Das SRL Exhibits New Lab Solutions at MEDLAB 2017
|
By LabMedica International staff writers Posted on 09 Feb 2017 |

Image: The AP22 IF BLOT ELITE system (Photo courtesy of Das SRL).
Das SRL, which designs, manufactures, and markets analyzers and automated equipment for application in immuno-enzimatic, immuno-fluorescence, and clinical chemistry, exhibited its AP22 IF BLOT ELITE system and NEO BIL Plus bilirubinometer at MEDLAB 2017.
Italy-based Das SRL offers systems for automated ELISA process on micro-plates and immunofluorescence on slides; readers and washers for laboratories using micro-titre plates or micro-strips; programmable semi-automated photometers for clinical chemistry analysis; and neonatal bilirubinometers for bilirubin monitoring using centrifuged whole blood samples.
The company’s AP22 IF BLOT ELITE is a fully automated system for ELISA and IFA and BLOT methods. The instrument is capable of running 2 ELISA micro-plates with 8 tests on line, 16 IFA slides or 24 BLOT strips. It can perform full automation for the Dot/Western Blot, from sample dilution and dispensing, reagent dispensing, incubation, and strip washing to dot imaging processing and assessment for each strip.
Also on display at the 4-day exhibition held from February 6-9, 2017, in Dubai, UAE was Das SRL’s NEO BIL Plus, an advanced version of traditional bilirubinometers, designed to analyze the 'total bilirubin' through a microcapillary tube. NEO BIL Plus features a special reading system, which can carry out a precise sample measurement of both bilirubin and interferentials at the same point of the capillary using a small quantity of serum and without any reagents.
Italy-based Das SRL offers systems for automated ELISA process on micro-plates and immunofluorescence on slides; readers and washers for laboratories using micro-titre plates or micro-strips; programmable semi-automated photometers for clinical chemistry analysis; and neonatal bilirubinometers for bilirubin monitoring using centrifuged whole blood samples.
The company’s AP22 IF BLOT ELITE is a fully automated system for ELISA and IFA and BLOT methods. The instrument is capable of running 2 ELISA micro-plates with 8 tests on line, 16 IFA slides or 24 BLOT strips. It can perform full automation for the Dot/Western Blot, from sample dilution and dispensing, reagent dispensing, incubation, and strip washing to dot imaging processing and assessment for each strip.
Also on display at the 4-day exhibition held from February 6-9, 2017, in Dubai, UAE was Das SRL’s NEO BIL Plus, an advanced version of traditional bilirubinometers, designed to analyze the 'total bilirubin' through a microcapillary tube. NEO BIL Plus features a special reading system, which can carry out a precise sample measurement of both bilirubin and interferentials at the same point of the capillary using a small quantity of serum and without any reagents.
Latest MEDLAB 2017 News
- AXA Diagnostics Showcases SkyLAB752 ELISA Analyzer
- Norma Diagnostika Showcases Icon Hematology Analyzers at MEDLAB
- AITbiotech Exhibits abTES Molecular Diagnostics Assays at MEDLAB 2017
- Binding Site Unveils Optilite Protein System
- Priorclave Showcases Energy-Efficient Lab Autoclaves in Dubai
- Diatron Exhibits Abacus 5 and Aquila Hematology Analyzers
- DRG Group Presents New Analyzer and ELISAs at Dubai Lab Exhibition
- Beckman Coulter Exhibits New Lab Solutions in Dubai
- Siemens Healthineers Showcases Latest Diagnostic Innovations
Channels
Clinical Chemistry
view channel
Rapid Biosensor Detects Pancreatic Cancer Biomarker for Early Detection
Pancreatic cancer is frequently identified only after it has progressed because early disease is typically asymptomatic, and survival remains extremely poor once advanced. Conventional laboratory assays... Read more
Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
Early detection across multiple cancers remains a major unmet need in population screening. Non-invasive approaches that can be delivered at scale may broaden access and shift diagnoses to earlier stages.... Read moreMolecular Diagnostics
view channel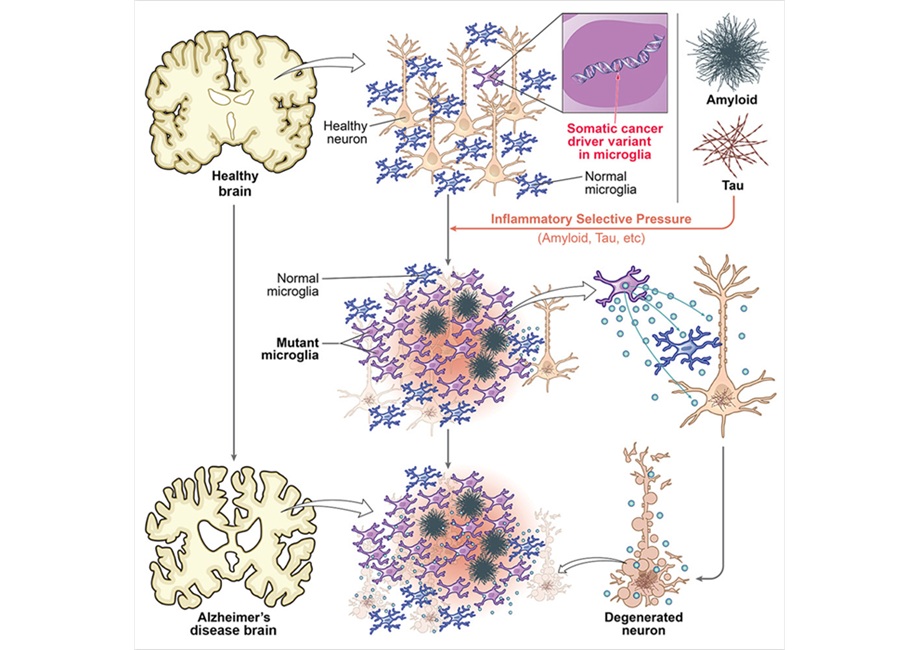
Cancer-Related Mutations in Immune Cells Linked to Alzheimer’s
Alzheimer’s disease is marked by protein aggregation and inflammatory changes in the brain’s immune system, yet its molecular drivers remain incompletely understood. With aging, human cells accumulate... Read more
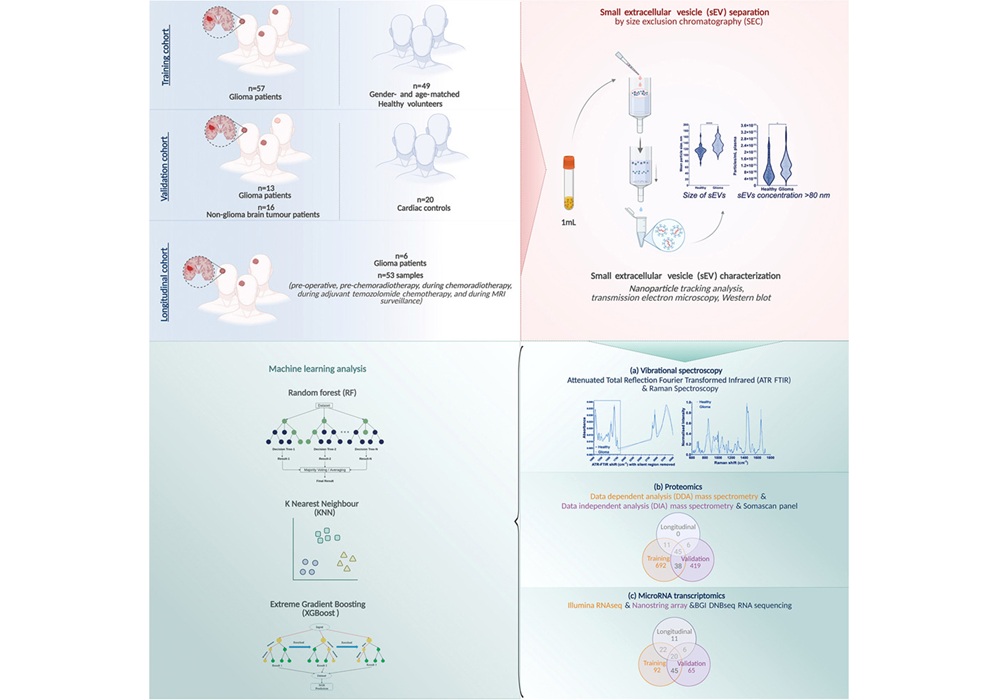
Composite Blood Biomarkers Enable Early Detection of Common Cancers
Early diagnosis of colorectal, lung, and ovarian cancers remains challenging, with many patients identified only after tumors have begun to spread. A scalable blood test could expand access to screening,... Read more
Machine Learning Model Uses DNA Methylation to Predict Tumor Origin in Cancers of Unknown Primary
Cancers of unknown primary (CUP) are metastatic malignancies in which the primary site cannot be identified, complicating treatment selection. Many patients consequently receive broad, nonspecific chemotherapy... Read moreHematology
view channel
Single Assay Enables Rapid HLA and ABO Genotyping for Transplant Matching
CareDx (Brisbane, CA, USA) has introduced AlloSeq Nano, a nanopore‑based HLA (human leukocyte antigen) and ABO genotyping solution unveiled at the European Federation for Immunogenetics (EFI) Conference 2026.... Read more
Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
Diffuse large B-cell lymphoma (DLBCL) is the most common form of non-Hodgkin lymphoma and often presents with aggressive clinical behavior. Although many patients respond to standard chemotherapy with... Read moreImmunology
view channel
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read more
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreMicrobiology
view channel
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read more
Label-Free Microscopy Methodd Enables Faster, Quantitative Detection of Malaria
Microscopy of blood smears remains a cornerstone for malaria diagnosis but can be slow, stain-dependent, and operator intensive. With more than 200 million infections and over 600,000 deaths annually,... Read more
Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
Melanoma remains prone to relapse even after surgery and adjuvant immunotherapy, with 25% to 40% of patients experiencing recurrence. Clinicians lack reliable pre-treatment indicators to identify those... Read more
Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
Gram-negative bloodstream infections and sepsis demand fast, precise antimicrobial therapy, yet conventional susceptibility workflows can delay targeted treatment. Clinical laboratories need platforms... Read morePathology
view channel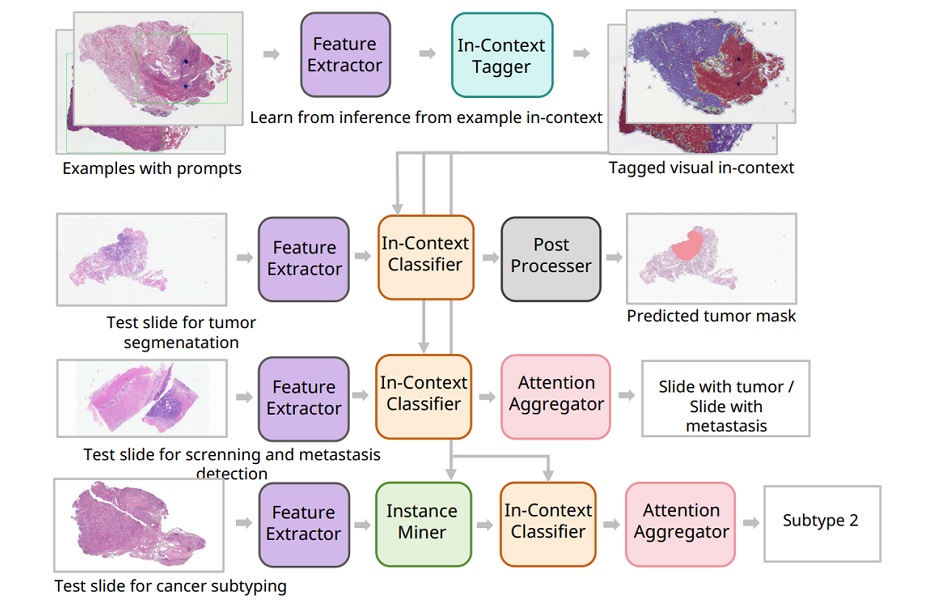
Plug-and-Play AI Pathology System Classifies Multiple Cancers from Few Slides
Pathologists are essential for cancer diagnosis and treatment planning, yet a global workforce shortage is straining services. Nearly 20 million new cases are diagnosed each year, and traditional artificial... Read more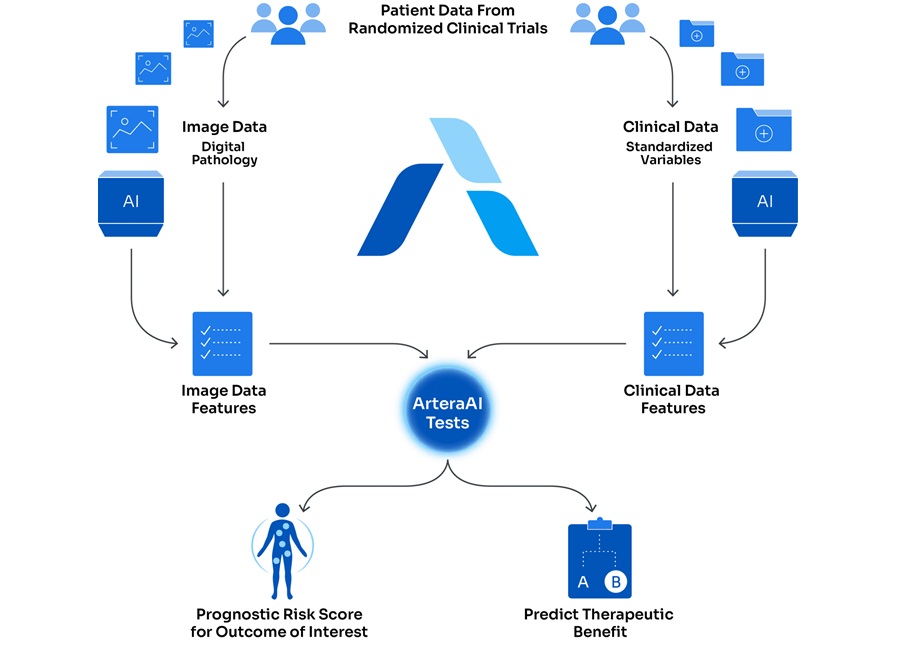
AI-Based Assays Support Risk Stratification in Prostate and Breast Cancer
Prostate and breast cancers are among the most commonly diagnosed malignancies worldwide, and heterogeneous disease biology complicates risk stratification and treatment selection. Clinicians increasingly... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
QuidelOrtho Corporation has completed the acquisition of LEX Diagnostics for approximately USD 100 million in cash. The transaction adds the LEX VELO System to QuidelOrtho’s portfolio. The platform received U.... Read more
Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
Seegene introduced STAgora, a real-time data analytics platform built on aggregated statistical testing data, at ESCMID Global 2026 in Munich, where it also presented an enhanced model of its automated... Read more
Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
Foundation Medicine, Inc., an independent affiliate of Roche, announced plans to expand its monitoring portfolio with SAGA Diagnostics’ Pathlight, a personalized, tumor-informed molecular residual disease... Read more