Technopath Showcases Third Party Quality Controls and Quality Control Software Solutions at AACC 2021
|
By LabMedica International staff writers Posted on 28 Sep 2021 |
Technopath Clinical Diagnostics (Tipperary, Ireland) showcased its third party quality controls and quality control software solutions at the 2021 AACC Annual Scientific Meeting & Clinical Lab Expo held at the Georgia World Congress Center in Atlanta, September 26-30.
The event allowed more than 400 exhibiting companies to showcase the latest product innovations, emerging technologies, and research, and provided the opportunity to browse exhibitors in person for the solutions to lab related needs.
Technopath is one of the largest manufacturers of liquid-ready Quality Control globally in compliance with the highest quality standards. At AACC 2021, the company highlighted Multichem, its class leading third party consolidated Quality Control product range which incorporates a greater number of analytes to enable extensive test menu and control product consolidation. The Multichem IA+ control, for example, allows consolidation of the company’s competitors’ immunoassay, cardiac marker, tumor marker and specialty immunoassay controls into one control. This consolidation allows laboratories to simplify their inventory, reduce costs and improve efficiencies.
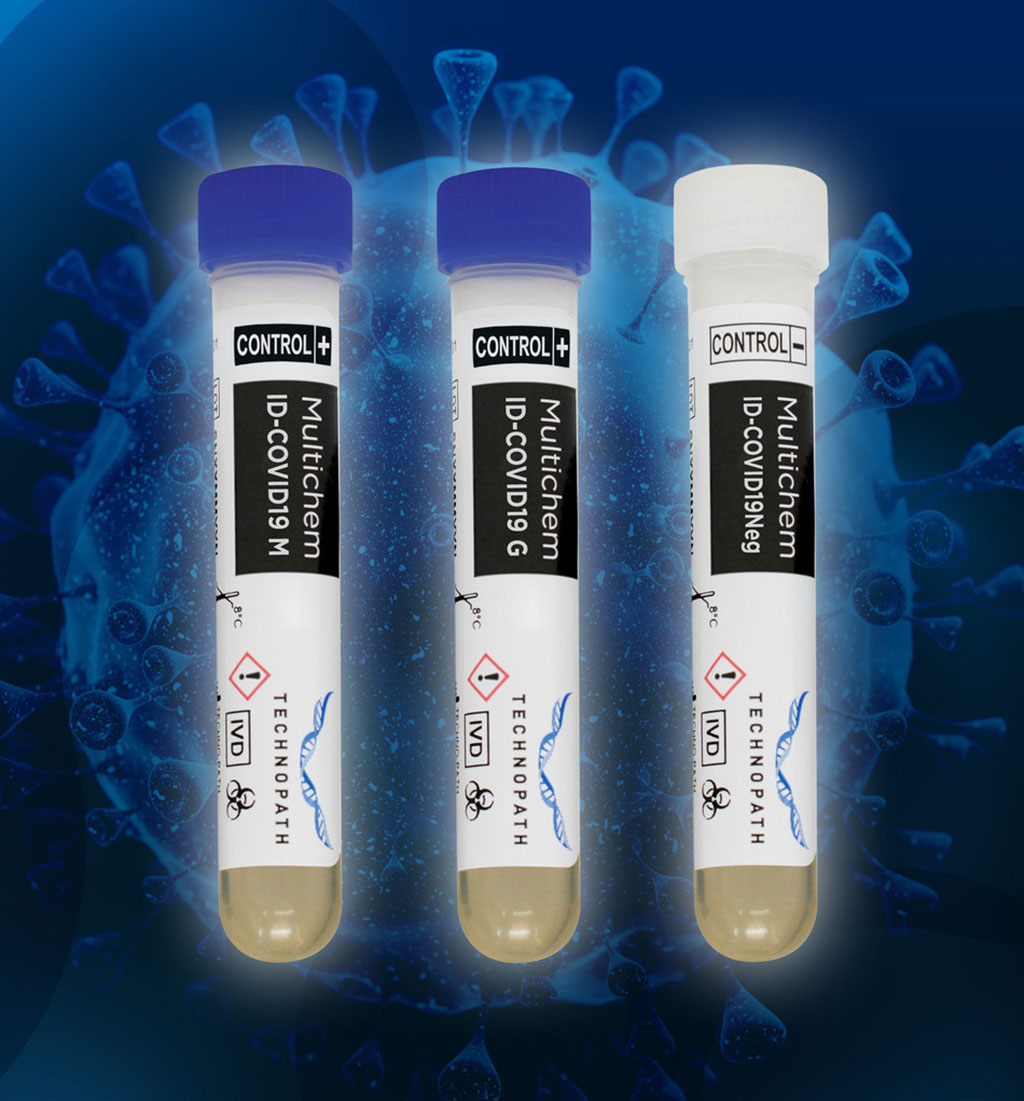
For instance, Multichem ID-COVID19 controls are the essential independent quality controls designed to test the precision of assays for antibodies IgG and IgM to SARS-CoV-2, to allow clinical laboratories to release patient results with confidence. The company also exhibited its flagship chemistry product, Multichem S Plus (S for Serum) which consolidates over 100 analytes in to a single product. Also on display was Technopath's leading diabetes product, Multichem A1c, which is a third party QC for hemoglobin A1c. Other products displayed at the event included flagship immunoassay product is Multichem IA Plus which consolidates over 90 analytes in to a single product and can replace up to four competitor products in the lab; and its leading urine chemistry control is Multichem U which provides a single product to cover the majority of standard urine chemistry assays, including Microalbumin, which some competitors provide a separate QC product for.
Alomgside its Multichem quality control product range, Technopath also demonstrated its IAMQC Software which provides laboratory managers and technologists with a range of QC software tools to analyze their QC results in real-time. IAMQC Software tools allow users to automate, centralize, standardize and improve QC processes in a laboratory setting. A combination of modules satisfies the varying levels of QC requirements in individual laboratories and are easily tailored to meet different QC management expectations.
Related Links:
Technopath Clinical Diagnostics
Latest Clinical Chem. News
- Automated NfL Assay Supports Monitoring of Neurological Disorders
- Blood-Based Screening Test Targets Early Detection of Colorectal Cancer
- New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
- CSF Biomarker Improves Diagnosis of Parkinson’s Disease and Lewy Body Dementia
- Simple Urine Home Test Kit Could Detect Early-Stage Breast Cancer
- Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
- New Tool Tracks Biomarker Changes to Predict Myeloma Progression
- New Plasma Tau Assay Improves Prediction of Alzheimer’s Progression
- First IVD Immunoassay to Detect Alzheimer’s Risk Gene Variant Receives CE Mark
- Routine Blood Markers Predict Heart Failure Risk in Prediabetes
- AI Model Enables Personalized Glucose Predictions for Type 1 Diabetes
- AI-Powered Blood Test Distinguishes Deadly Cardiac Events
- AI Sensor Detects Neurological Disorders Using Single Saliva Drop
- Blood Test Tracks Transplant Health Using Donor DNA
- New Blood Test Index Offers Earlier Detection of Liver Scarring
- Electronic Nose Smells Early Signs of Ovarian Cancer in Blood
Channels
Clinical Chemistry
view channel
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read more
Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
Alzheimer’s disease develops slowly, and biological changes can appear in blood many years before symptoms. While plasma assays for phosphorylated tau offer earlier detection, discerning whether these... Read moreMolecular Diagnostics
view channel
Whole Genome Sequencing in Routine Care Expands Rare Disease Detection
Rare diseases often involve prolonged diagnostic journeys that delay clinical decision-making and complicate family planning. As phenotypes become more heterogeneous, sequencing-based methods are increasingly... Read more
New AI Tool Improves Detection of Genetic Causes in Rare Disorders
Families affected by rare diseases often endure years of inconclusive testing and fragmented referrals before a definitive diagnosis. Despite broad access to genomic sequencing, many patients remain undiagnosed,... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel
New Bacterial Target Identified for Early Detection of Noma
Noma is a rapidly progressing orofacial infection that begins as gingivitis and can destroy oral and facial tissues, primarily affecting young children living in extreme poverty. Without treatment, it... Read more
Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
Streptococcus dysgalactiae subspecies equisimilis (SDSE) infections are increasing worldwide and include variants that may lead to severe disease. Researchers now report that whole-genome sequencing of... Read morePathology
view channel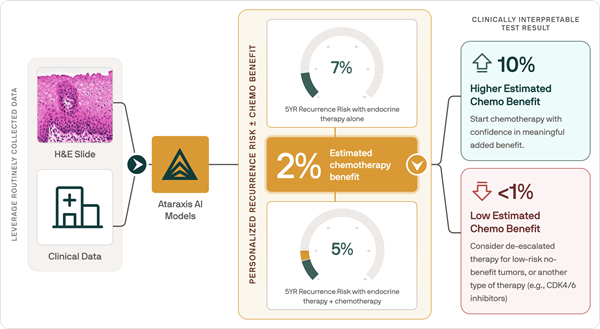
AI Tool Predicts Patient-Specific Chemotherapy Benefit in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer is typically guided by recurrence risk and population-level averages rather than patient-specific benefit. However, existing clinicopathologic... Read more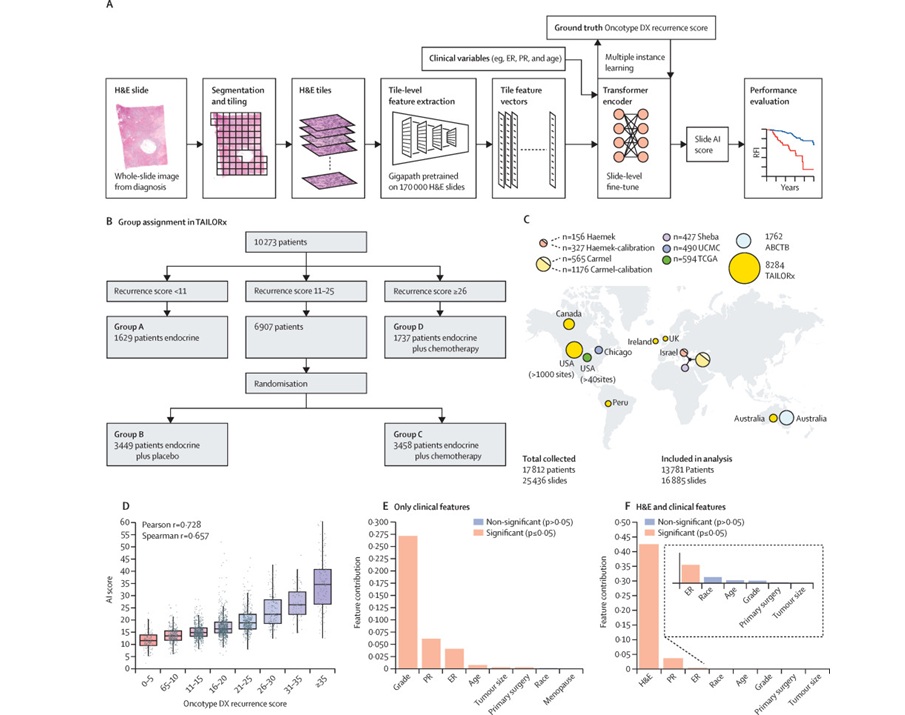
AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer remains a difficult decision because only a subset benefits and many undergo toxicity without gain. Genomic assays can help but are costly,... Read moreTechnology
view channel
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channel
Takara Bio USA and Hamilton Partner Partner to Automate NGS Library Preparation
Takara Bio USA, Inc. (San Jose, CA, USA), a wholly owned subsidiary of Takara Bio Inc., and Hamilton Company (Reno, NV, USA) announced a development and co-marketing agreement to deliver integrated, automated... Read more