Genetic Alterations Associated with Sporadic Congenital Hydrocephalus
|
By LabMedica International staff writers Posted on 02 Nov 2020 |
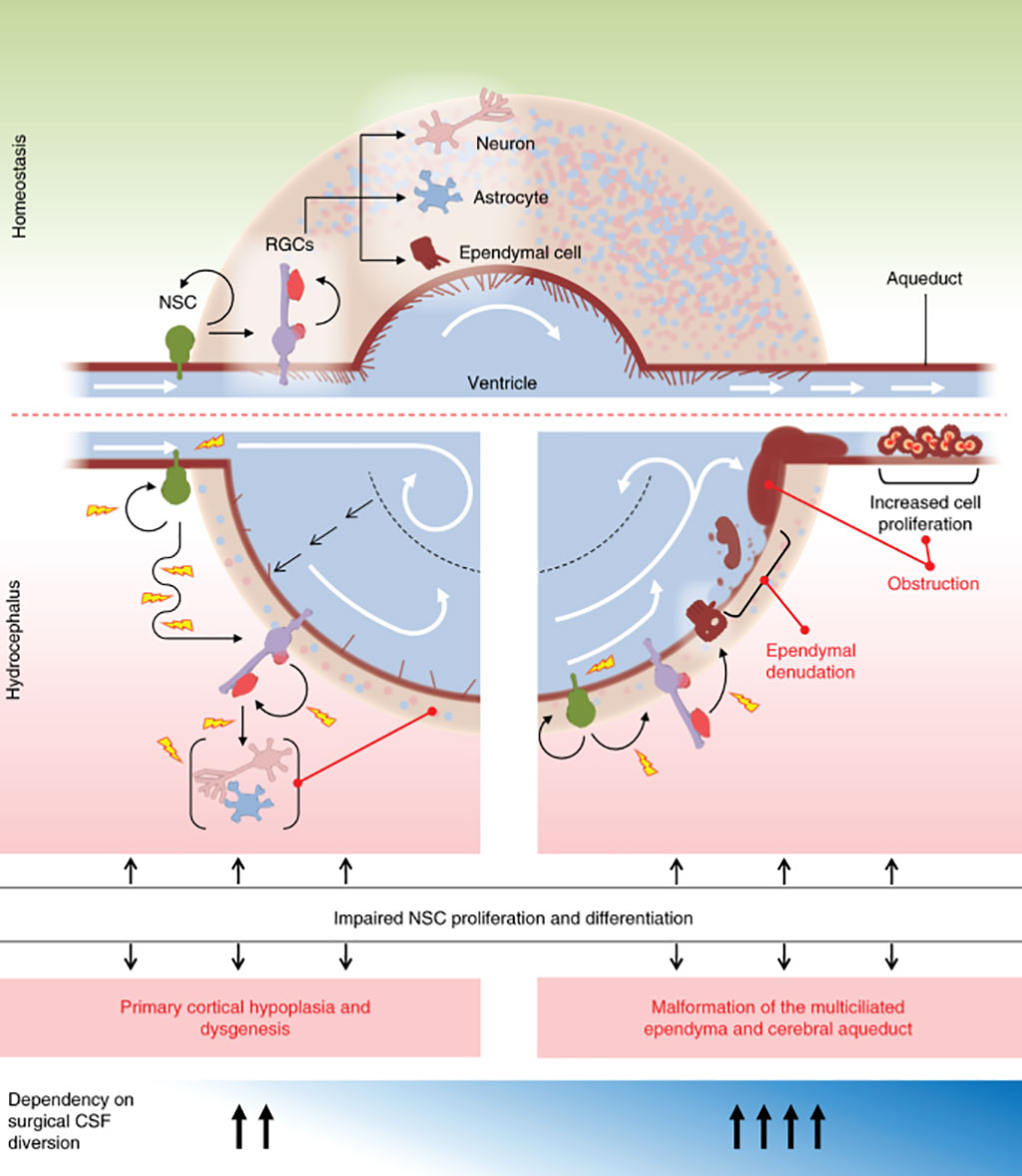
Image: A neural stem cell model of sporadic congenital hydrocephalus (Photo courtesy of Yale University School of Medicine).
Congenital hydrocephalus (CH), characterized by enlarged brain ventricles, is considered a disease of excessive cerebrospinal fluid (CSF) accumulation and thereby treated with neurosurgical CSF diversion with high morbidity and failure rates.
Congenital hydrocephalus is present in the infant prior to birth, meaning the fetus developed hydrocephalus in utero during fetal development. The most common cause of congenital hydrocephalus is aqueductal stenosis, which occurs when the narrow passage between the third and fourth ventricles in the brain is blocked or too narrow to allow sufficient CSF to drain.
A multidisciplinary team at Yale University School of Medicine (New Haven, CT, USA) performed whole-exome sequencing of 381 individuals with sporadic congenital hydrocephalus who were treated with neurosurgery, a cohort that included 232 parent-offspring trios. Another 1,798 trios of unaffected siblings and parents of individuals with autism spectrum disorder were analyzed in parallel as a control group.
The team uncovered 12 genes with two or more de novo mutations each that were predicted to be protein damaging. Five of these genes: TRIM71, SMARCC1, PTEN, PIK3CA, and FOXJ1, had significantly more mutations than expected. The investigators further estimated that about 22% of sporadic congenital hydrocephalus are due to rare, damaging mutations. The teams’ analysis additionally implicated a number of genes in the PI3K signaling pathway in congenital hydrocephalus. These genes regulate cell growth, proliferation, and differentiation in numerous tissues, including developing neural stem cells. In particular, they uncovered three de novo mutations in PI3KCA, three in PTEN, and two in MTOR.
Individuals with mutations in TRIM71, which maintains stem cell pluripotency, are more likely to have cranial nerve defect, non-obstructive inter-hemispheric cysts, and hearing loss. Meanwhile, individuals with mutations in SMARCC1, which regulates gene expression needed for neural stem cell proliferation, differentiation, and survival during telencephalon development, are more likely to have aqueductal stenosis and cardiac and skeletal abnormalities.
All together, the known, high-confidence, and probable risk genes for congenital hydrocephalus converge in gene co-expression networks of the mid-gestational human cortex, In particular, the congenital hydrocephalus risk genes converge on a network previously linked to autism spectrum disorder that is enriched for neuronal differentiation and RNA processing gene ontology terms.
Kristopher T. Kahle, MD, PhD, an assistant professor of neurosurgery and a senior author of the study, said, “It is possible that some of the neurological problems that many of these congenital hydrocephalus patients have may not be due to inadequately working shunts, but are in fact neurodevelopmental sequelae of a genetic condition that alters neuronal function.”
The authors concluded that that genetic disruptions affecting early brain development may drive sporadic congenital hydrocephalus. Their data implicate genetically encoded neural stem cell dysregulation and an associated impairment of fetal neurogliogenesis as primary pathophysiological events in a significant number of congenital hydrocephalus cases. The study was published on October 19, 2020 in the journal Nature Medicine.
Related Links:
Yale University School of Medicine
Congenital hydrocephalus is present in the infant prior to birth, meaning the fetus developed hydrocephalus in utero during fetal development. The most common cause of congenital hydrocephalus is aqueductal stenosis, which occurs when the narrow passage between the third and fourth ventricles in the brain is blocked or too narrow to allow sufficient CSF to drain.
A multidisciplinary team at Yale University School of Medicine (New Haven, CT, USA) performed whole-exome sequencing of 381 individuals with sporadic congenital hydrocephalus who were treated with neurosurgery, a cohort that included 232 parent-offspring trios. Another 1,798 trios of unaffected siblings and parents of individuals with autism spectrum disorder were analyzed in parallel as a control group.
The team uncovered 12 genes with two or more de novo mutations each that were predicted to be protein damaging. Five of these genes: TRIM71, SMARCC1, PTEN, PIK3CA, and FOXJ1, had significantly more mutations than expected. The investigators further estimated that about 22% of sporadic congenital hydrocephalus are due to rare, damaging mutations. The teams’ analysis additionally implicated a number of genes in the PI3K signaling pathway in congenital hydrocephalus. These genes regulate cell growth, proliferation, and differentiation in numerous tissues, including developing neural stem cells. In particular, they uncovered three de novo mutations in PI3KCA, three in PTEN, and two in MTOR.
Individuals with mutations in TRIM71, which maintains stem cell pluripotency, are more likely to have cranial nerve defect, non-obstructive inter-hemispheric cysts, and hearing loss. Meanwhile, individuals with mutations in SMARCC1, which regulates gene expression needed for neural stem cell proliferation, differentiation, and survival during telencephalon development, are more likely to have aqueductal stenosis and cardiac and skeletal abnormalities.
All together, the known, high-confidence, and probable risk genes for congenital hydrocephalus converge in gene co-expression networks of the mid-gestational human cortex, In particular, the congenital hydrocephalus risk genes converge on a network previously linked to autism spectrum disorder that is enriched for neuronal differentiation and RNA processing gene ontology terms.
Kristopher T. Kahle, MD, PhD, an assistant professor of neurosurgery and a senior author of the study, said, “It is possible that some of the neurological problems that many of these congenital hydrocephalus patients have may not be due to inadequately working shunts, but are in fact neurodevelopmental sequelae of a genetic condition that alters neuronal function.”
The authors concluded that that genetic disruptions affecting early brain development may drive sporadic congenital hydrocephalus. Their data implicate genetically encoded neural stem cell dysregulation and an associated impairment of fetal neurogliogenesis as primary pathophysiological events in a significant number of congenital hydrocephalus cases. The study was published on October 19, 2020 in the journal Nature Medicine.
Related Links:
Yale University School of Medicine
Latest Pathology News
- AI-Powered Atlas Maps Immune Structures Linked to Cancer Outcomes
- AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
- Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
- AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
- FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
- New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
- AI System Analyzes Routine Pathology Slides to Predict Cancer Outcomes
- New Tissue Mapping Approach Identifies High-Risk Form of Diabetic Kidney Disease
- Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
- Interpretable AI Reveals Hidden Cellular Features from Microscopy Images
- Tumor Immune Structure Predicts Response to Immunotherapy in Melanoma
- Plug-and-Play AI Pathology System Classifies Multiple Cancers from Few Slides
- AI-Based Assays Support Risk Stratification in Prostate and Breast Cancer
- AI Pathology Model Predicts Immunotherapy Response in Lung Cancer
- Study Reveals Moleclar Mechanism Driving Aggressive Skin Cancer
- AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
Channels
Clinical Chemistry
view channel
Urine-Based Test Shows Promise for Autism Screening in Children
Autism spectrum disorder (ASD) is commonly diagnosed through behavioral assessments, which can involve long waits that delay intervention. Earlier identification is linked to better developmental outcomes,... Read more
Liquid Biopsy Biomarkers May Improve Childhood Epilepsy Diagnosis
Childhood epilepsy remains a major neurological disorder with unmet needs for accurate, non-invasive biomarkers, as conventional tests such as electroencephalography and neuroimaging can have limited sensitivity... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channel
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read more
Immune Enzyme Linked to Treatment-Resistant Inflammatory Bowel Disease
Inflammatory bowel disease (IBD) affects nearly 3 million people in the United States and its prevalence continues to rise. Medications that target tumor necrosis factor (TNF)-alpha are widely used, but... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read morePathology
view channel
AI-Powered Atlas Maps Immune Structures Linked to Cancer Outcomes
Tertiary lymphoid structures are emerging as important indicators of antitumor immunity, but their heterogeneity and spatial context within tumors remain difficult to capture through routine diagnostics.... Read more
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read moreTechnology
view channel
Mailed Screening Kits Help Reduce Colorectal Cancer Screening Gaps
Colorectal cancer screening is a longstanding preventive priority, yet participation and follow-up remain uneven across patient groups. Safety‑net primary care settings often face barriers that limit screening... Read more
Algorithm Panel Aids Liver Fibrosis Assessment and Liver Cancer Surveillance
Chronic liver disease is common and often progresses silently, increasing the risk of cirrhosis and hepatocellular carcinoma when not detected early. With an estimated 1.5 billion people affected worldwide... Read moreIndustry
view channelWerfen and Oxford Nanopore Collaborate on Transplant Assay Development
Werfen (Barcelona, Spain), a global specialized diagnostics company, has announced a strategic collaboration with Oxford Nanopore Technologies (Oxford, UK), which develops nanopore-based sequencing technology,... Read more

























