Diagnostic Methods Compared for Fecal Helminth Eggs
|
By LabMedica International staff writers Posted on 24 Jun 2018 |
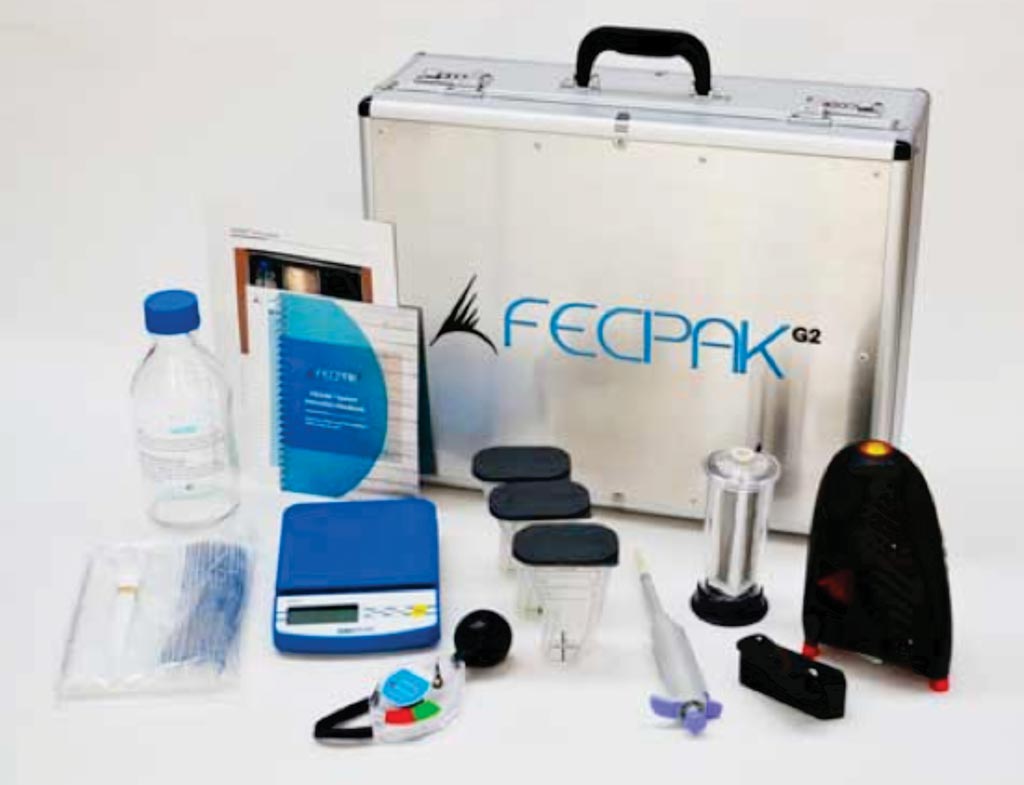
Image: The FECPAKG2 is a complete remote-location parasite assessment tool (Photo courtesy of Techion Group).
For estimating prevalence of soil-transmitted helminthiasis, assessing infection intensities, evaluating drug efficacy and monitoring drug resistance, accurate diagnostic methods are essential. The currently recommended Kato-Katz method has already been in use for decades.
A comparison has been made between the Kato-Katz method and a novel method, which is an online, remote location, parasite diagnostic system previously used in veterinary medicine. The new method is based on the flotation-dilution principle and its novelty is the accumulation of parasite eggs into one viewing area within a fluid meniscus.
An international team of scientists working with the Swiss Tropical and Public Health Institute (Basel, Switzerland) collected two stool samples from adolescent participants (age 15–18 years) at baseline and 14 to 21 days after treatment in the framework of a randomized clinical trial on Pemba Island, Tanzania. Stool samples were analyzed with different diagnostic efforts: i) one or ii) two Kato-Katz thick smears from the first sample, iii) two Kato-Katz thick smears from two samples and iv) FECPAKG2 from the first sample. For FECPAKG2 an image of the fecal sample was then captured, and stored offline on a computer and uploaded onto a cloud once connected to the Internet. Subsequently, the image can be analyzed at any time by specialists around the world.
The team reported that complete data for all diagnostic efforts were available from 615 participants at baseline and 231 hookworm-positive participants at follow-up. At baseline FECPAKG2 revealed a sensitivity of 75.6% (72.0–77.7) for detecting Ascaris lumbricoides, 71.5% (67.4–95.3) for hookworm and 65.8% (64.9–66.2) for Trichuris trichiura, which was significantly lower than any of the Kato-Katz methods and highly dependent on infection intensity. Despite that the egg counts based on FECPAKG2 were relatively lower compared to Kato-Katz by a ratio of 0.38 (0.32–0.43) for A. lumbricoides, 0.36 (0.33–0.40) for hookworm and 0.08 (0.07–0.09) for T. trichiura, the egg reduction rates (ERR) were correctly estimated with FECPAKG2.
The authors concluded that the sensitivity to identify any soil-transmitted helminthes (STH) infection was considerably lower for FECPAKG2 compared to Kato-Katz. Following rigorous development, FECPAKG2 might be an interesting tool with unique features for epidemiological and clinical studies. The study was published on June 4, 2018 in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Swiss Tropical and Public Health Institute
A comparison has been made between the Kato-Katz method and a novel method, which is an online, remote location, parasite diagnostic system previously used in veterinary medicine. The new method is based on the flotation-dilution principle and its novelty is the accumulation of parasite eggs into one viewing area within a fluid meniscus.
An international team of scientists working with the Swiss Tropical and Public Health Institute (Basel, Switzerland) collected two stool samples from adolescent participants (age 15–18 years) at baseline and 14 to 21 days after treatment in the framework of a randomized clinical trial on Pemba Island, Tanzania. Stool samples were analyzed with different diagnostic efforts: i) one or ii) two Kato-Katz thick smears from the first sample, iii) two Kato-Katz thick smears from two samples and iv) FECPAKG2 from the first sample. For FECPAKG2 an image of the fecal sample was then captured, and stored offline on a computer and uploaded onto a cloud once connected to the Internet. Subsequently, the image can be analyzed at any time by specialists around the world.
The team reported that complete data for all diagnostic efforts were available from 615 participants at baseline and 231 hookworm-positive participants at follow-up. At baseline FECPAKG2 revealed a sensitivity of 75.6% (72.0–77.7) for detecting Ascaris lumbricoides, 71.5% (67.4–95.3) for hookworm and 65.8% (64.9–66.2) for Trichuris trichiura, which was significantly lower than any of the Kato-Katz methods and highly dependent on infection intensity. Despite that the egg counts based on FECPAKG2 were relatively lower compared to Kato-Katz by a ratio of 0.38 (0.32–0.43) for A. lumbricoides, 0.36 (0.33–0.40) for hookworm and 0.08 (0.07–0.09) for T. trichiura, the egg reduction rates (ERR) were correctly estimated with FECPAKG2.
The authors concluded that the sensitivity to identify any soil-transmitted helminthes (STH) infection was considerably lower for FECPAKG2 compared to Kato-Katz. Following rigorous development, FECPAKG2 might be an interesting tool with unique features for epidemiological and clinical studies. The study was published on June 4, 2018 in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Swiss Tropical and Public Health Institute
Latest Microbiology News
- Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
- Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
- Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
- Label-Free Microscopy Method Enables Faster, Quantitative Detection of Malaria
- Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
- RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
- Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
- New Bacterial Target Identified for Early Detection of Noma
- Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
- Rapid Urine Test Speeds Antibiotic Selection for UTIs
Channels
Clinical Chemistry
view channel
Blood-Based Alzheimer’s Test Gains CE Mark for Amyloid Pathology Detection
Alzheimer’s disease is the most common cause of dementia, yet confirmatory testing remains invasive and hard to access. Diagnosis currently takes an average of 3.5 years, and about 75% of people with dementia... Read more
FDA-Cleared Assay Enables Comprehensive Automated Testosterone Testing
Accurate evaluation of androgen status often requires concordant measurement of total testosterone, free testosterone, and sex hormone‑binding globulin. Reference methods such as equilibrium dialysis with... Read moreMolecular Diagnostics
view channel
Gene Panel Shows Promise for Predicting Chemotherapy Response in TNBC
Triple-negative breast cancer (TNBC) is an aggressive subtype commonly treated with chemotherapy, yet outcomes vary widely among patients. Understanding the tumor features that drive this variability remains... Read more
Realistic Mock Samples Aim to Speed Cervical Cancer Test Development
Cervical cancer remains highly preventable, yet screening access is limited in many low- and middle-income settings. Gold-standard tests for high-risk human papillomavirus (HPV) detect viral DNA or messenger... Read more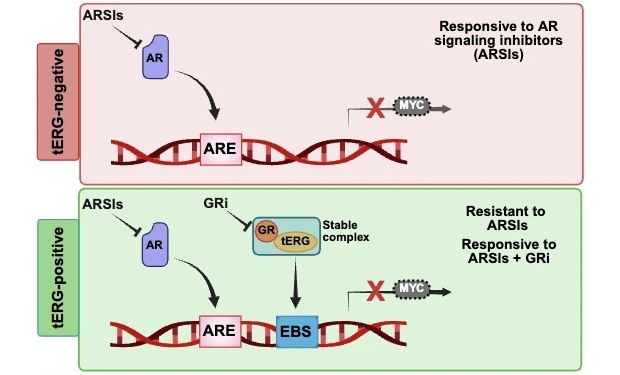
Molecular Marker Identifies Hormone Therapy Resistance Pathway in Prostate Cancer
Most prostate cancers depend on androgen signaling, making hormone suppression or blockade a central treatment strategy. Although many patients respond initially, tumors often adapt and eventually progress,... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
Immune aging is associated with weaker responses to vaccination, greater risks of infection, and higher levels of inflammation. Leveraging routinely ordered laboratory tests to quantify that responsiveness... Read more
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Point-of-Care Testing Enhances Health Literacy and Self-Management in Chronic Disease
Limited access to general practitioners and pathology services can delay diagnosis and monitoring for people in regional and remote communities. Rapid, on-the-spot testing can shorten turnaround times... Read more
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more