Plasma-Based Algorithm Accurately Predicts Likelihood of Developing Alzheimer’s Disease
|
By LabMedica International staff writers Posted on 31 May 2021 |
Image: Neurons were grown in tissue culture and stained with antibody to microtubule associated protein 2 (MAP2) protein in green and MAP tau in red. MAP2 is found only in dendrites and perikarya, while tau is found in these sites and in axons as well. DNA is shown in blue (Photo courtesy of EnCor Biotechnology Inc. via Wikimedia Commons)
A team of Swedish researchers has created an algorithm for use by physicians lacking access to advanced diagnostic instruments, which accurately predicts future risk of developing Alzheimer’s disease (AD).
Early and accurate diagnosis of AD is necessary to take advantage of a new generation of drugs designed to slow down the progression of the disease.
In this regard, investigators at Lund University (Sweden) hypothesized that the combination of plasma phosphorylated tau protein (P-tau) and other accessible biomarkers might provide accurate prediction about the risk of developing AD. They tested this theory by analyzing blood samples from 340 participants with subjective cognitive decline and mild cognitive impairment from the Swedish BioFINDER study and 543 participants from the North American Alzheimer’s Disease Neuroimaging Initiative (ADNI). Plasma P-tau, plasma Abeta42/Abeta40, plasma neurofilament light, APOE genotype, brief cognitive tests, and an AD-specific magnetic resonance imaging measure were examined using progression to AD as outcome.
Results revealed that within four years of the analysis, plasma P-tau217 predicted AD accurately (area under the curve (AUC) = 0.83) in the BioFINDER group. Combining plasma P-tau217, memory, executive function and APOE produced higher accuracy (AUC = 0.91). In the ADNI group, this model had similar AUC (0.90) using plasma P-tau181 instead of P-tau217.
The diagnostic model was used to predict the probability of an individual progressing to AD. Within two and six years, similar models had AUCs of 0.90–0.91 in both cohorts. Significantly, measuring cerebrospinal fluid P-tau, Abeta42/Abeta40, and neurofilament light instead of plasma biomarkers did not significantly improve the accuracy. Furthermore, this simple prognostic algorithm was significantly more accurate than clinical predictions by dementia experts who examined the patients, but did not have access to data generated by the algorithm.
“A combination of a simple blood test (measuring a variant of the tau protein and a risk gene for Alzheimer's) and three brief cognitive tests that only take 10 minutes to complete, predicted with over 90% certainty which patients would develop Alzheimer's dementia within four years. This simple prognostic algorithm was significantly more accurate than the clinical predictions by the dementia experts who examined the patients, but did not have access to expensive spinal fluid testing or PET scans,” said senior author Dr. Oskar Hansson, professor of neurology at Lund University. “The algorithm will enable us to recruit people with Alzheimer's at an early stage, which is when new drugs have a better chance of slowing the course of the disease.”
The Alzheimer’s disease diagnostic algorithm was described in the May 24, 2021, online edition of the journal Nature Medicine.
Related Links:
Lund University
Early and accurate diagnosis of AD is necessary to take advantage of a new generation of drugs designed to slow down the progression of the disease.
In this regard, investigators at Lund University (Sweden) hypothesized that the combination of plasma phosphorylated tau protein (P-tau) and other accessible biomarkers might provide accurate prediction about the risk of developing AD. They tested this theory by analyzing blood samples from 340 participants with subjective cognitive decline and mild cognitive impairment from the Swedish BioFINDER study and 543 participants from the North American Alzheimer’s Disease Neuroimaging Initiative (ADNI). Plasma P-tau, plasma Abeta42/Abeta40, plasma neurofilament light, APOE genotype, brief cognitive tests, and an AD-specific magnetic resonance imaging measure were examined using progression to AD as outcome.
Results revealed that within four years of the analysis, plasma P-tau217 predicted AD accurately (area under the curve (AUC) = 0.83) in the BioFINDER group. Combining plasma P-tau217, memory, executive function and APOE produced higher accuracy (AUC = 0.91). In the ADNI group, this model had similar AUC (0.90) using plasma P-tau181 instead of P-tau217.
The diagnostic model was used to predict the probability of an individual progressing to AD. Within two and six years, similar models had AUCs of 0.90–0.91 in both cohorts. Significantly, measuring cerebrospinal fluid P-tau, Abeta42/Abeta40, and neurofilament light instead of plasma biomarkers did not significantly improve the accuracy. Furthermore, this simple prognostic algorithm was significantly more accurate than clinical predictions by dementia experts who examined the patients, but did not have access to data generated by the algorithm.
“A combination of a simple blood test (measuring a variant of the tau protein and a risk gene for Alzheimer's) and three brief cognitive tests that only take 10 minutes to complete, predicted with over 90% certainty which patients would develop Alzheimer's dementia within four years. This simple prognostic algorithm was significantly more accurate than the clinical predictions by the dementia experts who examined the patients, but did not have access to expensive spinal fluid testing or PET scans,” said senior author Dr. Oskar Hansson, professor of neurology at Lund University. “The algorithm will enable us to recruit people with Alzheimer's at an early stage, which is when new drugs have a better chance of slowing the course of the disease.”
The Alzheimer’s disease diagnostic algorithm was described in the May 24, 2021, online edition of the journal Nature Medicine.
Related Links:
Lund University
Latest Molecular Diagnostics News
- Adaptive PCR Platform Improves Consistency in Small-Batch NGS Workflows
- Portable Test Uses CRISPR to Rapidly Identify STIs and Resistance Markers
- New Molecular Test Boosts Accuracy of Bile Duct Cancer Diagnosis
- First IVDR‑Certified IGH Clonality Assay Supports Diagnosis of B-Cell Malignancies
- Plasma ctDNA Testing Predicts Breast Cancer Recurrence After Neoadjuvant Therapy
- New Respiratory Panel Expands Pathogen Detection to 25 Targets
- Nasal Swab May Reveal Early Signs of Alzheimer’s Disease
- Blood Biomarker Predicts Cognitive Outcomes After Cardiac Arrest
- Liquid Biopsy Enables Faster Diagnosis of Childhood Cancer in Africa
- Blood Test Helps Guide Treatment in Older Women with Breast Cancer
- Rapid Host-Response Test Distinguishes Bacterial and Viral Infections in Minutes
- Liquid Biopsy Method Pinpoints Disease Source From a Single Drop of Blood
- Study Reveals Widespread Errors in Gene Variant Naming
- New Blood Test Aims to Transform Liver Cancer Surveillance
- New Biomarkers Indicate Higher Liver Cancer Risk in Chronic Hepatitis B Patients
- Precision Analyzer Reveals ‘Chameleon Proteins’ Causing Intractable Diseases
Channels
Clinical Chemistry
view channel
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read more
Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
Alzheimer’s disease develops slowly, and biological changes can appear in blood many years before symptoms. While plasma assays for phosphorylated tau offer earlier detection, discerning whether these... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel
Rapid Urine Test Speeds Antibiotic Selection for UTIs
Urinary tract infections are a common reason for antibiotic prescribing and have led to more than 800,000 hospital admissions across England in the past five years, according to National Health Service data.... Read more
WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
Tuberculosis (TB) remains a leading infectious killer, with more than 3,300 deaths and 29,000 new illnesses every day. Diagnostic delays and dependence on centralized laboratory networks continue to impede... Read morePathology
view channel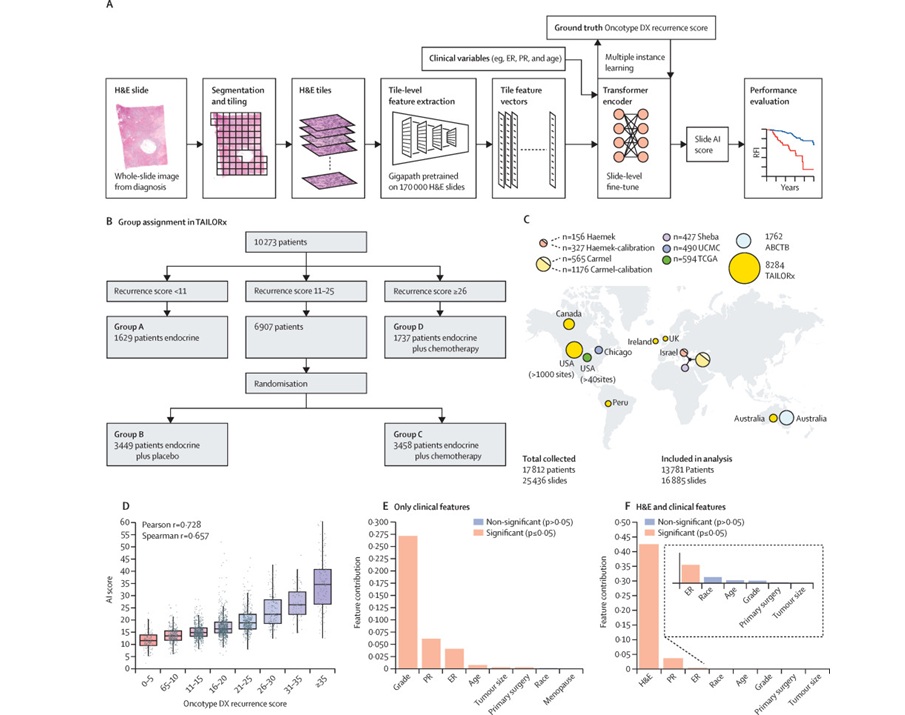
AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer remains a difficult decision because only a subset benefits and many undergo toxicity without gain. Genomic assays can help but are costly,... Read more
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read moreTechnology
view channel
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channel
Lunit and CellCarta Collaborate to Expand AI Pathology in CDx Development
Lunit (Seoul, South Korea), a leading provider of AI for cancer diagnostics and precision oncology, and CellCarta (Montreal, QC, Canada), a global contract research organization (CRO) laboratory serving... Read more