Mobile Platform Offers Patients Ultrafast Genetic Diagnosis
|
By LabMedica International staff writers Posted on 01 Jan 2024 |

Molecular diagnostics traditionally requires knowledge of handling complex equipment and sample preparation. Now, a simple mobile platform for genetic diagnostics that is currently under development can be handled by healthcare professionals without any laboratory training.
Genomtec SA (Wrocław, Poland) is developing its flagship technological solution named Genomtec ID platform which will offer patients ultrafast mobile genetic diagnosis. The Genomtec ID platform is designed to facilitate diagnostic procedures on-site, including locations like public clinics, consulting rooms, hospitals, pharmacies, and emergency wards, eliminating the necessity for complex, time-consuming lab operations. The technology at the heart of Genomtec ID is the patented Streamlined Nucleic Acid Amplification Technology (SNAAT). This innovation ensures precise pathogen identification within 15 minutes, making it a rapid diagnostic solution. The Genomtec ID system combines the LAMP (loop-mediated isothermal amplification) method with microfluidics and a contactless photonic heating system. This integration allows for simultaneous isolation, purification, concentration, amplification, and detection of specific DNA or RNA fragments of pathogens, significantly speeding up the process while ensuring accuracy on par with or superior to conventional lab-based PCR methods.
One of the standout features of the SNAAT method is its capability for multiplexing, allowing for the simultaneous detection of multiple diagnostic targets on a microfluidic card. This means a single test can identify up to five pathogens at once. The use of a passive microfluidic card, devoid of embedded electronics or electric parts, for nucleic acid isolation, purification, and concentration, not only enhances the detection threshold for target nucleic acids but also reduces the manufacturing costs of disposable reaction cards. The robust diagnostic parameters offered by SNAAT, including sensitivity, specificity, and repeatability, are largely due to its resistance to common inhibitors found in biological samples like blood or drugs. This resistance makes SNAAT an innovative and reliable technology for point-of-care care testing (POCT), promising a new era of convenient and rapid diagnostics accessible to a broader range of healthcare settings.
Related Links:
Genomtec SA
Latest Molecular Diagnostics News
- New AI Tool Improves Detection of Genetic Causes in Rare Disorders
- Adaptive PCR Platform Improves Consistency in Small-Batch NGS Workflows
- Portable Test Uses CRISPR to Rapidly Identify STIs and Resistance Markers
- New Molecular Test Boosts Accuracy of Bile Duct Cancer Diagnosis
- First IVDR‑Certified IGH Clonality Assay Supports Diagnosis of B-Cell Malignancies
- Plasma ctDNA Testing Predicts Breast Cancer Recurrence After Neoadjuvant Therapy
- New Respiratory Panel Expands Pathogen Detection to 25 Targets
- Nasal Swab May Reveal Early Signs of Alzheimer’s Disease
- Blood Biomarker Predicts Cognitive Outcomes After Cardiac Arrest
- Liquid Biopsy Enables Faster Diagnosis of Childhood Cancer in Africa
- Blood Test Helps Guide Treatment in Older Women with Breast Cancer
- Rapid Host-Response Test Distinguishes Bacterial and Viral Infections in Minutes
- Liquid Biopsy Method Pinpoints Disease Source From a Single Drop of Blood
- Study Reveals Widespread Errors in Gene Variant Naming
- New Blood Test Aims to Transform Liver Cancer Surveillance
- New Biomarkers Indicate Higher Liver Cancer Risk in Chronic Hepatitis B Patients
Channels
Clinical Chemistry
view channel
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read more
Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
Alzheimer’s disease develops slowly, and biological changes can appear in blood many years before symptoms. While plasma assays for phosphorylated tau offer earlier detection, discerning whether these... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel
Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
Streptococcus dysgalactiae subspecies equisimilis (SDSE) infections are increasing worldwide and include variants that may lead to severe disease. Researchers now report that whole-genome sequencing of... Read more
Rapid Urine Test Speeds Antibiotic Selection for UTIs
Urinary tract infections are a common reason for antibiotic prescribing and have led to more than 800,000 hospital admissions across England in the past five years, according to National Health Service data.... Read morePathology
view channel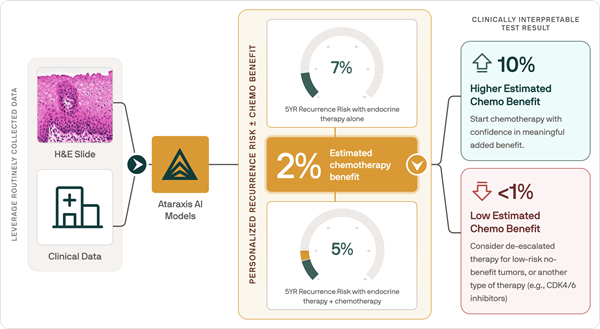
AI Tool Predicts Patient-Specific Chemotherapy Benefit in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer is typically guided by recurrence risk and population-level averages rather than patient-specific benefit. However, existing clinicopathologic... Read more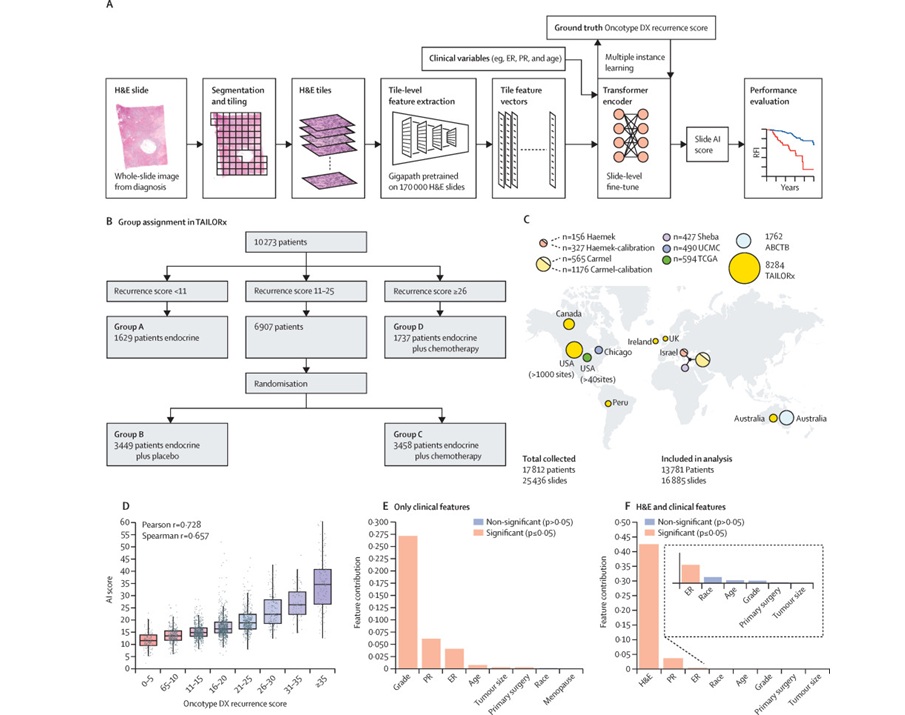
AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer remains a difficult decision because only a subset benefits and many undergo toxicity without gain. Genomic assays can help but are costly,... Read moreTechnology
view channel
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channel
Takara Bio USA and Hamilton Partner Partner to Automate NGS Library Preparation
Takara Bio USA, Inc. (San Jose, CA, USA), a wholly owned subsidiary of Takara Bio Inc., and Hamilton Company (Reno, NV, USA) announced a development and co-marketing agreement to deliver integrated, automated... Read more