Ultra-Rapid Culture-Free Sepsis Test Reduces Testing Time from Days to Hours
|
By LabMedica International staff writers Posted on 26 Jul 2024 |
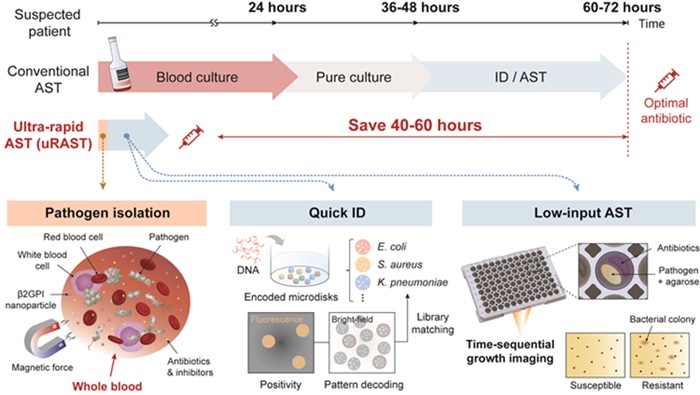
Sepsis, a critical emergency condition, results from an overactive inflammatory response to pathogens like bacteria or fungi in the blood, leading to organ damage and the possibility of sudden death. It holds a 30-day mortality rate of over 30%, which is more than double that of heart attacks. Prompt administration of the correct antibiotic is vital for reducing this high mortality rate. To determine the best treatment, three independent tests are typically required: blood culture to confirm the infection, pathogen identification to pinpoint the specific infecting organism, and antimicrobial susceptibility testing (AST) to identify the most effective antibiotic. Currently, obtaining AST results, which are crucial for selecting the appropriate antibiotic, can take more than 2-3 days. Delays in these results contribute to inappropriate antibiotic use, accelerating the emergence of multidrug-resistant 'superbugs.' While advancements have shortened the timeframe needed for AST, no global progress has been made in reducing the time required for the blood culture process, which is the most time-consuming. Now, an ultra-rapid AST method that bypasses the need for traditional blood culture has demonstrated the potential to reduce the turnaround time of reporting drug susceptibility profiles by more than 40–60 hours compared with hospital AST workflows.
The ultra-Rapid Antimicrobial Susceptibility Testing (uRAST) developed by researchers from the Department of Electrical and Computer Engineering at Seoul National University (Seoul, Korea), in collaboration with QuantaMatrix Inc. (Seoul, Korea), is the world's first to bypass the lengthy blood culture phase, allowing for the completion of all necessary tests for an effective antibiotic regimen within a single day. The uRAST technology employs nanoparticles coated with immune proteins that specifically bind to pathogens, enabling the direct isolation of these pathogens from a patient's blood. The researchers have also integrated new technologies that rapidly conduct pathogen identification and AST, considerably speeding up the testing process. In a clinical trial involving 190 patients suspected of having sepsis, uRAST delivered complete test results within just 13 hours, slashing 40-60 hours off the time required by traditional diagnostic methods. Moreover, uRAST achieved accuracy levels that meet FDA standards.
Another significant aspect of this research published on July 25th in Nature is the integration of fully automated technology that consolidates all necessary sepsis diagnostics into one streamlined process. Traditionally, each test is performed separately and manually, causing delays—particularly outside of normal laboratory operating hours. For instance, if a blood culture is completed after-hours, further testing must wait until the next day, thus missing the critical window for effective sepsis intervention. This research demonstrated the potential for continuous, 24/7 diagnostic operations by automating the entire sequence of necessary tests for sepsis, significantly improving the prospects for timely patient care.
Related Links:
Seoul National University
QuantaMatrix Inc.
Latest Microbiology News
- Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
- Label-Free Microscopy Method Enables Faster, Quantitative Detection of Malaria
- Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
- RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
- Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
- New Bacterial Target Identified for Early Detection of Noma
- Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
- Rapid Urine Test Speeds Antibiotic Selection for UTIs
- WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
Channels
Clinical Chemistry
view channel
Noninvasive Urine Test May Support Earlier Diagnosis of Psychiatric Disorders
Delays in diagnosing serious psychiatric conditions can leave patients without timely support and complicate treatment planning. For bipolar disorder, average time to diagnosis can exceed nine years, and... Read moreAt-Home Blood and Cognitive Tests Support Dementia Risk Stratification
Dementia places a substantial burden on patients and health systems, and identifying individuals at elevated risk remains difficult at scale. In the UK, almost a million people are estimated to be living... Read moreMolecular Diagnostics
view channel
Emerging Biomarkers Advance Early Detection of MASLD and Liver Cancer Risk
Metabolic dysfunction-associated steatotic liver disease (MASLD) affects about 30% of people worldwide and can advance to metabolic dysfunction-associated steatohepatitis (MASH), fibrosis, cirrhosis, and... Read more
Patented Isothermal Amplification Chemistry Advances Decentralized Testing
Molecular diagnostics offer high sensitivity for pathogen detection but typically rely on thermal cycling and specialized instruments, limiting their use outside centralized laboratories.... Read more
Finger-Prick Blood Test Aids Early Tuberculosis Detection and Risk Stratification
Household contacts of people with tuberculosis face an estimated 2% risk of developing disease, yet most are asymptomatic at the time of screening. Early-stage cases are often missed because symptom checks... Read more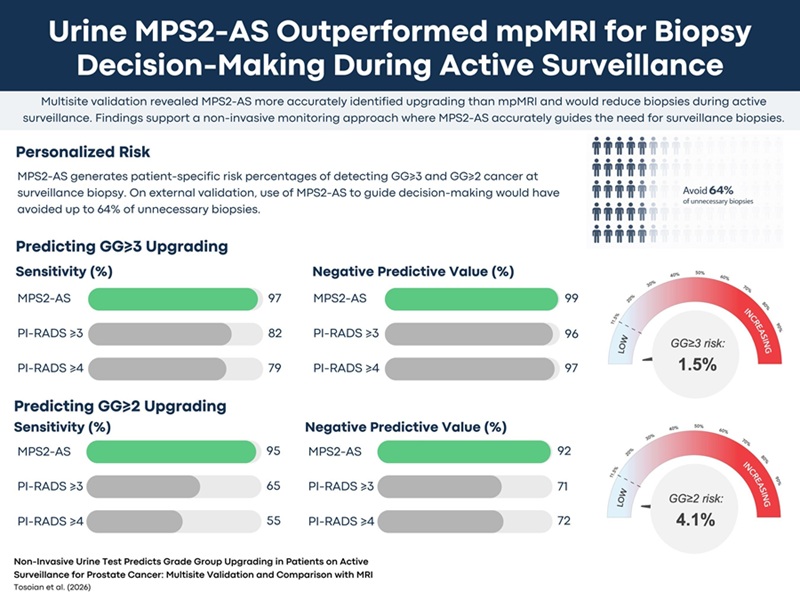
Urine Test Beats MRI in Identifying Prostate Cancer Upgrading During Active Surveillance
Active surveillance is common for men with low-risk prostate cancer, yet deciding when to repeat biopsy remains challenging. Prostate-specific antigen (PSA) testing and magnetic resonance imaging (MRI)... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read more
Point-of-Care Tests Could Expand Access to Mpox Diagnosis
Mpox outbreaks in non-endemic regions have underscored the need for rapid, accessible diagnostics to limit transmission. Polymerase chain reaction (PCR) remains the clinical reference, yet it depends on... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read more
Tumor-on-a-Chip Platform Models Pancreatic Cancer Treatment Response
Pancreatic cancer remains one of the hardest malignancies to treat because tumors are embedded within a dense microenvironment that shapes growth and therapy response. Standard laboratory models often... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more