Next-Gen POC Whole Blood Hemostasis System Recognizes Specific Needs of EDs and ORs 
|
By LabMedica International staff writers Posted on 01 Nov 2022 |
Current hemostatic tests provide only a subset of needed information, or take too long to be useful in critical bleeding situations, forcing clinicians to use iterative transfusion protocols that do not account for the patient’s actual coagulation status. This approach leads to over-transfusion and transfusion of inappropriate products. Up to 60% of red blood cell (RBC) transfusions lack clinical justification. Further, blood products are costly, making up about 1-2% of a hospital’s budget. Hospitals around the world are thus seeking ways to reduce their transfusion rates to improve outcomes and reduce costs.
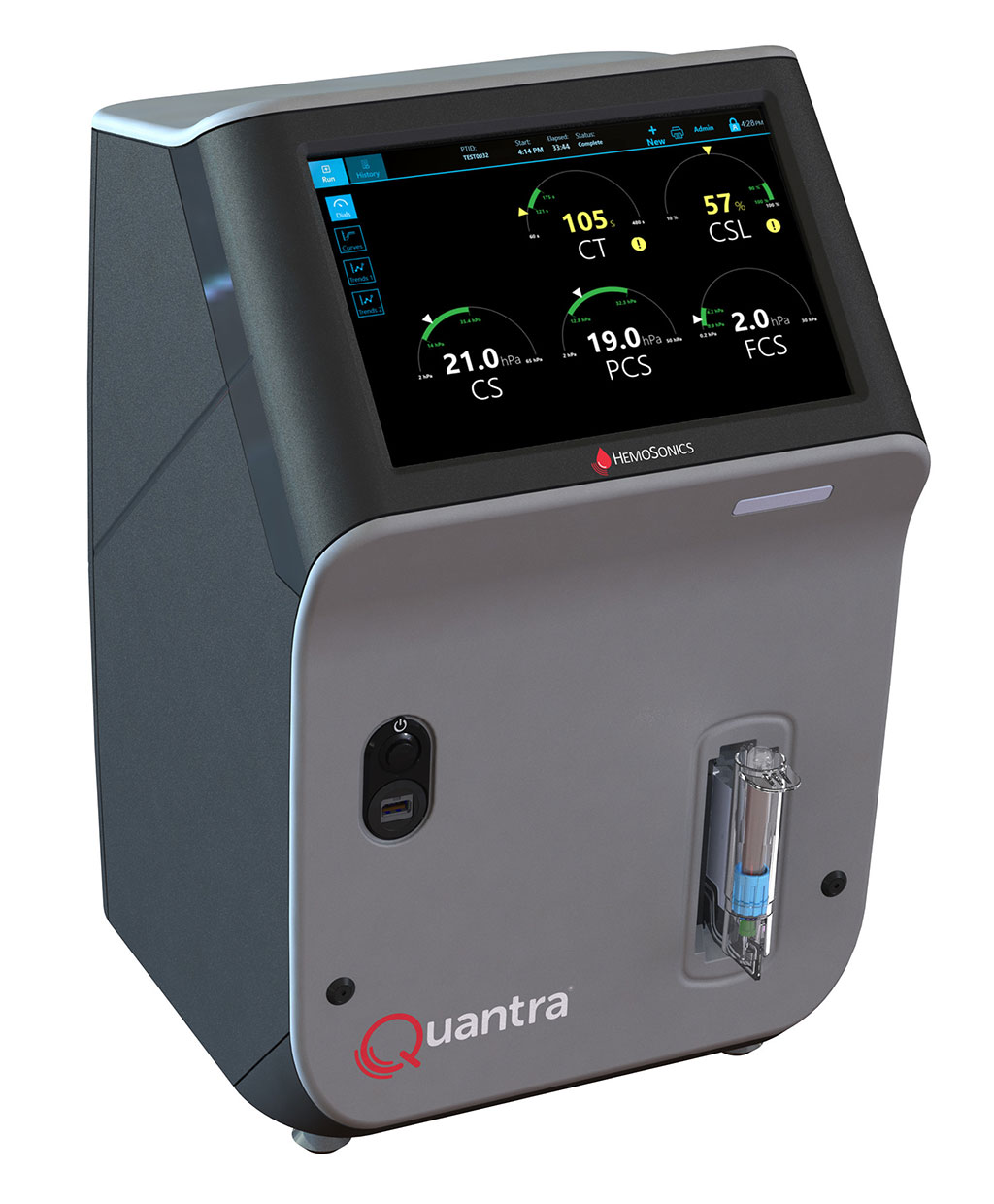
Now, a next-generation point-of-care (POC) hemostasis system leverages innovative medical-grade ultrasound technology to measure the coagulation properties of a whole blood sample. The system enables clinicians to deliver patient-centered coagulation therapy that improves care and optimizes blood product usage. With its rapid results, ease of operation, and dials screen for straightforward interpretation, it is the only whole blood hemostasis testing system specifically cleared for use in POC settings, such as operating rooms, emergency departments, and intensive care units.
HemoSonics, LLC’s (Durham, NC, USA) innovative Quantra hemostasis analyzer platform, based on patented SEER sonorheometry, enables fully automated, rapid point-of-care viscoelastic testing and gives physicians the results they need at the POC to make more informed decisions. SEER uses state-of-the-art ultrasound technology to measure clot stiffness (shear modulus of elasticity) over time using ultrasound-induced resonance. The Quantra system’s innovative closed-cartridge design requires no open-tube blood manipulation after sample collection and it automatically runs internal QC every eight hours and with every test. The Quantra system’s innovative ultrasound technology, proprietary closed-cartridge design, unparalleled ease of use, and ease of interpretation make it uniquely suited for use in critical or emergency care settings.
HemoSonics has now received 510(k) market clearance from the U.S. Food and Drug Administration (FDA) for the Quantra Hemostasis System with QStat Cartridge. The FDA clearance of the QStat Cartridge expands the Quantra System's indications for use to include trauma, and liver transplantation procedures. The QStat Cartridge assay, in addition to the System's already established QPlus Cartridge assay, increases Quantra's overall diagnostic capabilities to now cover the broadest range of clinical indications of any point-of-care hemostasis analyzer available in the U.S. market today.
"The Quantra Hemostasis System with QStat Cartridge is breaking new ground and leading innovation in the point-of-care and laboratory-based whole blood hemostasis testing market. Today's clearance significantly expands the clinical indications of the Quantra System, creating the greatest opportunity to address the critical unmet clinical needs of our healthcare provider partners and the patients that we serve," said Robert Roda, President, and Chief Executive Officer of HemoSonics.
"Point-of-care data is the answer to PBM-guided patient decisions. It is a triple win; Patients get improved outcomes, public health is improved, and cost savings are achieved," said Dr. Bruce Spiess, Medical Director of HemoSonics. "The Quantra Hemostasis System with QStat and QPlus Cartridges will assist more clinicians in determining which specific blood products are needed to rapidly treat individual patients. It has the potential to positively impact patient outcomes for hundreds of thousands of trauma patients and thousands of liver transplant recipients each year by optimizing blood product usage and conserving critically low blood supplies."
Related Links:
HemoSonics, LLC
Latest Point of Care News
Channels
Clinical Chemistry
view channel
Urine-Based Test Shows Promise for Autism Screening in Children
Autism spectrum disorder (ASD) is commonly diagnosed through behavioral assessments, which can involve long waits that delay intervention. Earlier identification is linked to better developmental outcomes,... Read more
Liquid Biopsy Biomarkers May Improve Childhood Epilepsy Diagnosis
Childhood epilepsy remains a major neurological disorder with unmet needs for accurate, non-invasive biomarkers, as conventional tests such as electroencephalography and neuroimaging can have limited sensitivity... Read moreMolecular Diagnostics
view channel
Blood-Based MRD Monitoring Supports Relapse Prevention in Leukemia
In myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML), molecular blood testing for minimal residual disease (MRD) can detect signs of impending relapse before clinical symptoms emerge.... Read more
Genomic Test Predicts Chemotherapy Benefit in Metastatic Prostate Cancer
Metastatic prostate cancer remains a major treatment challenge, particularly when deciding whether to add chemotherapy to hormonal therapy. In the United States, about 334,000 men are diagnosed with prostate... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channel
Study Points to Autoimmune Pathway Behind Long COVID Symptoms
Long COVID leaves many SARS-CoV-2 survivors with persistent fatigue, cognitive issues, palpitations, and musculoskeletal pain for months or years. Estimates cited in new research suggest 4%–20% of infected... Read more
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read morePathology
view channel
AI-Powered Atlas Maps Immune Structures Linked to Cancer Outcomes
Tertiary lymphoid structures are emerging as important indicators of antitumor immunity, but their heterogeneity and spatial context within tumors remain difficult to capture through routine diagnostics.... Read more
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channel