Novel Biomarker Identified for Two Different Cancers
|
By LabMedica International staff writers Posted on 17 Apr 2014 |
Image: Histopathology of laryngeal squamous cell carcinoma (Photo courtesy of Masaryk University).
A new biomarker linked to better outcomes for patients with head and neck cancers and non-small-cell lung cancer (NSCLC) has been identified.
The biomarker could help scientists develop new diagnostics and therapies and help physicians determine the best long-term treatments for patients with these cancers.
Scientists from the University of Pittsburgh (PA, USA) examined tumor samples from 187 patients with non-small-cell lung cancer and 60 patients with head and neck squamous cell carcinomas (HNSCC). The median follow-up for overall survival (OS) and disease-free survival (DFS) for NSCLC patients was 69.0 months and 35.9 months, respectively. The median follow-up for HNSCC patients was 41 months.
The team used antibody-based immunological techniques, cell cultures, immunoprecipitation, immunohistochemistry and in situ quantification. For tumor analysis, tissue microarrays of formalin-fixed and paraffin-embedded tumors and control tissues were used. Each tumor was represented by three distinct cores for NSCLC and two for HNSCC. The nuclear signal intensity was quantified by automated quantitative analysis for NSCLC and Aperio software for HNSCC (Aperio Technologies, Vista, CA, USA).
The investigators found that the expression of a protein called choline phosphate cytidylyltransferase-α CCT-α or CCTα, which acts as an “antigen” that prompts the immune system to produce antibodies against it. CCTα was associated with longer survival rates, including for patients with NSCLC who were treated with surgery alone, without the use of platinum-based chemotherapy drugs and associated toxic side effects.
Laura J. Niedernhofer, MD, PhD, an associate professor and a senior author of the study said, “Based on what we found, a high CCTα expression appears to be indicative of survival, making CCTα a promising biomarker. Our findings suggest that CCTα may, in fact, be more important in determining outcomes in patients with both types of cancer than the already established excision repair cross-complementation group 1 (ERCC1) protein.”
Related Links:
University of Pittsburgh
Aperio Technologies
The biomarker could help scientists develop new diagnostics and therapies and help physicians determine the best long-term treatments for patients with these cancers.
Scientists from the University of Pittsburgh (PA, USA) examined tumor samples from 187 patients with non-small-cell lung cancer and 60 patients with head and neck squamous cell carcinomas (HNSCC). The median follow-up for overall survival (OS) and disease-free survival (DFS) for NSCLC patients was 69.0 months and 35.9 months, respectively. The median follow-up for HNSCC patients was 41 months.
The team used antibody-based immunological techniques, cell cultures, immunoprecipitation, immunohistochemistry and in situ quantification. For tumor analysis, tissue microarrays of formalin-fixed and paraffin-embedded tumors and control tissues were used. Each tumor was represented by three distinct cores for NSCLC and two for HNSCC. The nuclear signal intensity was quantified by automated quantitative analysis for NSCLC and Aperio software for HNSCC (Aperio Technologies, Vista, CA, USA).
The investigators found that the expression of a protein called choline phosphate cytidylyltransferase-α CCT-α or CCTα, which acts as an “antigen” that prompts the immune system to produce antibodies against it. CCTα was associated with longer survival rates, including for patients with NSCLC who were treated with surgery alone, without the use of platinum-based chemotherapy drugs and associated toxic side effects.
Laura J. Niedernhofer, MD, PhD, an associate professor and a senior author of the study said, “Based on what we found, a high CCTα expression appears to be indicative of survival, making CCTα a promising biomarker. Our findings suggest that CCTα may, in fact, be more important in determining outcomes in patients with both types of cancer than the already established excision repair cross-complementation group 1 (ERCC1) protein.”
Related Links:
University of Pittsburgh
Aperio Technologies
Latest Pathology News
- Study Reveals Moleclar Mechanism Driving Aggressive Skin Cancer
- AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
- Collaboration Applies AI Pathology to Predict Response to Antibody-Drug Conjugates
- Biomarker Predicts Immunotherapy Response and Prognosis in Colorectal Cancer
- AI Improves Completeness of Complex Cancer Pathology Reports
- AI Tool Predicts Chemotherapy Response in Small Cell Lung Cancer
- Tumor-Specific Biomarker Predicts Neoadjuvant Immunotherapy Response in Gastric Cancer
- AI Tool Predicts Patient-Specific Chemotherapy Benefit in Breast Cancer
- AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
- Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
- New Chromogenic Culture Media Enable Rapid Detection of Candida Infections
- AI-Powered Tool to Transform Dermatopathology Workflow
- AI Tool Predicts Chemotherapy Response from Biopsy Slides
- Sex Differences in Alzheimer’s Biomarkers Linked to Faster Cognitive Decline
- World’s First Optical Microneedle Device to Enable Blood-Sampling-Free Clinical Testing
- Novel mcPCR Technology to Transform Testing of Clinical Samples
Channels
Clinical Chemistry
view channel
Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
Early detection across multiple cancers remains a major unmet need in population screening. Non-invasive approaches that can be delivered at scale may broaden access and shift diagnoses to earlier stages.... Read more
Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Alzheimer's disease often advances silently for years, making timely risk stratification difficult in routine practice. Current approaches to detect pathology can involve lumbar puncture or positron emission... Read moreMolecular Diagnostics
view channel
Machine Learning Model Uses DNA Methylation to Predict Tumor Origin in Cancers of Unknown Primary
Cancers of unknown primary (CUP) are metastatic malignancies in which the primary site cannot be identified, complicating treatment selection. Many patients consequently receive broad, nonspecific chemotherapy... Read more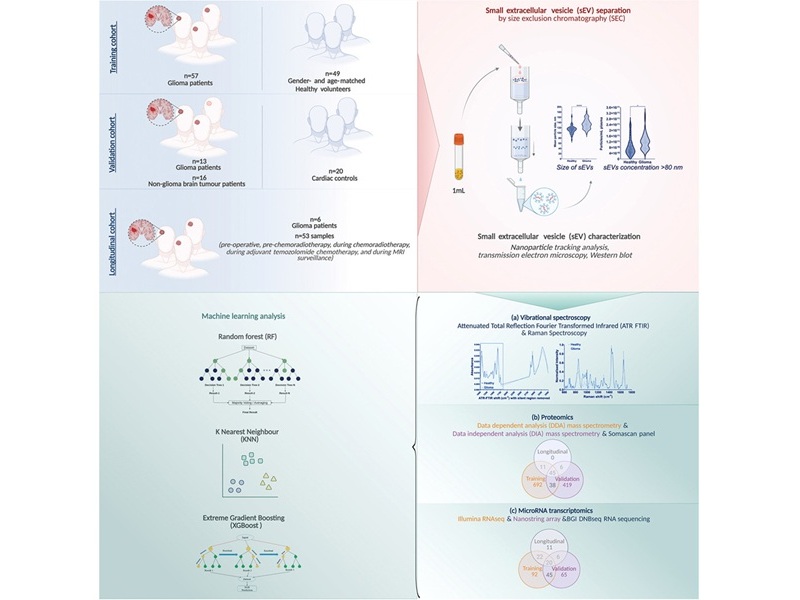
Blood Test Enables Early Detection and Classification of Glioma
High-grade gliomas, particularly glioblastoma, are fast-growing brain tumors that are often diagnosed late and typically require invasive procedures for confirmation. Current pathways rely on symptoms,... Read more
Multi-Biomarker Blood Test Detects Early-Stage Cancers Across Types
Abbott is showcasing its Cancerguard multi-cancer early detection (MCED) test at the American Association for Cancer Research (AACR) Annual Meeting 2026, where new data highlight continued progress in... Read more
New Sample-to-Answer PCR System Supports High-Throughput Infectious Disease Testing
Clinical laboratories face mounting demand for rapid, high‑volume molecular testing for infectious diseases, including routine monitoring in immunocompromised patients. Consolidated, sample‑to‑answer workflows... Read moreHematology
view channel
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read more
Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
Chronic myelomonocytic leukemia (CMML) is a rare blood cancer in which acquired genetic mutations in bone marrow stem cells drive disease. Stem cell transplantation is the only curative option but carries... Read moreMicrobiology
view channel
Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
Gram-negative bloodstream infections and sepsis demand fast, precise antimicrobial therapy, yet conventional susceptibility workflows can delay targeted treatment. Clinical laboratories need platforms... Read more
Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
Antibiotic resistance in early life is challenging to characterize, particularly around the timing and drivers of gene acquisition in newborns. Meconium, the first stool passed by infants, was long considered... Read morePathology
view channel
AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
Reliable prognostic profiling and biomarker screening are essential to guide oncology treatment decisions, while laboratories must balance speed and resource constraints. Earlier identification of high‑risk... Read more
Study Reveals Moleclar Mechanism Driving Aggressive Skin Cancer
Cutaneous squamous cell carcinoma (cSCC) is the world’s second most common skin cancer, and while many cases are treatable, a subset becomes highly aggressive and therapy‑resistant. Identifying molecular... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
QuidelOrtho Corporation has completed the acquisition of LEX Diagnostics for approximately USD 100 million in cash. The transaction adds the LEX VELO System to QuidelOrtho’s portfolio. The platform received U.... Read more
Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
Seegene introduced STAgora, a real-time data analytics platform built on aggregated statistical testing data, at ESCMID Global 2026 in Munich, where it also presented an enhanced model of its automated... Read more
Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
Foundation Medicine, Inc., an independent affiliate of Roche, announced plans to expand its monitoring portfolio with SAGA Diagnostics’ Pathlight, a personalized, tumor-informed molecular residual disease... Read more