Molecular Urine Test Identifies Lung Carcinoma Mutations
|
By LabMedica International staff writers Posted on 20 May 2015 |
The monitoring of circulating tumor DNA (ctDNA) is a promising technique that may provide clinicians with a faster, cheaper and less invasive way to evaluate the clinical status and response to therapy of cancer patients.
Disease progression in patients with metastatic non-small-cell lung cancer (NSCLC) is often heralded by the acquisition of epidermal growth factor receptor (EGFR) T790M resistance mutation following treatment with anti-EGFR inhibitors.
Scientists at Moores Cancer Center (La Jolla, CA, USA) took urine samples from patients with metastatic NSCLC who progressed on erlotinib treatment and ctDNA was extracted by a method that preferentially isolates short, fragmented ctDNA. Droplet digital polymerase chain reaction system (Bio-Rad; Hercules, CA, USA) was used to quantify the ctDNA and yielded an average of total amplifiable ctDNA per sample of 0.4 µg (range, 0.04 µg to 2.4 µg). Spiked cell lines were used for analytical characterization and demonstrated that the EGFR T790M assay had a lower limit of detection of two copies within a background of 60 ng of wild-type DNA, yielding an analytical sensitivity of 0.01%. EGFR status was analyzed using a PCR method that amplifies short target DNA fragments using kinetically-favorable binding conditions for a wild type blocking oligonucleotide, followed by massively parallel deep sequencing using a MiSeq desktop sequencer (Illumina; San Diego CA, USA).
EGFR T790M mutation in urine was detected in 15 of 22 (68%) of patients receiving anti-EGFR treatment until progression. Urine EGFR T790M mutation was detected in 10 out of 10 tissue-positive patients, giving 100% concordance. Urine ctDNA testing identified five additional patients who may be eligible for treatment with anti-T790M drugs, three of whom were tissue negative. The investigators showed that EGFR T790M mutation can be detected in urinary ctDNA up to three months before radiographic progression on first-line anti-EGFR tyrosine kinase inhibitor (TKI). When T790M positive patients were treated with third generation anti-EGFR TKIs, a decrease in ctDNA T790M load was observed as early as four hours after therapy on first day of treatment. The initial decrease in urinary T790M was followed by a spike in T790M during the first week of therapy.
Hatim Husain, MD, the lead author of the study and his colleagues concluded that this ctDNA assay for multiple genes via next-generation sequencing (NGS) might become a "liquid biopsy" that could serve as an alternative to invasive tissue biopsy. EGFR T790M mutation in urine was detected months before radiographic detection, and furthermore, urine ctDNA testing identified tissue negative patients who may be eligible for treatment with third generation anti-EGFR TKIs and monitoring of urinary T790M dynamics may predict clinical benefit or initial tumour burden. The study was presented at the EUROPEAN Lung Cancer Conference held April 15–18, 2015, in Geneva (Switzerland).
Related Links:
Moores Cancer Center
Bio-Rad
Illumina
Disease progression in patients with metastatic non-small-cell lung cancer (NSCLC) is often heralded by the acquisition of epidermal growth factor receptor (EGFR) T790M resistance mutation following treatment with anti-EGFR inhibitors.
Scientists at Moores Cancer Center (La Jolla, CA, USA) took urine samples from patients with metastatic NSCLC who progressed on erlotinib treatment and ctDNA was extracted by a method that preferentially isolates short, fragmented ctDNA. Droplet digital polymerase chain reaction system (Bio-Rad; Hercules, CA, USA) was used to quantify the ctDNA and yielded an average of total amplifiable ctDNA per sample of 0.4 µg (range, 0.04 µg to 2.4 µg). Spiked cell lines were used for analytical characterization and demonstrated that the EGFR T790M assay had a lower limit of detection of two copies within a background of 60 ng of wild-type DNA, yielding an analytical sensitivity of 0.01%. EGFR status was analyzed using a PCR method that amplifies short target DNA fragments using kinetically-favorable binding conditions for a wild type blocking oligonucleotide, followed by massively parallel deep sequencing using a MiSeq desktop sequencer (Illumina; San Diego CA, USA).
EGFR T790M mutation in urine was detected in 15 of 22 (68%) of patients receiving anti-EGFR treatment until progression. Urine EGFR T790M mutation was detected in 10 out of 10 tissue-positive patients, giving 100% concordance. Urine ctDNA testing identified five additional patients who may be eligible for treatment with anti-T790M drugs, three of whom were tissue negative. The investigators showed that EGFR T790M mutation can be detected in urinary ctDNA up to three months before radiographic progression on first-line anti-EGFR tyrosine kinase inhibitor (TKI). When T790M positive patients were treated with third generation anti-EGFR TKIs, a decrease in ctDNA T790M load was observed as early as four hours after therapy on first day of treatment. The initial decrease in urinary T790M was followed by a spike in T790M during the first week of therapy.
Hatim Husain, MD, the lead author of the study and his colleagues concluded that this ctDNA assay for multiple genes via next-generation sequencing (NGS) might become a "liquid biopsy" that could serve as an alternative to invasive tissue biopsy. EGFR T790M mutation in urine was detected months before radiographic detection, and furthermore, urine ctDNA testing identified tissue negative patients who may be eligible for treatment with third generation anti-EGFR TKIs and monitoring of urinary T790M dynamics may predict clinical benefit or initial tumour burden. The study was presented at the EUROPEAN Lung Cancer Conference held April 15–18, 2015, in Geneva (Switzerland).
Related Links:
Moores Cancer Center
Bio-Rad
Illumina
Read the full article by registering today, it's FREE! 

Register now for FREE to LabMedica.com and get access to news and events that shape the world of Clinical Laboratory Medicine. 
- Free digital version edition of LabMedica International sent by email on regular basis
- Free print version of LabMedica International magazine (available only outside USA and Canada).
- Free and unlimited access to back issues of LabMedica International in digital format
- Free LabMedica International Newsletter sent every week containing the latest news
- Free breaking news sent via email
- Free access to Events Calendar
- Free access to LinkXpress new product services
- REGISTRATION IS FREE AND EASY!
Sign in: Registered website members
Sign in: Registered magazine subscribers
Latest Pathology News
- AI Improves Completeness of Complex Cancer Pathology Reports
- AI Tool Predicts Chemotherapy Response in Small Cell Lung Cancer
- Tumor-Specific Biomarker Predicts Neoadjuvant Immunotherapy Response in Gastric Cancer
- AI Tool Predicts Patient-Specific Chemotherapy Benefit in Breast Cancer
- AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
- Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
- New Chromogenic Culture Media Enable Rapid Detection of Candida Infections
- AI-Powered Tool to Transform Dermatopathology Workflow
- AI Tool Predicts Chemotherapy Response from Biopsy Slides
- Sex Differences in Alzheimer’s Biomarkers Linked to Faster Cognitive Decline
- World’s First Optical Microneedle Device to Enable Blood-Sampling-Free Clinical Testing
- Novel mcPCR Technology to Transform Testing of Clinical Samples
- Pathogen-Agnostic Testing Reveals Hidden Respiratory Threats in Negative Samples
- Molecular Imaging to Reduce Need for Melanoma Biopsies
- Urine Specimen Collection System Improves Diagnostic Accuracy and Efficiency
- AI-Powered 3D Scanning System Speeds Cancer Screening
Channels
Clinical Chemistry
view channel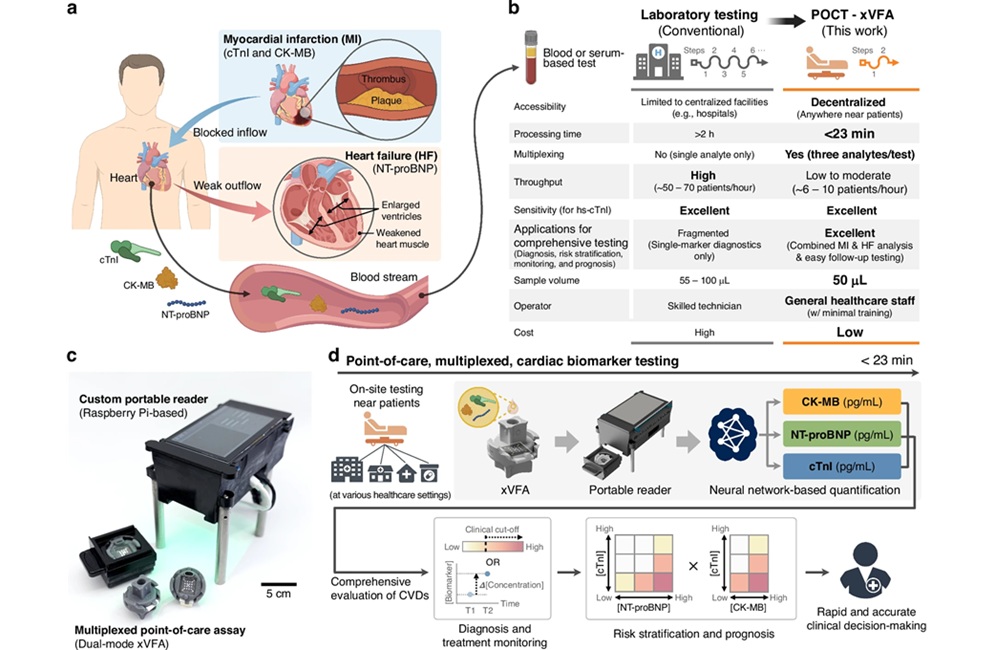
AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
Cardiovascular diseases are a leading cause of death, responsible for nearly 20 million deaths each year. Timely triage of myocardial infarction and heart failure hinges on rapid cardiac biomarker measurement,... Read moreNext Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
Clinical laboratories continue to face staffing shortages, limited space, and growing test volumes that pressure chemistry and electrolyte workflows. Maintaining rapid turnaround times increasingly depends... Read moreMolecular Diagnostics
view channel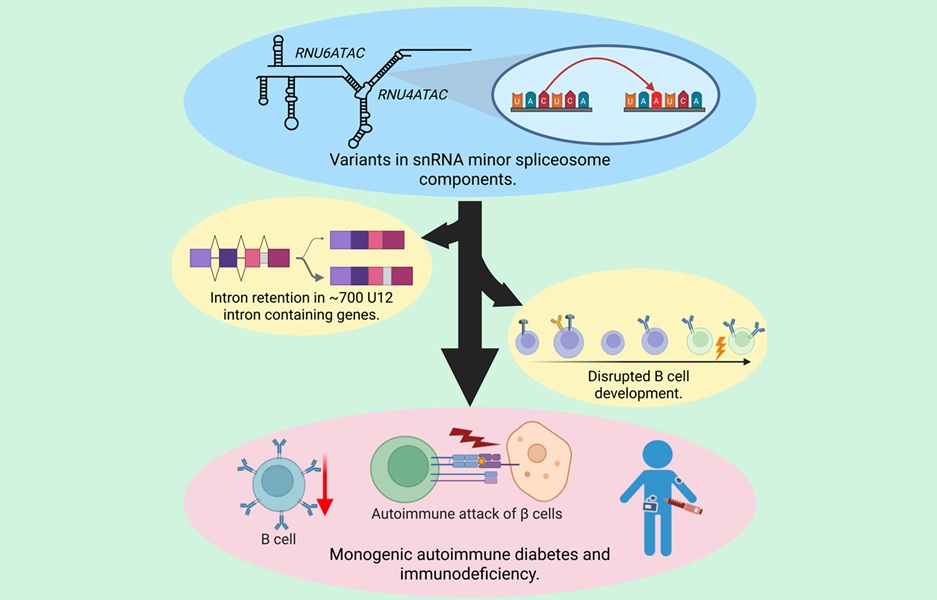
Genome Sequencing Identifies Noncoding Variants Causing Neonatal Diabetes
Neonatal diabetes is a rare form of diabetes that presents in the first six months of life and is driven by genetic changes, yet many affected families still lack definitive diagnoses. Genetic studies... Read more
Genetic Markers Predict GLP-1 Weight-Loss Response and Side Effects
Glucagon-like peptide-1 (GLP-1) receptor agonists such as semaglutide and tirzepatide are now widely used for weight management, yet individual responses vary considerably, with some patients experiencing... Read moreHematology
view channel
Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
Chronic myelomonocytic leukemia (CMML) is a rare blood cancer in which acquired genetic mutations in bone marrow stem cells drive disease. Stem cell transplantation is the only curative option but carries... Read more
New Platelet Function Assay Enables Monitoring of Antiplatelet Therapy
Monitoring response to antiplatelet therapy remains challenging for many clinical laboratories. Aggregation-based assays and cartridge systems often require specialized personnel, dedicated instruments,... Read moreImmunology
view channelCombined Screening Approach Identifies Early Leprosy Cases
Leprosy remains a significant public health concern, with more than 200,000 new cases reported globally each year and early disease often escaping routine laboratory detection. In its initial phase, bacterial... Read more
Antibody Blood Test Identifies Active TB and Distinguishes Latent Infection
Active tuberculosis (TB) remains a leading cause of death and illness worldwide, yet distinguishing contagious disease from latent infection continues to challenge clinicians. Standard screening tools... Read more
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel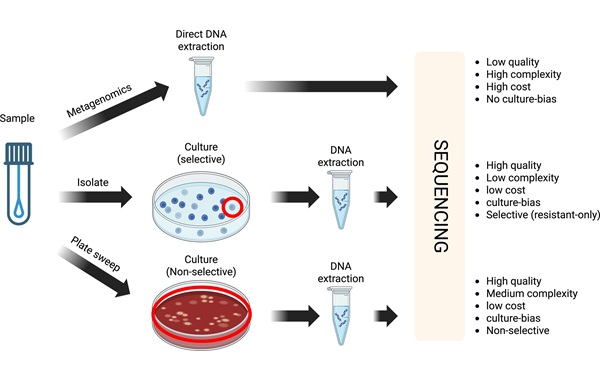
Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
Intensive care units face persistent threats from hospital-acquired infections, increasingly driven by drug-resistant bacteria. Rapidly pinpointing environmental reservoirs and transmission hotspots remains... Read more
New Bacterial Target Identified for Early Detection of Noma
Noma is a rapidly progressing orofacial infection that begins as gingivitis and can destroy oral and facial tissues, primarily affecting young children living in extreme poverty. Without treatment, it... Read moreTechnology
view channel
Noninvasive Sputum Test Detects Early Lung Cancer
Early detection remains critical for improving outcomes in lung cancer, yet clinicians increasingly encounter indeterminate pulmonary nodules found incidentally or through screening, complicating decision-making.... Read more
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channel
GRAIL Partners with Epic to Integrate Multi-Cancer Test into EHR
GRAIL’s Galleri multi-cancer early detection (MCED) test is being integrated into Epic’s electronic health record (EHR) platform through Epic Aura. The collaboration is designed to let clinicians at interested... Read moreGlobal Partnership Aims to Streamline NGS Tumor Profiling in Oncology Trials
CellCarta and Pillar Biosciences announced a global, multi-year strategic partnership on April 2, 2026 to broaden access to operationally streamlined next-generation sequencing (NGS) tumor profiling for... Read more