Microfluidic Device Rapidly Detects Urinary Tract Infections
|
By LabMedica International staff writers Posted on 23 Aug 2015 |
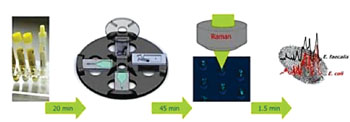
Image: The workflow of the Lab-on-a-Disc device for identifying bacteria in urine samples. From left to right (i) Urine sample from patient; (ii) Capturing bacteria in V-cup-structures on a Lab-on-a-Disk platform by centrifugation; (iii) Raman spectroscopic analysis of captured bacteria within the V-cups; (iv) Fingerprint-like spectroscopic information on the UTI pathogen (Photo courtesy of Ute Neugebauer).

Image: The alpha300 R Superior Confocal Raman Imaging System (Photo courtesy of WITec).
A polymeric centrifugal microfluidic platform has been developed for the rapid and sensitive identification of bacteria directly from urine, thus eliminating time-consuming cultivation steps.
Untreated urinary tract infections can quickly move to a life-threatening condition and cases may trigger sepsis, which occurs when the immune system, in an attempt to fight off the infection, inadvertently activates body-wide inflammation that can cause blood clots and leaky blood vessels.
Scientists at the Jena University Hospital (Germany) have created a Lab-on-a-Disc platform that combines microfluidics and Raman microscopy, a modern optical detection method. The platform utilizes the rotationally induced centrifugal field to efficiently capture bacteria directly from suspension within a glass-polymer hybrid chip. Once trapped in an array of small V-shaped structures, the bacteria are readily available for spectroscopic characterization, such as Raman spectroscopic fingerprinting, providing valuable information on the characteristics of the captured bacteria.
Anonymized urine samples were provided by the hospital’s Institute of Medical Microbiology. They originated from different patients with single pathogen urinary tract infections (UTIs) of Enterococcus faecalis and Escherichia coli. To remove bigger particles such as leukocytes or epithelial cells, the urine samples were run through membrane filters, centrifuged, the pellet washed twice with phosphate buffered saline (PBS), and is finally re-suspended in PBS before being loaded into the device. A CRM 300 micro-Raman setup (WITec, Ulm, Germany), equipped with a 600 lines/mm grating was used for micro-Raman measurements.
The whole procedure, including sample preparation, requires about one hour to obtain a valuable result, marking a significant reduction in diagnosis time when compared to the 24 hours and more, typically required for standard microbiological methods. Characterization of the captured bacteria by label-free conventional micro-Raman spectroscopy allows rapid identification of the pathogens with their characteristic features, which is valuable for first screening analysis. The device has been easily adapted for fluorescence measurements, paving the way for the development of microfluidics-based immunochemical assays, illustrating a high potential of the device for numerous applications in spectroscopy-based point-of-care diagnostics.
Ulrich-Christian Schröder, a doctoral student and lead author of the study said, “Our device works by loading a few microliters of a patient's urine sample into a tiny chip, which is then rotated with a high angular velocity so that any bacteria is guided by centrifugal force through microfluidic channels to a small chamber where 'V-cup capture units' collect it for optical investigation. We were able to identify Escherichia coli and Enterococcus faecalis within 70 minutes, directly from patients' urine samples.” The study was published on August 11, 2015, in the journal Biomicrofluidics.
Related Links:
Jena University Hospital
WITec
Untreated urinary tract infections can quickly move to a life-threatening condition and cases may trigger sepsis, which occurs when the immune system, in an attempt to fight off the infection, inadvertently activates body-wide inflammation that can cause blood clots and leaky blood vessels.
Scientists at the Jena University Hospital (Germany) have created a Lab-on-a-Disc platform that combines microfluidics and Raman microscopy, a modern optical detection method. The platform utilizes the rotationally induced centrifugal field to efficiently capture bacteria directly from suspension within a glass-polymer hybrid chip. Once trapped in an array of small V-shaped structures, the bacteria are readily available for spectroscopic characterization, such as Raman spectroscopic fingerprinting, providing valuable information on the characteristics of the captured bacteria.
Anonymized urine samples were provided by the hospital’s Institute of Medical Microbiology. They originated from different patients with single pathogen urinary tract infections (UTIs) of Enterococcus faecalis and Escherichia coli. To remove bigger particles such as leukocytes or epithelial cells, the urine samples were run through membrane filters, centrifuged, the pellet washed twice with phosphate buffered saline (PBS), and is finally re-suspended in PBS before being loaded into the device. A CRM 300 micro-Raman setup (WITec, Ulm, Germany), equipped with a 600 lines/mm grating was used for micro-Raman measurements.
The whole procedure, including sample preparation, requires about one hour to obtain a valuable result, marking a significant reduction in diagnosis time when compared to the 24 hours and more, typically required for standard microbiological methods. Characterization of the captured bacteria by label-free conventional micro-Raman spectroscopy allows rapid identification of the pathogens with their characteristic features, which is valuable for first screening analysis. The device has been easily adapted for fluorescence measurements, paving the way for the development of microfluidics-based immunochemical assays, illustrating a high potential of the device for numerous applications in spectroscopy-based point-of-care diagnostics.
Ulrich-Christian Schröder, a doctoral student and lead author of the study said, “Our device works by loading a few microliters of a patient's urine sample into a tiny chip, which is then rotated with a high angular velocity so that any bacteria is guided by centrifugal force through microfluidic channels to a small chamber where 'V-cup capture units' collect it for optical investigation. We were able to identify Escherichia coli and Enterococcus faecalis within 70 minutes, directly from patients' urine samples.” The study was published on August 11, 2015, in the journal Biomicrofluidics.
Related Links:
Jena University Hospital
WITec
Latest Microbiology News
- Gut Microbiome Signatures Help Identify Risk of IBD Progression
- FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
- New AMR Assay Supports Rapid Infection Control Screening in Hospitals
- Diagnostic Gaps Complicate Bundibugyo Ebola Outbreak Response in Congo
- Study Finds Hidden Mpox Infections May Drive Ongoing Spread
- Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
- Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
- Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
- Label-Free Microscopy Method Enables Faster, Quantitative Detection of Malaria
- Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
Channels
Clinical Chemistry
view channel
Urine-Based Test Shows Promise for Autism Screening in Children
Autism spectrum disorder (ASD) is commonly diagnosed through behavioral assessments, which can involve long waits that delay intervention. Earlier identification is linked to better developmental outcomes,... Read more
Liquid Biopsy Biomarkers May Improve Childhood Epilepsy Diagnosis
Childhood epilepsy remains a major neurological disorder with unmet needs for accurate, non-invasive biomarkers, as conventional tests such as electroencephalography and neuroimaging can have limited sensitivity... Read moreMolecular Diagnostics
view channel
Updated Guidance Prioritizes Stool-Based Colorectal Cancer Screening Tests
Colorectal cancer is the second-leading cause of cancer death in the United States and claimed an estimated 55,000 lives in 2026. Incidence is rising among adults younger than 50, even as overall mortality... Read more
Digital PCR Assays Support Surveillance of Bundibugyo Ebolavirus Outbreak
QIAGEN (Venlo, Netherlands) has introduced two custom-designed research-use-only (RUO) QIAcuity dPCR assays to support infectious disease research and surveillance connected to the Bundibugyo ebolavirus outbreak.... Read more
Blood-Based Proteomic Test May Predict Treatment Response in Non-Small Cell Lung Cancer
Lung cancer remains the leading cause of cancer death, with non-small cell lung cancer (NSCLC) accounting for most cases. Treatment decisions are often made without a clear indication of how a patient... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channel
Immune Enzyme Linked to Treatment-Resistant Inflammatory Bowel Disease
Inflammatory bowel disease (IBD) affects nearly 3 million people in the United States and its prevalence continues to rise. Medications that target tumor necrosis factor (TNF)-alpha are widely used, but... Read more
Simple Blood Test Could Replace Biopsies for Lung Transplant Rejection Monitoring
Lung transplant recipients face some of the highest rates of acute cellular rejection, and routine surveillance often relies on repeated surgical biopsies. These procedures can cause complications such... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read morePathology
view channel
AI-Powered Atlas Maps Immune Structures Linked to Cancer Outcomes
Tertiary lymphoid structures are emerging as important indicators of antitumor immunity, but their heterogeneity and spatial context within tumors remain difficult to capture through routine diagnostics.... Read more
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read moreIndustry
view channelWerfen and Oxford Nanopore Collaborate on Transplant Assay Development
Werfen (Barcelona, Spain), a global specialized diagnostics company, has announced a strategic collaboration with Oxford Nanopore Technologies (Oxford, UK), which develops nanopore-based sequencing technology,... Read more
























