Fecal Calprotectin Predicts Therapy Outcome in Ulcerative Colitis Patients
|
By LabMedica International staff writers Posted on 24 Aug 2021 |
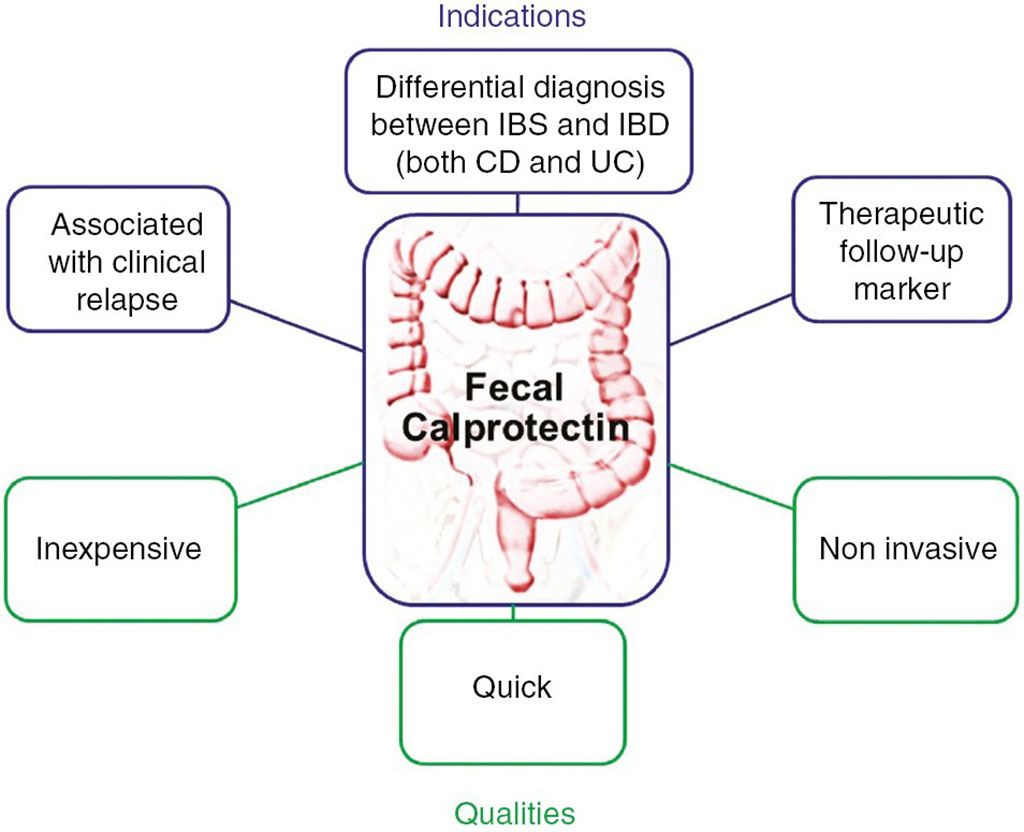
Image: Schematic representation of calprotectin indications in clinical practices and main advantages (Photo courtesy of Hôpital Beaujon)
Ulcerative colitis (UC) is a chronic intestinal disorder of unknown etiology and characterized by a relapsing and remitting course. The diagnosis and assessment of the disease activity has been based on clinical symptoms, laboratory measurements, findings of endoscopy and pathological examinations.
The development of a non-invasive and simple biomarker for evaluating the disease activity is considered necessary for the clinical management of UC. Calprotectin is a complex of mammalian proteins found in the cytosol of human neutrophils, monocytes and macrophages. Granulocyte and monocyte adsorptive apheresis (GMA) is widely used as a remission induction therapy for active UC patients.
A large team of Gastroenterologists at the (Asahikawa Medical University, Asahikawa, Japan) and their colleagues conducted a multicenter prospective observation study of patients who received 10 sessions of GMA, twice a week, for five consecutive weeks. A total 36 patients with active UC were enrolled in the study. Fecal calprotectin was measured at entry, one week, two weeks, at the end of GMA and on the day of endoscopy within 24 weeks after GMA.
The patients’ stool samples were homogenized by mixing with a predefined extraction buffer volume. After centrifugation, the supernatants were subjected to a fluorescence enzyme immunoassay using EliA Calprotectin 2 (Thermo Fisher Scientific, Tokyo, Japan). Laboratory values, including the white blood cell (WBC) count and C- reactive protein (CRP) level were also measured at the same time points as FC measurement: at entry, one and two weeks and at the end of GMA and on the day when endoscopy was performed within 24 weeks after GMA. The team analyzed the relationships between the clinical outcome (clinical remission [CR] and endoscopic remission [ER]) and the change in FC concentration.
The investigators reported that the overall CR and ER rates were 50.0% and 19.2%, respectively. After GMA, the median FC concentration in patients with ER was significantly lower than that in patients without ER (469 mg/kg versus 3,107 mg/kg). When the cut-off value of FC concentration was set at 1,150 mg/kg for assessing ER after GMA, the sensitivity and specificity were 0.8 and 0.81, respectively. The FC concentration had significantly decreased by one week. An ROC analysis demonstrated that the reduction rate of FC (ΔFC) at one week was the most accurate predictor of CR at the end of GMA (AUC = 0.852). When the cut-off value of ΔFC was set at ≤ 40% at one week for predicting CR at the end of GMA, the sensitivity and specificity were 76.9% and 84.6%, respectively.
The authors concluded that they had evaluated the utility of FC as a biomarker for assessing ER after GMA and predicting CR in the early phase during GMA in patients with active UC. The findings will benefit patients with active UC by allowing them to avoid unnecessary invasive procedures and will help establish new strategies for GMA. The study was published on August 6, 2021 in the journal BMC Gastroenterology.
Related Links:
Asahikawa Medical University
Thermo Fisher Scientific
The development of a non-invasive and simple biomarker for evaluating the disease activity is considered necessary for the clinical management of UC. Calprotectin is a complex of mammalian proteins found in the cytosol of human neutrophils, monocytes and macrophages. Granulocyte and monocyte adsorptive apheresis (GMA) is widely used as a remission induction therapy for active UC patients.
A large team of Gastroenterologists at the (Asahikawa Medical University, Asahikawa, Japan) and their colleagues conducted a multicenter prospective observation study of patients who received 10 sessions of GMA, twice a week, for five consecutive weeks. A total 36 patients with active UC were enrolled in the study. Fecal calprotectin was measured at entry, one week, two weeks, at the end of GMA and on the day of endoscopy within 24 weeks after GMA.
The patients’ stool samples were homogenized by mixing with a predefined extraction buffer volume. After centrifugation, the supernatants were subjected to a fluorescence enzyme immunoassay using EliA Calprotectin 2 (Thermo Fisher Scientific, Tokyo, Japan). Laboratory values, including the white blood cell (WBC) count and C- reactive protein (CRP) level were also measured at the same time points as FC measurement: at entry, one and two weeks and at the end of GMA and on the day when endoscopy was performed within 24 weeks after GMA. The team analyzed the relationships between the clinical outcome (clinical remission [CR] and endoscopic remission [ER]) and the change in FC concentration.
The investigators reported that the overall CR and ER rates were 50.0% and 19.2%, respectively. After GMA, the median FC concentration in patients with ER was significantly lower than that in patients without ER (469 mg/kg versus 3,107 mg/kg). When the cut-off value of FC concentration was set at 1,150 mg/kg for assessing ER after GMA, the sensitivity and specificity were 0.8 and 0.81, respectively. The FC concentration had significantly decreased by one week. An ROC analysis demonstrated that the reduction rate of FC (ΔFC) at one week was the most accurate predictor of CR at the end of GMA (AUC = 0.852). When the cut-off value of ΔFC was set at ≤ 40% at one week for predicting CR at the end of GMA, the sensitivity and specificity were 76.9% and 84.6%, respectively.
The authors concluded that they had evaluated the utility of FC as a biomarker for assessing ER after GMA and predicting CR in the early phase during GMA in patients with active UC. The findings will benefit patients with active UC by allowing them to avoid unnecessary invasive procedures and will help establish new strategies for GMA. The study was published on August 6, 2021 in the journal BMC Gastroenterology.
Related Links:
Asahikawa Medical University
Thermo Fisher Scientific
Latest Clinical Chem. News
- Urine-Based Nanosensor Tracks Lung Cancer and Fibrosis Noninvasively
- FDA-Cleared Assay Enables Comprehensive Automated Testosterone Testing
- CE-Marked Blood Biomarker Test Advances Automated Alzheimer’s Diagnostics
- Blood-Based Alzheimer’s Test Gains CE Mark for Amyloid Pathology Detection
- Noninvasive Urine Test May Support Earlier Diagnosis of Psychiatric Disorders
- At-Home Blood and Cognitive Tests Support Dementia Risk Stratification
- Ultrasensitive Test Detects Key Biomarker of Frontotemporal Dementia Subtype
- Routine Blood Tests Years Before Pregnancy Could Identify Preeclampsia Risk
- Blood Test Detects Testicular Cancer Missed by Standard Markers
- Routine Blood Tests Identify Biomarkers Linked to PTSD
- Proteomic Data Underscore Need for Age-Specific Pediatric Reference Ranges
- Routine Blood Count Ratio Linked to Future Alzheimer’s and Dementia Risk
- Label-Free Microfluidic Device Enriches Tumor Cells and Clusters from Pleural Effusions
- Rapid Biosensor Detects Pancreatic Cancer Biomarker for Early Detection
- Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
- Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Channels
Molecular Diagnostics
view channel
Sensitive Protein Marker Aids Diagnosis of Small Cell Prostate Cancer
Accurate identification of aggressive prostate cancer subtypes can be difficult when tumors lose expression of lineage markers used in routine pathology. Small cell carcinoma of the prostate, in particular,... Read more
Rapid Multiplex PCR Test Detects 11 Gastrointestinal Pathogens from Single Sample
Cepheid’s Xpert GI Panel has received CE marking under the In Vitro Diagnostic Medical Devices Regulation (IVDR) and is expected to begin shipping to countries that accept the CE mark in the coming weeks.... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
Immune aging is associated with weaker responses to vaccination, greater risks of infection, and higher levels of inflammation. Leveraging routinely ordered laboratory tests to quantify that responsiveness... Read more
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read moreMicrobiology
view channel
Study Finds Hidden Mpox Infections May Drive Ongoing Spread
Mpox continues to circulate despite vaccination, and many cases show no known link to a symptomatic partner. The role of people without symptoms has remained uncertain, limiting clarity on how transmission persists.... Read more
Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
Antimicrobial resistance (AMR) poses a growing threat to patient safety, with carbapenem-resistant Enterobacterales causing difficult-to-treat infections and leaving clinicians with limited therapeutic options.... Read more
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Point-of-Care Testing Enhances Health Literacy and Self-Management in Chronic Disease
Limited access to general practitioners and pathology services can delay diagnosis and monitoring for people in regional and remote communities. Rapid, on-the-spot testing can shorten turnaround times... Read more
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read moreIndustry
view channel
AI-Powered Multi-Functional Analyzer Wins German Innovation Award
Hematology services are increasingly delivered across distributed care settings, where limited staffing and complex workflows can extend turnaround times. Advanced morphology review still often depends... Read more