Serum Based Antigen Test Detects Childhood Tuberculosis
|
By LabMedica International staff writers Posted on 01 Jun 2021 |
Image: Computer-generated Mycobacterium tuberculosis bacteria, Ziehl-Neelsen stain. The reagents used are carbol fuchsin, acid alcohol, and methylene blue counterstain Acid-fast bacilli stain red and the background is blue (Photo courtesy of microbiologyinpictures)
Approximately one million children develop tuberculosis (TB) and 205,000 die of TB-related causes each year. Eighty percent of these deaths occur in children < 5 years old, with the majority (96%) of deaths occurring among children who did not receive treatment, where missed diagnoses are likely responsible for undertreatment.
Children with TB, particularly infants, frequently have paucibacillary TB, exhibit non-specific symptoms, and are likely to rapidly progress to disseminated or extrapulmonary TB in the absence of appropriate treatment. This clinical presentation, combined with difficulty obtaining respiratory samples, makes it challenging to diagnose pediatric TB and monitor treatment responses using standard sputum-based methods.
A multidisciplinary team of medical scientists led by Tulane University School of Medicine (New Orleans, LA, USA) used a small blood sample that can be easily obtained from children of any age to detect a specific protein (CFP-10) that the bacteria secrete to maintain the infection that develops into TB. Since this protein is present at very low levels in the blood, the assay uses an antibody specific for this protein to enrich it from other proteins in blood and a mass spectrometer to detect it with high sensitivity and accuracy.
The team used this test to screen stored blood samples collected from 284 HIV-infected and 235 children without the virus who participated in a large clinical trial conducted from 2004 to 2008. Serum CFP-10pep was analyzed using a nanoparticle-based immunoenrichment assay read by matrix assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF-MS) (Bruker Microflex LRF, Bremen, Germany) that detects Mtb-specific CFP-10pep from trypsin-digested serum or EDTA plasma samples.
The group found their test identified children diagnosed with TB by the current gold-standard TB tests with 100% accuracy. The assay also detected 83.7% of TB cases that were missed by these tests, but that were later diagnosed by a standard checklist employing an array of other information collected by each child's physician (unconfirmed TB cases). The test also detected CFP-10 in 77% of the blood samples that were collected 24 weeks before children were diagnosed with TB by other methods, indicating its strong potential for early TB diagnosis. The biomarker from some positive cases can be detected as early as 60 weeks before their TB diseases were confirmed.
Tony Y. Hu, PhD, a Professor of Biochemistry and Molecular Biology and senior author of the study, said, “This is a breakthrough for infants with tuberculosis because we don't have this kind of screening technology to catch early infections among those youngest groups who are most likely to be undiagnosed. I hope this method can be pushed forward quickly to reach these children as early as possible.”
The authors concluded that their results suggest that serum CFP-10pep signal could improve TB diagnosis in children, as it exceeds the WHO-specified sensitivity requirements for new non-sputum diagnostics, and exhibits similar performance for all TB manifestations, including culture-negative TB, HIV/TB co-infection, and extrapulmonary TB, which are normally challenging to diagnose. The study was published on May 18, 2021 in the journal BMC Medicine.
Related Links:
Tulane University School of Medicine
Bruker
Children with TB, particularly infants, frequently have paucibacillary TB, exhibit non-specific symptoms, and are likely to rapidly progress to disseminated or extrapulmonary TB in the absence of appropriate treatment. This clinical presentation, combined with difficulty obtaining respiratory samples, makes it challenging to diagnose pediatric TB and monitor treatment responses using standard sputum-based methods.
A multidisciplinary team of medical scientists led by Tulane University School of Medicine (New Orleans, LA, USA) used a small blood sample that can be easily obtained from children of any age to detect a specific protein (CFP-10) that the bacteria secrete to maintain the infection that develops into TB. Since this protein is present at very low levels in the blood, the assay uses an antibody specific for this protein to enrich it from other proteins in blood and a mass spectrometer to detect it with high sensitivity and accuracy.
The team used this test to screen stored blood samples collected from 284 HIV-infected and 235 children without the virus who participated in a large clinical trial conducted from 2004 to 2008. Serum CFP-10pep was analyzed using a nanoparticle-based immunoenrichment assay read by matrix assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF-MS) (Bruker Microflex LRF, Bremen, Germany) that detects Mtb-specific CFP-10pep from trypsin-digested serum or EDTA plasma samples.
The group found their test identified children diagnosed with TB by the current gold-standard TB tests with 100% accuracy. The assay also detected 83.7% of TB cases that were missed by these tests, but that were later diagnosed by a standard checklist employing an array of other information collected by each child's physician (unconfirmed TB cases). The test also detected CFP-10 in 77% of the blood samples that were collected 24 weeks before children were diagnosed with TB by other methods, indicating its strong potential for early TB diagnosis. The biomarker from some positive cases can be detected as early as 60 weeks before their TB diseases were confirmed.
Tony Y. Hu, PhD, a Professor of Biochemistry and Molecular Biology and senior author of the study, said, “This is a breakthrough for infants with tuberculosis because we don't have this kind of screening technology to catch early infections among those youngest groups who are most likely to be undiagnosed. I hope this method can be pushed forward quickly to reach these children as early as possible.”
The authors concluded that their results suggest that serum CFP-10pep signal could improve TB diagnosis in children, as it exceeds the WHO-specified sensitivity requirements for new non-sputum diagnostics, and exhibits similar performance for all TB manifestations, including culture-negative TB, HIV/TB co-infection, and extrapulmonary TB, which are normally challenging to diagnose. The study was published on May 18, 2021 in the journal BMC Medicine.
Related Links:
Tulane University School of Medicine
Bruker
Latest Microbiology News
- New Bacterial Target Identified for Early Detection of Noma
- Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
- Rapid Urine Test Speeds Antibiotic Selection for UTIs
- WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
- Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
- WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
- New Imaging Approach Could Help Predict Dangerous Gut Infection
- Rapid Sequencing Could Transform Tuberculosis Care
- Blood-Based Viral Signature Identified in Crohn’s Disease
- Hidden Gut Viruses Linked to Colorectal Cancer Risk
- Three-Test Panel Launched for Detection of Liver Fluke Infections
- Rapid Test Promises Faster Answers for Drug-Resistant Infections
- CRISPR-Based Technology Neutralizes Antibiotic-Resistant Bacteria
- Comprehensive Review Identifies Gut Microbiome Signatures Associated With Alzheimer’s Disease
- AI-Powered Platform Enables Rapid Detection of Drug-Resistant C. Auris Pathogens
Channels
Clinical Chemistry
view channel
AI-Based Blood Test Diagnose Multiple Brain Disorders from Blood Sample
Diagnosing the cause of age-related cognitive symptoms remains challenging because clinical presentations of neurodegenerative diseases often overlap, and multiple pathologies can co-occur... Read more
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read moreMolecular Diagnostics
view channel
RNA Profiling Uncovers Therapeutic Targets in Solid Tumors
Many patients with advanced solid tumors exhaust broad DNA panel testing yet still lack biomarkers that match guideline-recommended therapies, limiting access to targeted options. Expanding molecular profiling... Read more
Whole Genome Sequencing in Routine Care Expands Rare Disease Detection
Rare diseases often involve prolonged diagnostic journeys that delay clinical decision-making and complicate family planning. As phenotypes become more heterogeneous, sequencing-based methods are increasingly... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
Antibody Blood Test Identifies Active TB and Distinguishes Latent Infection
Active tuberculosis (TB) remains a leading cause of death and illness worldwide, yet distinguishing contagious disease from latent infection continues to challenge clinicians. Standard screening tools... Read more
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read morePathology
view channel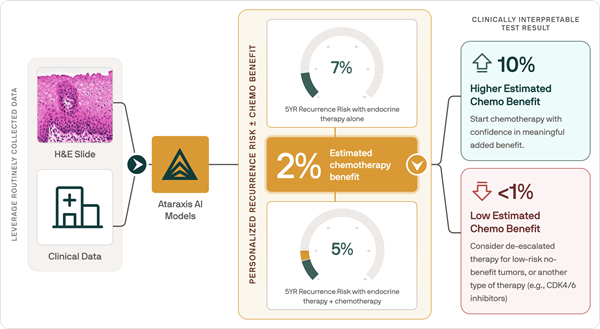
AI Tool Predicts Patient-Specific Chemotherapy Benefit in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer is typically guided by recurrence risk and population-level averages rather than patient-specific benefit. However, existing clinicopathologic... Read more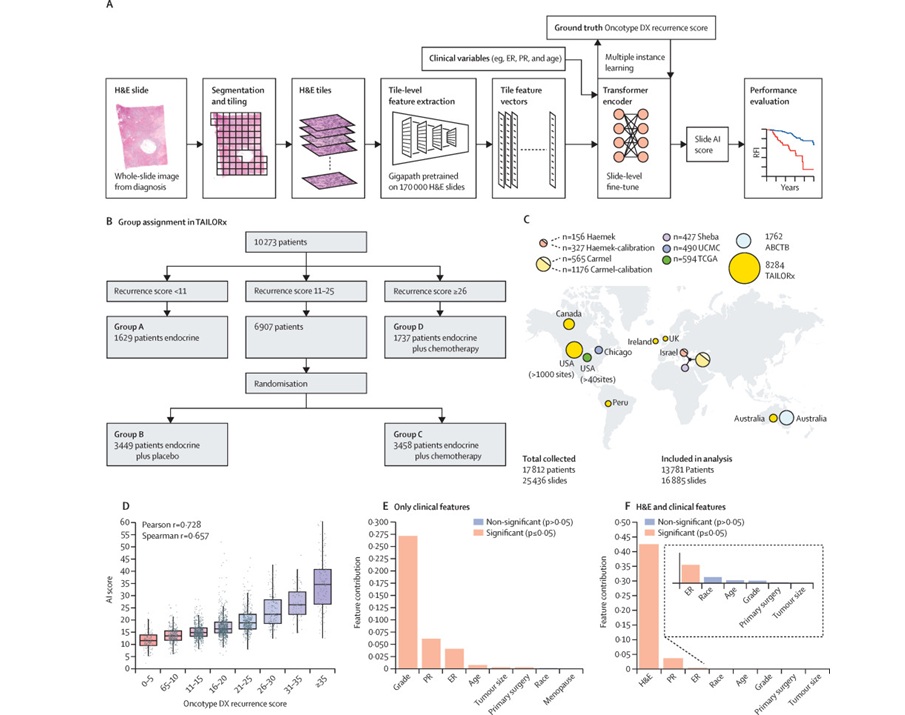
AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer remains a difficult decision because only a subset benefits and many undergo toxicity without gain. Genomic assays can help but are costly,... Read moreTechnology
view channel
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channel
Takara Bio USA and Hamilton Partner Partner to Automate NGS Library Preparation
Takara Bio USA, Inc. (San Jose, CA, USA), a wholly owned subsidiary of Takara Bio Inc., and Hamilton Company (Reno, NV, USA) announced a development and co-marketing agreement to deliver integrated, automated... Read more




.jpg)