Genome Sequencing Identifies Myeloma Precursor Disease with Progression Risk
|
By LabMedica International staff writers Posted on 08 Apr 2021 |

Image: The BD FACSAria III sorter is equipped with five lasers, 17 fluorescence channels and two channels for Forward Scatter and Side Scatter (Photo courtesy of BD Biosciences)
Multiple myeloma (MM) is the second most common hematological malignancy and is consistently preceded by the asymptomatic expansion of clonal plasma cells, termed either monoclonal gammopathy of undetermined significance (MGUS) or smoldering myeloma (SMM).
These two precursor conditions are found in 2%–3% of the general population aged older than 40 years. Only a small fraction of MGUS will ultimately progress to MM, whereas ~60% of persons with SMM will progress within 10 years of initial diagnosis. Currently, the differentiation between MGUS and SMM is based on indirect measures and surrogate markers of disease burden.
Hematologists and Oncologists at the Memorial Sloan Kettering Cancer Center (New York, NY, USA) and their colleagues interrogated genome sequence data for 80 multiple myeloma, 18 MGUS, and 14 SMM cases, including a single SMM case classified as high risk based on an available prognostic model. The team compared genome features in 17 precursor cases that progressed to multiple myeloma within two years and 15 stable precursor cases, uncovering a set of “myeloma-defining genomic events” that included chromothripsis, aneuploidy, driver gene mutations, apolipoprotein B mRNA editing enzyme, catalytic polypeptide-like (APOBEC) mutational profiles, and templated insertions.
For all samples, bone marrow plasma cells (BMPCs) were isolated from bone marrow aspirates and sorted on a BD FACSAria III instrument (BD Biosciences, San Jose, CA, USA). For matched control DNA from each patient, bone marrow T cells or peripheral blood mononuclear cells were used. The T cells were isolated from the BM aspirates and sorted also using the BD FACSAria III. Standard input whole-genome sequencing were run on a NovaSeq 6000 in a 150 bp/150 bp paired-end run (Illumina, San Diego, CA, USA).
The scientists reported that clinically stable cases of MGUS and SMM are characterized by a different genomic landscape and by differences in the temporal acquisition of myeloma-defining genomic events in comparison to progressive entities. In contrast, the investigators reported, the more clinically stable set of precursor gammopathies were missing such alterations. They also tended to surface in individuals diagnosed with MGUS or SMM somewhat later in life (between around 28 and 65 years old), compared to precursor conditions in those with progressive disease, who were diagnosed between the ages of five and 46 years.
The authors concluded that despite its limited sample size, their study provides proof of principle that whole genome sequencing (WGS) has the potential to accurately differentiate stable and progressive precursor conditions in low disease burden clinical states. The application of this technology in the clinic has the potential to significantly alter the management of individual patients, but will require confirmation in larger studies. The study was published on March 25, 2021 in the journal Nature Communications.
Related Links:
Memorial Sloan Kettering Cancer Center
BD Biosciences
Illumina
These two precursor conditions are found in 2%–3% of the general population aged older than 40 years. Only a small fraction of MGUS will ultimately progress to MM, whereas ~60% of persons with SMM will progress within 10 years of initial diagnosis. Currently, the differentiation between MGUS and SMM is based on indirect measures and surrogate markers of disease burden.
Hematologists and Oncologists at the Memorial Sloan Kettering Cancer Center (New York, NY, USA) and their colleagues interrogated genome sequence data for 80 multiple myeloma, 18 MGUS, and 14 SMM cases, including a single SMM case classified as high risk based on an available prognostic model. The team compared genome features in 17 precursor cases that progressed to multiple myeloma within two years and 15 stable precursor cases, uncovering a set of “myeloma-defining genomic events” that included chromothripsis, aneuploidy, driver gene mutations, apolipoprotein B mRNA editing enzyme, catalytic polypeptide-like (APOBEC) mutational profiles, and templated insertions.
For all samples, bone marrow plasma cells (BMPCs) were isolated from bone marrow aspirates and sorted on a BD FACSAria III instrument (BD Biosciences, San Jose, CA, USA). For matched control DNA from each patient, bone marrow T cells or peripheral blood mononuclear cells were used. The T cells were isolated from the BM aspirates and sorted also using the BD FACSAria III. Standard input whole-genome sequencing were run on a NovaSeq 6000 in a 150 bp/150 bp paired-end run (Illumina, San Diego, CA, USA).
The scientists reported that clinically stable cases of MGUS and SMM are characterized by a different genomic landscape and by differences in the temporal acquisition of myeloma-defining genomic events in comparison to progressive entities. In contrast, the investigators reported, the more clinically stable set of precursor gammopathies were missing such alterations. They also tended to surface in individuals diagnosed with MGUS or SMM somewhat later in life (between around 28 and 65 years old), compared to precursor conditions in those with progressive disease, who were diagnosed between the ages of five and 46 years.
The authors concluded that despite its limited sample size, their study provides proof of principle that whole genome sequencing (WGS) has the potential to accurately differentiate stable and progressive precursor conditions in low disease burden clinical states. The application of this technology in the clinic has the potential to significantly alter the management of individual patients, but will require confirmation in larger studies. The study was published on March 25, 2021 in the journal Nature Communications.
Related Links:
Memorial Sloan Kettering Cancer Center
BD Biosciences
Illumina
Latest Hematology News
- Next Generation Instrument Screens for Hemoglobin Disorders in Newborns
- First 4-in-1 Nucleic Acid Test for Arbovirus Screening to Reduce Risk of Transfusion-Transmitted Infections
- POC Finger-Prick Blood Test Determines Risk of Neutropenic Sepsis in Patients Undergoing Chemotherapy
- First Affordable and Rapid Test for Beta Thalassemia Demonstrates 99% Diagnostic Accuracy
- Handheld White Blood Cell Tracker to Enable Rapid Testing For Infections
- Smart Palm-size Optofluidic Hematology Analyzer Enables POCT of Patients’ Blood Cells
- Automated Hematology Platform Offers High Throughput Analytical Performance
- New Tool Analyzes Blood Platelets Faster, Easily and Accurately
- First Rapid-Result Hematology Analyzer Reports Measures of Infection and Severity at POC
- Bleeding Risk Diagnostic Test to Reduce Preventable Complications in Hospitals
- True POC Hematology Analyzer with Direct Capillary Sampling Enhances Ease-of-Use and Testing Throughput
- Point of Care CBC Analyzer with Direct Capillary Sampling Enhances Ease-of-Use and Testing Throughput
- Blood Test Could Predict Outcomes in Emergency Department and Hospital Admissions
- Novel Technology Diagnoses Immunothrombosis Using Breath Gas Analysis
- Advanced Hematology System Allows Labs to Process Up To 119 Complete Blood Count Results per Hour
- Unique AI-Based Approach Automates Clinical Analysis of Blood Data
Channels
Clinical Chemistry
view channel
3D Printed Point-Of-Care Mass Spectrometer Outperforms State-Of-The-Art Models
Mass spectrometry is a precise technique for identifying the chemical components of a sample and has significant potential for monitoring chronic illness health states, such as measuring hormone levels... Read more.jpg)
POC Biomedical Test Spins Water Droplet Using Sound Waves for Cancer Detection
Exosomes, tiny cellular bioparticles carrying a specific set of proteins, lipids, and genetic materials, play a crucial role in cell communication and hold promise for non-invasive diagnostics.... Read more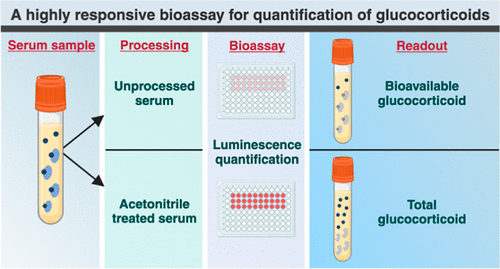
Highly Reliable Cell-Based Assay Enables Accurate Diagnosis of Endocrine Diseases
The conventional methods for measuring free cortisol, the body's stress hormone, from blood or saliva are quite demanding and require sample processing. The most common method, therefore, involves collecting... Read moreMolecular Diagnostics
view channel
Simple Blood Test Could Enable First Quantitative Assessments for Future Cerebrovascular Disease
Cerebral small vessel disease is a common cause of stroke and cognitive decline, particularly in the elderly. Presently, assessing the risk for cerebral vascular diseases involves using a mix of diagnostic... Read more
New Genetic Testing Procedure Combined With Ultrasound Detects High Cardiovascular Risk
A key interest area in cardiovascular research today is the impact of clonal hematopoiesis on cardiovascular diseases. Clonal hematopoiesis results from mutations in hematopoietic stem cells and may lead... Read moreImmunology
view channel
Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
Transplanted organs constantly face the risk of being rejected by the recipient's immune system which differentiates self from non-self using T cells and B cells. T cells are commonly associated with acute... Read more
AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
Current strategies for matching cancer patients with specific treatments often depend on bulk sequencing of tumor DNA and RNA, which provides an average profile from all cells within a tumor sample.... Read more
Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
Cancer treatment typically adheres to a standard of care—established, statistically validated regimens that are effective for the majority of patients. However, the disease’s inherent variability means... Read moreMicrobiology
view channelEnhanced Rapid Syndromic Molecular Diagnostic Solution Detects Broad Range of Infectious Diseases
GenMark Diagnostics (Carlsbad, CA, USA), a member of the Roche Group (Basel, Switzerland), has rebranded its ePlex® system as the cobas eplex system. This rebranding under the globally renowned cobas name... Read more
Clinical Decision Support Software a Game-Changer in Antimicrobial Resistance Battle
Antimicrobial resistance (AMR) is a serious global public health concern that claims millions of lives every year. It primarily results from the inappropriate and excessive use of antibiotics, which reduces... Read more
New CE-Marked Hepatitis Assays to Help Diagnose Infections Earlier
According to the World Health Organization (WHO), an estimated 354 million individuals globally are afflicted with chronic hepatitis B or C. These viruses are the leading causes of liver cirrhosis, liver... Read more
1 Hour, Direct-From-Blood Multiplex PCR Test Identifies 95% of Sepsis-Causing Pathogens
Sepsis contributes to one in every three hospital deaths in the US, and globally, septic shock carries a mortality rate of 30-40%. Diagnosing sepsis early is challenging due to its non-specific symptoms... Read morePathology
view channel.jpg)
Use of DICOM Images for Pathology Diagnostics Marks Significant Step towards Standardization
Digital pathology is rapidly becoming a key aspect of modern healthcare, transforming the practice of pathology as laboratories worldwide adopt this advanced technology. Digital pathology systems allow... Read more
First of Its Kind Universal Tool to Revolutionize Sample Collection for Diagnostic Tests
The COVID pandemic has dramatically reshaped the perception of diagnostics. Post the pandemic, a groundbreaking device that combines sample collection and processing into a single, easy-to-use disposable... Read moreAI-Powered Digital Imaging System to Revolutionize Cancer Diagnosis
The process of biopsy is important for confirming the presence of cancer. In the conventional histopathology technique, tissue is excised, sliced, stained, mounted on slides, and examined under a microscope... Read more
New Mycobacterium Tuberculosis Panel to Support Real-Time Surveillance and Combat Antimicrobial Resistance
Tuberculosis (TB), the leading cause of death from an infectious disease globally, is a contagious bacterial infection that primarily spreads through the coughing of patients with active pulmonary TB.... Read moreTechnology
view channel
New Diagnostic System Achieves PCR Testing Accuracy
While PCR tests are the gold standard of accuracy for virology testing, they come with limitations such as complexity, the need for skilled lab operators, and longer result times. They also require complex... Read more
DNA Biosensor Enables Early Diagnosis of Cervical Cancer
Molybdenum disulfide (MoS2), recognized for its potential to form two-dimensional nanosheets like graphene, is a material that's increasingly catching the eye of the scientific community.... Read more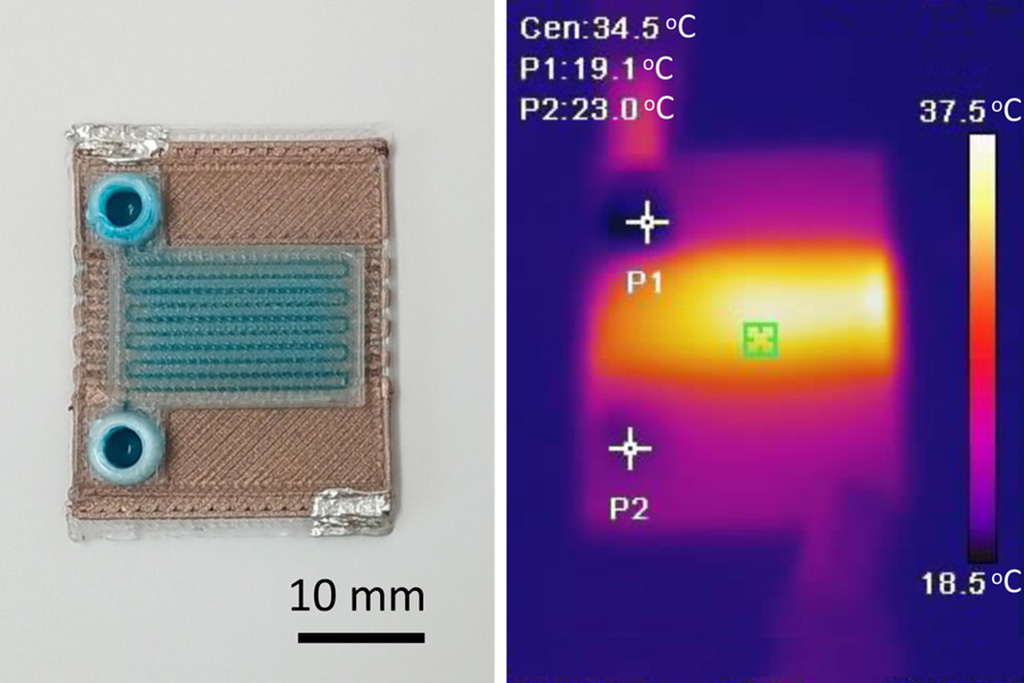
Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
Microfluidics, which are miniature devices that control the flow of liquids and facilitate chemical reactions, play a key role in disease detection from small samples of blood or other fluids.... Read more
Breakthrough in Diagnostic Technology Could Make On-The-Spot Testing Widely Accessible
Home testing gained significant importance during the COVID-19 pandemic, yet the availability of rapid tests is limited, and most of them can only drive one liquid across the strip, leading to continued... Read moreIndustry
view channel_1.jpg)
Thermo Fisher and Bio-Techne Enter Into Strategic Distribution Agreement for Europe
Thermo Fisher Scientific (Waltham, MA USA) has entered into a strategic distribution agreement with Bio-Techne Corporation (Minneapolis, MN, USA), resulting in a significant collaboration between two industry... Read more
ECCMID Congress Name Changes to ESCMID Global
Over the last few years, the European Society of Clinical Microbiology and Infectious Diseases (ESCMID, Basel, Switzerland) has evolved remarkably. The society is now stronger and broader than ever before... Read more