Repeatability and Reproducibility of a Handheld Quantitative G6PD Diagnostic
|
By LabMedica International staff writers Posted on 03 Mar 2022 |
Novel treatment regimens for the radical cure of P. vivax malaria are more effective than current options but require prior quantitative G6PD testing. The reference method for quantitative G6PD measurement is spectrophotometry but, due to its operational characteristics, is not suitable for routine use. Furthermore, poor inter-laboratory reproducibility of spectrophotometry has prevented quantitative global definitions of G6PD deficiency. SD Biosensor (ROK) have developed a novel handheld “Biosensor” device (G6PD STANDARD), which measures G6PD activity within two minutes and has operational characteristics suited to point of care diagnosis.
Approximately 400 million people worldwide are affected by glucose-6-phosphate dehydrogenase enzyme deficiency (G6PDd), with allele frequencies reaching up to 35% in malaria endemic areas. The introduction of novel short course treatment regimens for the radical cure of Plasmodium vivax requires reliable point-of-care diagnosis that can identify G6PD deficient individuals.
The G6PD gene is located on the X-chromosome; males are either hemizygous deficient or normal, whereas females are homozygous deficient, normal, or heterozygous for the gene. Heterozygous females have two distinct red blood cell (RBC) populations, G6PD normal and G6PD deficient that circulate in a ratio determined through the random process of lyonization Therefore, the G6PD activity levels of heterozygous females is dependent on the proportion of deficient cells, those cells at greatest risk of drug induced hemolysis.
An international team of medical scientists led by those at the Charles Darwin University (Darwin, Australia) determined the repeatability of a point-of-care assay (precision when repeated under constant conditions) and reproducibility (assay precision under different conditions, such as across devices, operators and sites), since robust performance of these characteristics is necessary for rolling-out universal thresholds for clinical decisions.
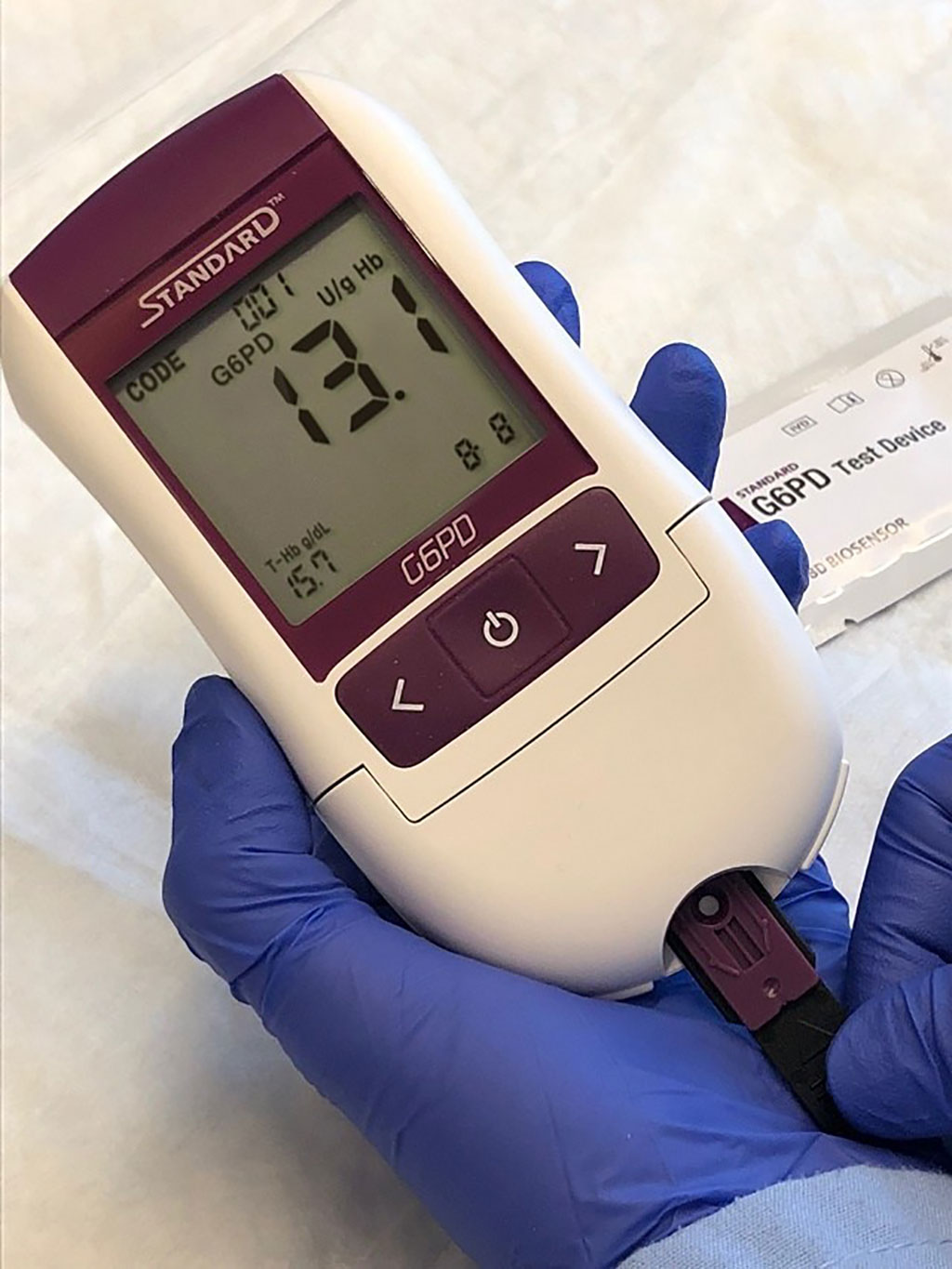
A hand-held quantitative G6PD diagnostic developed by SD Biosensor (STANDARD G6PD test, Suwon-si, Republic of Korea), and was tested in the study. The Biosensor device quantifies hemoglobin (Hb) concentration using a photo-reflectance based algorithm informed by the sample’s color intensity. This is measured on a separate spot to that for the G6PD activity. The handheld device displays G6PD activity (in U/gHb) and hemoglobin (Hb) levels (in g/dL) two minutes after applying the blood buffer solution, however the manufacturer indicates that results cannot be considered if Hb readings are equal to or below 7g/dL. To ensure that identical samples were tested across all sites, commercial controls were used with all controls within one phase being from the same lot (Analytical Control Systems, Inc., Fishers, IN; USA). Spectrophotometry was performed using kits from Pointe Scientific (Canton, MI, USA).
The investigators reported that when tested at one site, the mean coefficient of variation (CV) was 0.111, 0.172 and 0.260 for high, intermediate, and low controls across all devices respectively; combined G6PD Biosensor readings correlated well with spectrophotometry (rs = 0.859). When tested in different laboratories, correlation was lower (rs = 0.604) and G6PD activity determined by Biosensor for the low and intermediate controls overlapped. The use of lyophilized human blood samples rather than fresh blood may have affected these findings. Biosensor G6PD readings between sites did not differ significantly, whereas spectrophotometry readings differed markedly between sites.
The authors concluded that their findings suggest that the Biosensor offers reproducible quantitative diagnosis of G6PD status at the point-of-care in the hands of well-trained technicians. If repeatability and reproducibility as well as the previously reported accuracy are confirmed under real life conditions, the Biosensor has the potential to simplify access to effective radical cure of P. vivax malaria. The study was published on February 17, 2022 in the journal PLOS Neglected Tropical Diseases.
Related Links:
Charles Darwin University
SD Biosensor
Analytical Control Systems
Pointe Scientific
Latest Clinical Chem. News
- Automated NfL Assay Supports Monitoring of Neurological Disorders
- Blood-Based Screening Test Targets Early Detection of Colorectal Cancer
- CSF Biomarker Improves Diagnosis of Parkinson’s Disease and Lewy Body Dementia
- Simple Urine Home Test Kit Could Detect Early-Stage Breast Cancer
- New Tool Tracks Biomarker Changes to Predict Myeloma Progression
- New Plasma Tau Assay Improves Prediction of Alzheimer’s Progression
- First IVD Immunoassay to Detect Alzheimer’s Risk Gene Variant Receives CE Mark
- Routine Blood Markers Predict Heart Failure Risk in Prediabetes
- AI Model Enables Personalized Glucose Predictions for Type 1 Diabetes
- AI-Powered Blood Test Distinguishes Deadly Cardiac Events
- AI Sensor Detects Neurological Disorders Using Single Saliva Drop
- Blood Test Tracks Transplant Health Using Donor DNA
- New Blood Test Index Offers Earlier Detection of Liver Scarring
- Electronic Nose Smells Early Signs of Ovarian Cancer in Blood
- Simple Blood Test Offers New Path to Alzheimer’s Assessment in Primary Care
- Existing Hospital Analyzers Can Identify Fake Liquid Medical Products
Channels
Molecular Diagnostics
view channel
New Respiratory Panel Expands Pathogen Detection to 25 Targets
Respiratory infections often present with overlapping symptoms, complicating differential diagnosis in acute and community settings. The stakes are higher for older adults, young children, and people with... Read more
Simple Nasal Swab May Reveal Early Signs of Alzheimer’s Disease
Alzheimer’s disease affects millions worldwide but remains difficult to detect at its earliest, pre-symptomatic stage. Clinicians need tools that can identify biological changes before cognitive symptoms... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read more
Microfluidic Chip Detects Cancer Recurrence from Immune Response Signals
Early identification of treatment response and relapse remains a major challenge in solid tumors, where minimal residual disease is difficult to detect with routine imaging and blood tests.... Read moreMicrobiology
view channel
Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
Accurate and rapid identification of bacterial infections remains challenging in acute care, where delays can hinder timely, targeted therapy. Infectious diseases are a major cause of mortality worldwide,... Read more
Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
Direct-to-consumer gut microbiome kits promise personalized insights by profiling fecal bacteria and generating health readouts, but their analytical accuracy remains uncertain. A new study shows that... Read more
WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
Tuberculosis (TB) remains one of the world’s leading infectious disease killers, yet millions of cases go undiagnosed or are detected too late. Barriers such as reliance on sputum samples, limited laboratory... Read morePathology
view channel
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read more
AI-Powered Tool to Transform Dermatopathology Workflow
Skin cancer accounts for the largest number of cancer diagnoses in the United States, placing sustained pressure on pathology services. Diagnostic interpretation can be variable for challenging melanocytic... Read moreTechnology
view channel
Online Tool Supports Family Screening for Inherited Cancer Risk
Genetic test results in oncology often have implications for relatives who may share inherited cancer risk. Many health systems lack structured processes to help patients alert family members, limiting... Read more
Portable Breath Sensor Detects Pneumonia Biomarkers in Minutes
Pneumonia is commonly confirmed with chest X-rays or laboratory assays that can take hours, delaying clinical decisions in acute and outpatient settings. Breath-based diagnostics promise faster answers... Read moreIndustry
view channel
Integrated DNA Technologies Expands into Clinical Diagnostics
Integrated DNA Technologies (IDT; Coralville, Iowa, USA) has announced the launch of Archer FUSIONPlex-HT Dx and VARIANTPlex-HT Dx. This launch marks the company’s first in vitro diagnostic (IVD) offerings... Read more