Potential Noninvasive Biomarker of EoE Disease Activity Identified
|
By LabMedica International staff writers Posted on 24 May 2016 |
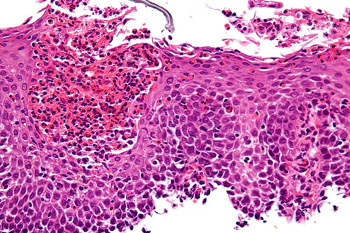
Image: A micrograph of eosinophilic esophagitis (EoE), H&E stain. Characteristic features are present: Abundant eosinophils – criteria vary; one common definition is: > 20 eosinophils/0.24 mm2; Papillae are elongated; papillae reach into the top 1/3 of the epithelial layer; Basal cell hyperplasia; > 3 cells thick or >15% of epithelial thickness; Spongiosis (Photo courtesy of Michael Bonert / Wikimedia).
Researchers have identified a potential blood-based marker of disease activity for the severe and often painful food allergic disease eosinophilic esophagitis (EoE) – possibly leading to a blood test, which could spare EoE patients, often children, the discomfort and risk of the currently used invasive endoscopic monitoring procedures.
Researchers at the Cincinnati Center for Eosinophilic Disorders (CCED) of Cincinnati Children’s Hospital Medical Center (Cincinnati, OH, USA) led the study. “Adults and children with EoE can be on highly restricted diets of formula alone or only a few foods,” said Patricia C. Fulkerson, MD, PhD, senior study author, “One of the major obstacles to families participating in studies to introduce foods back into the child’s diet is the need for endoscopy after each food is tried to see whether or not it triggers disease activity.”
The disease activity of EoE is currently monitored using peak esophageal eosinophil count, which requires endoscopy to collect esophageal tissue biopsies. People with EoE, a lifelong disease, must continue monitoring disease activity, even after effective treatment with restricted diets or steroids. Treatment changes, such as reintroducing a single food, require additional endoscopic exams to assess for disease flare-ups.
Prior research has demonstrated that testing blood of EoE patients is not a clinically useful indication of active disease because eosinophil levels in blood do not correlate well with levels in the esophagus. This led the team to investigate a precursor cell to eosinophils, a lineage-committed eosinophil progenitor (EoP), as a potential marker. They found elevated EoP levels in the blood of pediatric patients with active EoE disease, suggesting a promising, blood-based marker.
The authors emphasize that additional research is needed to validate the marker before routine clinical use. “This clinical study is the first to investigate EoP levels in patients with EoE and identifies a potential new noninvasive biomarker,” said study author Vincent A. Mukkada, MD, physician at Cincinnati Children’s and CCED member, “This work is an essential step toward improving outcomes for patients with EoE. It will be followed by repeated testing of more patients and with sequential measurements of EoP levels in the same patient during different disease states.”
Allergic diseases have been on the rise over the past 20 years. The CCED team has previously reported that incidence of EoE is estimated at 1 of 1,000 people. Their research has also shown that EoE is caused by a combination of genetic and environmental factors, and is primarily mediated by an immunologic response to foods. The hallmark of EoE is swelling and inflammation in the esophagus, accompanied by high levels of eosinophils.
The study, by Morris DW et al, was published May 16, 2016, in the Journal of Allergy and Clinical Immunology.
Related Links:
Cincinnati Children’s Hospital Medical Center
Researchers at the Cincinnati Center for Eosinophilic Disorders (CCED) of Cincinnati Children’s Hospital Medical Center (Cincinnati, OH, USA) led the study. “Adults and children with EoE can be on highly restricted diets of formula alone or only a few foods,” said Patricia C. Fulkerson, MD, PhD, senior study author, “One of the major obstacles to families participating in studies to introduce foods back into the child’s diet is the need for endoscopy after each food is tried to see whether or not it triggers disease activity.”
The disease activity of EoE is currently monitored using peak esophageal eosinophil count, which requires endoscopy to collect esophageal tissue biopsies. People with EoE, a lifelong disease, must continue monitoring disease activity, even after effective treatment with restricted diets or steroids. Treatment changes, such as reintroducing a single food, require additional endoscopic exams to assess for disease flare-ups.
Prior research has demonstrated that testing blood of EoE patients is not a clinically useful indication of active disease because eosinophil levels in blood do not correlate well with levels in the esophagus. This led the team to investigate a precursor cell to eosinophils, a lineage-committed eosinophil progenitor (EoP), as a potential marker. They found elevated EoP levels in the blood of pediatric patients with active EoE disease, suggesting a promising, blood-based marker.
The authors emphasize that additional research is needed to validate the marker before routine clinical use. “This clinical study is the first to investigate EoP levels in patients with EoE and identifies a potential new noninvasive biomarker,” said study author Vincent A. Mukkada, MD, physician at Cincinnati Children’s and CCED member, “This work is an essential step toward improving outcomes for patients with EoE. It will be followed by repeated testing of more patients and with sequential measurements of EoP levels in the same patient during different disease states.”
Allergic diseases have been on the rise over the past 20 years. The CCED team has previously reported that incidence of EoE is estimated at 1 of 1,000 people. Their research has also shown that EoE is caused by a combination of genetic and environmental factors, and is primarily mediated by an immunologic response to foods. The hallmark of EoE is swelling and inflammation in the esophagus, accompanied by high levels of eosinophils.
The study, by Morris DW et al, was published May 16, 2016, in the Journal of Allergy and Clinical Immunology.
Related Links:
Cincinnati Children’s Hospital Medical Center
Latest Pathology News
- AI-Powered Atlas Maps Immune Structures Linked to Cancer Outcomes
- AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
- Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
- AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
- FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
- New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
- AI System Analyzes Routine Pathology Slides to Predict Cancer Outcomes
- New Tissue Mapping Approach Identifies High-Risk Form of Diabetic Kidney Disease
- Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
- Interpretable AI Reveals Hidden Cellular Features from Microscopy Images
- Tumor Immune Structure Predicts Response to Immunotherapy in Melanoma
- Plug-and-Play AI Pathology System Classifies Multiple Cancers from Few Slides
- AI-Based Assays Support Risk Stratification in Prostate and Breast Cancer
- AI Pathology Model Predicts Immunotherapy Response in Lung Cancer
- Study Reveals Moleclar Mechanism Driving Aggressive Skin Cancer
- AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
Channels
Clinical Chemistry
view channel
Alzheimer’s Blood Biomarkers Linked to Early Cognitive Differences Before Dementia
Blood-based screening for Alzheimer’s disease offers a noninvasive, lower-cost alternative to brain imaging or spinal fluid testing, yet its ability to flag the earliest cognitive changes has been unclear.... Read more
Urine-Based Test Shows Promise for Autism Screening in Children
Autism spectrum disorder (ASD) is commonly diagnosed through behavioral assessments, which can involve long waits that delay intervention. Earlier identification is linked to better developmental outcomes,... Read moreMolecular Diagnostics
view channel
Simple Cytogenetic Method Could Improve Classification of ALL Subtypes
Many cancers deviate from the normal chromosome number, but the clinical impact of extreme chromosome loss remains unclear. This widespread genomic disruption is associated with aggressive disease and... Read more
Blood-Based Assay Enables Noninvasive Monitoring of Sarcoma Immunotherapy Response
Sarcomas remain difficult to monitor during immunotherapy, as low tumor mutation burden can limit traditional circulating tumor DNA approaches and repeat tissue biopsies are often impractical in advanced disease.... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read morePathology
view channel
AI-Powered Atlas Maps Immune Structures Linked to Cancer Outcomes
Tertiary lymphoid structures are emerging as important indicators of antitumor immunity, but their heterogeneity and spatial context within tumors remain difficult to capture through routine diagnostics.... Read more
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channel