Dengue Rapid Diagnostic Tests Recycled For Serotyping
|
By LabMedica International staff writers Posted on 25 May 2016 |
Image: The SD Bioline Dengue Duo Rapid Diagnostic Test (RDT) (Photo courtesy of Standard Diagnostics).
Dengue virus infection causes major public health problems in tropical and subtropical areas, but in many endemic areas, inadequate access to laboratory facilities is a major obstacle to surveillance and study of dengue epidemiology.
Dengue Rapid Diagnostic Tests (RDT), in which a drop of blood is loaded onto a paper strip in a plastic cassette, are simple to use and have good diagnostic accuracy. However, four types of dengue virus circulate in most tropical areas and their patterns of circulation are of epidemiological importance since they play a role in the severity and propagation of the disease.
An international team of tropical medicine specialists led by those at Mahidol University (Salaya, Thailand) collected blood samples from 99 consenting patients admitted with symptoms meeting the international criteria for dengue infection from August to November 2013. At another hospital 362 consenting patients with undifferentiated fever who tested negative by malaria RDT were enrolled from July to October 2012, and both venous whole blood and capillary whole blood from finger pricks were collected.
The samples were assayed with the SD Bioline Dengue Duo RDT (Standard Diagnostics, Kyonggi-do, Korea) which is an in vitro immunochromatographic assay for the detection of dengue virus NS1 Ag and anti-dengue IgM/IgG antibodies in human serum, plasma, or whole blood, from finger-prick or venous blood. This test comprises a pair of test devices, a dengue NS1 Ag test on the left side, and a dengue IgM/IgG antibody (Ab) test on the right side. Dengue RNA was purified from the sample pad of the NS1 RDT loaded with virus isolates of the four serotypes, then quantified by real time reverse transcriptase polymerase chain reaction (RT-PCR).
All dengue positive neat, RDT and filter paper (FP) samples were tested by real-time RT-PCR for serotyping. All four DENV serotypes were found, with the majority of patients having DENV-3 (81%; 42/52) followed by DENV-2 (10%; 5/52), DENV-4 (4%; 2/52), and DENV-1(4%; 2/52) with one sample that could not be typed. There was 100% concordance between RDT and serum RT-PCR of infecting dengue serotype. The dengue serotypes at in the rural area at Salavan were mostly DENV-1 (80%; 113/142) followed by DENV-2 (12%; 17/142) and DENV-3 (4%; 6/142).
The authors concluded that their technique may also permit dengue envelope sequencing for deeper molecular epidemiology analysis from RNA purified from RDTs. This could greatly increase availability of dengue epidemiological data from previously inaccessible tropical areas by facilitating dengue confirmation tests and strain identification to aid surveillance and public health interventions. The study was published on May 9, 2016, in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Mahidol University
Standard Diagnostics
Dengue Rapid Diagnostic Tests (RDT), in which a drop of blood is loaded onto a paper strip in a plastic cassette, are simple to use and have good diagnostic accuracy. However, four types of dengue virus circulate in most tropical areas and their patterns of circulation are of epidemiological importance since they play a role in the severity and propagation of the disease.
An international team of tropical medicine specialists led by those at Mahidol University (Salaya, Thailand) collected blood samples from 99 consenting patients admitted with symptoms meeting the international criteria for dengue infection from August to November 2013. At another hospital 362 consenting patients with undifferentiated fever who tested negative by malaria RDT were enrolled from July to October 2012, and both venous whole blood and capillary whole blood from finger pricks were collected.
The samples were assayed with the SD Bioline Dengue Duo RDT (Standard Diagnostics, Kyonggi-do, Korea) which is an in vitro immunochromatographic assay for the detection of dengue virus NS1 Ag and anti-dengue IgM/IgG antibodies in human serum, plasma, or whole blood, from finger-prick or venous blood. This test comprises a pair of test devices, a dengue NS1 Ag test on the left side, and a dengue IgM/IgG antibody (Ab) test on the right side. Dengue RNA was purified from the sample pad of the NS1 RDT loaded with virus isolates of the four serotypes, then quantified by real time reverse transcriptase polymerase chain reaction (RT-PCR).
All dengue positive neat, RDT and filter paper (FP) samples were tested by real-time RT-PCR for serotyping. All four DENV serotypes were found, with the majority of patients having DENV-3 (81%; 42/52) followed by DENV-2 (10%; 5/52), DENV-4 (4%; 2/52), and DENV-1(4%; 2/52) with one sample that could not be typed. There was 100% concordance between RDT and serum RT-PCR of infecting dengue serotype. The dengue serotypes at in the rural area at Salavan were mostly DENV-1 (80%; 113/142) followed by DENV-2 (12%; 17/142) and DENV-3 (4%; 6/142).
The authors concluded that their technique may also permit dengue envelope sequencing for deeper molecular epidemiology analysis from RNA purified from RDTs. This could greatly increase availability of dengue epidemiological data from previously inaccessible tropical areas by facilitating dengue confirmation tests and strain identification to aid surveillance and public health interventions. The study was published on May 9, 2016, in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Mahidol University
Standard Diagnostics
Latest Microbiology News
- Gut Microbiome Signatures Help Identify Risk of IBD Progression
- FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
- New AMR Assay Supports Rapid Infection Control Screening in Hospitals
- Diagnostic Gaps Complicate Bundibugyo Ebola Outbreak Response in Congo
- Study Finds Hidden Mpox Infections May Drive Ongoing Spread
- Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
- Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
- Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
- Label-Free Microscopy Method Enables Faster, Quantitative Detection of Malaria
- Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
Channels
Clinical Chemistry
view channel
Urine-Based Test Shows Promise for Autism Screening in Children
Autism spectrum disorder (ASD) is commonly diagnosed through behavioral assessments, which can involve long waits that delay intervention. Earlier identification is linked to better developmental outcomes,... Read more
Liquid Biopsy Biomarkers May Improve Childhood Epilepsy Diagnosis
Childhood epilepsy remains a major neurological disorder with unmet needs for accurate, non-invasive biomarkers, as conventional tests such as electroencephalography and neuroimaging can have limited sensitivity... Read moreMolecular Diagnostics
view channel
Tumor Mutation Marker Helps Refine Lung Cancer Prognosis and Guide Therapy Selection
Lung cancer remains the leading cause of cancer death worldwide, while heterogeneous tumor genetics continue to complicate treatment decisions. Although molecular testing is increasingly used to match... Read more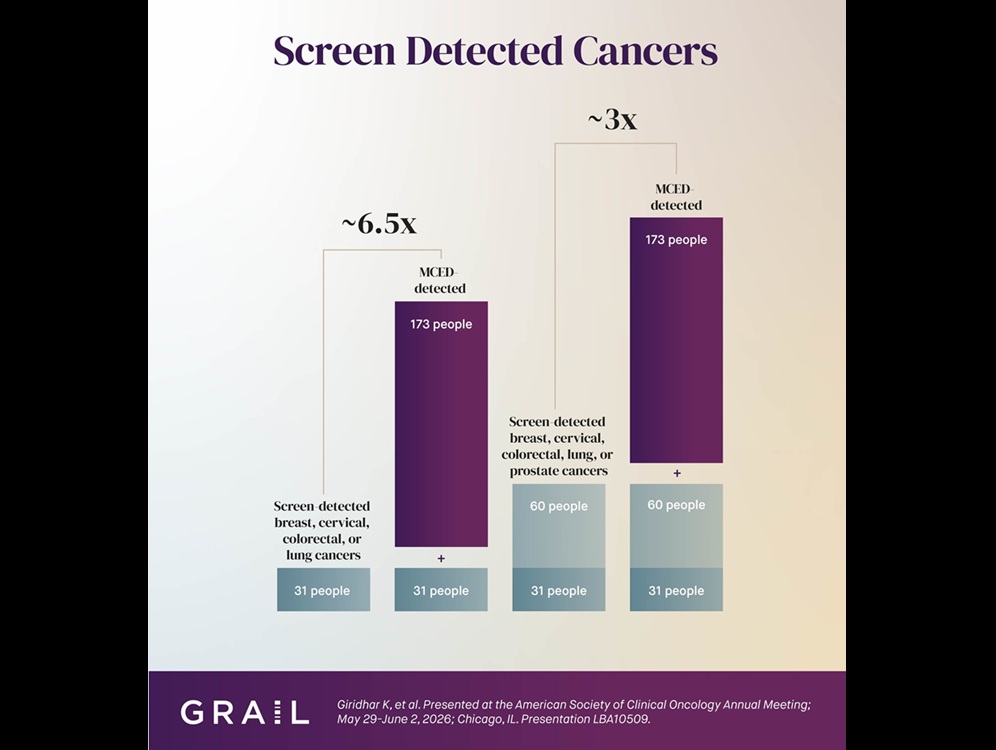
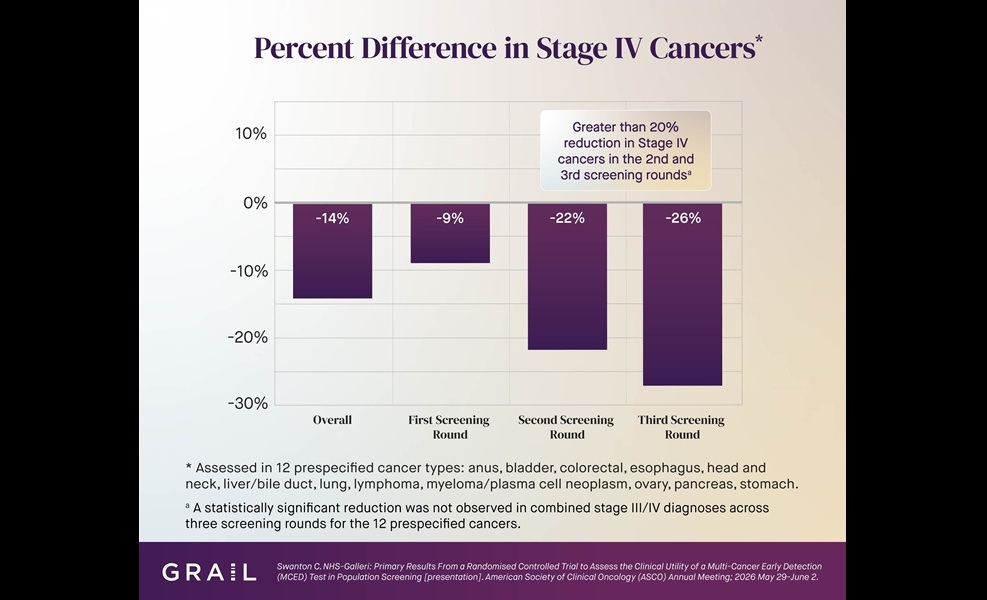
Multi-Cancer Test Boosts Detection When Added to Standard Screening
Most cancers are still diagnosed outside organ-specific screening, often at advanced stages. In the United States, guideline-recommended screening detects only 14% of cancers, underscoring the need for... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channel
Study Points to Autoimmune Pathway Behind Long COVID Symptoms
Long COVID leaves many SARS-CoV-2 survivors with persistent fatigue, cognitive issues, palpitations, and musculoskeletal pain for months or years. Estimates cited in new research suggest 4%–20% of infected... Read more
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read morePathology
view channel
AI-Powered Atlas Maps Immune Structures Linked to Cancer Outcomes
Tertiary lymphoid structures are emerging as important indicators of antitumor immunity, but their heterogeneity and spatial context within tumors remain difficult to capture through routine diagnostics.... Read more
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channelPartnership Expands Access to Alzheimer’s Blood Tests in Latin America and Caribbean
Alzheimer’s disease assessment remains challenging in many regions where aging populations are increasing demand for care, but access to dementia specialists and advanced imaging remains limited.... Read more




.jpg)