evolved remarkably. The society is now stronger and broader than ever before... Read more