Transformative Fingertip Blood Collection Device Produces Lab-Quality Test Results
|
By LabMedica International staff writers Posted on 08 Dec 2023 |
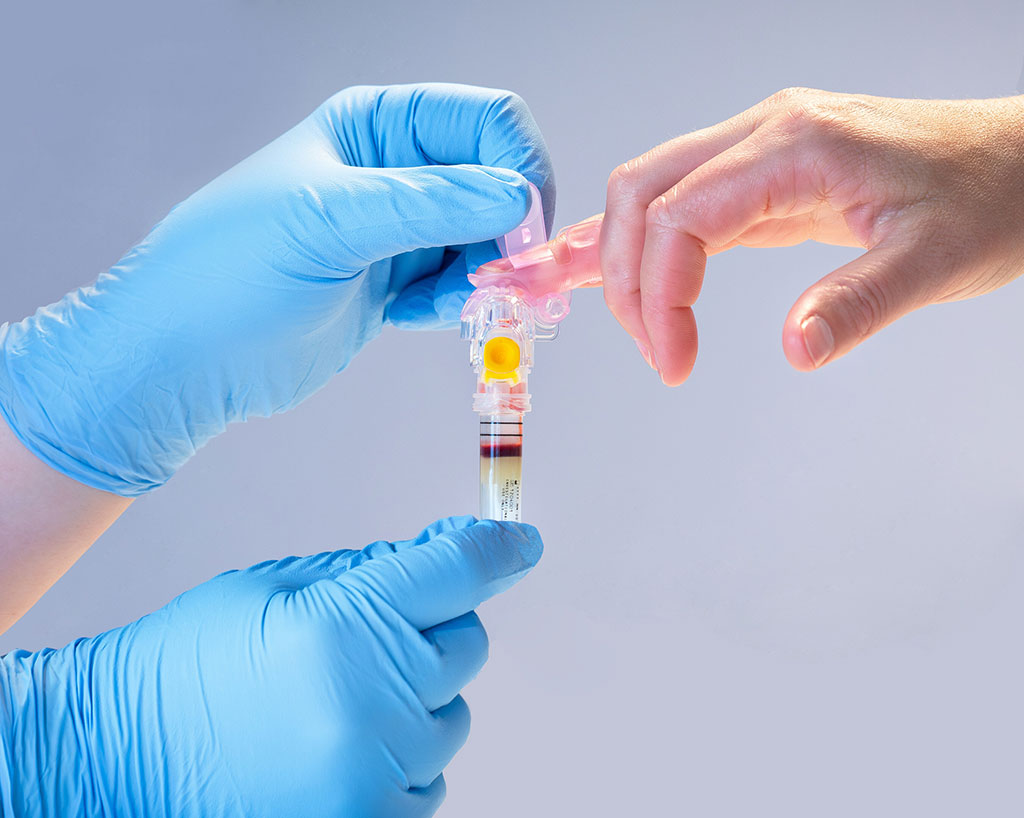
The traditional process of "blood work" for patients typically involves a visit to a laboratory service center where a professional, such as a nurse or phlebotomist, draws blood from the patient's vein. Now, a new blood collection device is set to revolutionize this standard procedure by allowing blood testing in more accessible and convenient locations, offering advantages for patients, healthcare providers, and collection sites.
Becton, Dickinson and Company’s (BD, Franklin Lakes, NJ, USA) BD MiniDraw Capillary Blood Collection System is an innovative device that collects blood samples through a fingerstick, yielding laboratory-quality results for several common blood tests. Unlike the conventional venous blood draw, this system is less invasive, utilizing capillary blood from the patient's finger, which can be done by a trained healthcare worker without the need for a vein puncture by a phlebotomist. The device’s design enables the acquisition of lab-quality results from a smaller blood volume than what is typically required for venous collections. Moreover, it offers more convenience to patients by allowing blood collection at new venues, such as retail pharmacies, potentially enhancing the overall patient experience and revolutionizing diagnostic testing.
The use of the BD MiniDraw Collection System benefits patients by enabling them to have blood drawn in familiar places they frequently visit, like retail pharmacies or grocery stores. These locations often have extended operating hours and may be more conveniently located near their homes or workplaces. For these alternative collection sites, such as retail settings, the system not only complements existing health and wellness programs, like vaccine administration but also has the potential to increase foot traffic. Healthcare providers can benefit too, as capillary blood collection could be offered right in a physician's office without needing specialized phlebotomy staff. This ease and reduced invasiveness of the process might lead to improved patient compliance with blood testing, which is crucial for regular health assessments and could aid in the early detection and better management of chronic diseases.
BD has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the BD MiniDraw Collection System. This clearance covers low-volume blood collection for various tests, including lipid panels, select chemistry tests, and hemoglobin and hematocrit (H&H) testing, which are some of the most frequently requested laboratory tests. They are essential for diagnosing and monitoring various chronic conditions, from hypertension to high cholesterol. Looking ahead, BD aims to broaden the range of blood tests facilitated by the BD MiniDraw Collection System.
"Because the BD MiniDraw Collection System enables blood collection at non-traditional sites that may be more convenient—like your local pharmacy rather than a standalone lab—we can expand health equity and access, and make it easier for patients to get the blood tests they need both for preventative care and the management of chronic conditions," said Dave Hickey, executive vice president and president of Life Sciences for BD. "This innovation is our strategy and pipeline coming to life, and proof of our commitment to enabling diagnostics and care in alternate settings."
Related Links:
BD
Latest Pathology News
- AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
- Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
- AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
- FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
- New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
- AI System Analyzes Routine Pathology Slides to Predict Cancer Outcomes
- New Tissue Mapping Approach Identifies High-Risk Form of Diabetic Kidney Disease
- Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
- Interpretable AI Reveals Hidden Cellular Features from Microscopy Images
- Tumor Immune Structure Predicts Response to Immunotherapy in Melanoma
- Plug-and-Play AI Pathology System Classifies Multiple Cancers from Few Slides
- AI-Based Assays Support Risk Stratification in Prostate and Breast Cancer
- AI Pathology Model Predicts Immunotherapy Response in Lung Cancer
- Study Reveals Moleclar Mechanism Driving Aggressive Skin Cancer
- AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
- Collaboration Applies AI Pathology to Predict Response to Antibody-Drug Conjugates
Channels
Clinical Chemistry
view channel
Liquid Biopsy Biomarkers May Improve Childhood Epilepsy Diagnosis
Childhood epilepsy remains a major neurological disorder with unmet needs for accurate, non-invasive biomarkers, as conventional tests such as electroencephalography and neuroimaging can have limited sensitivity... Read more
Blood-Based Sensor Detects Early Signs of Alzheimer’s and Parkinson’s
Alzheimer’s disease and Parkinson’s disease are increasing as populations age, yet diagnosis remains largely symptom-driven and often occurs after irreversible brain damage has begun. Earlier detection,... Read moreMolecular Diagnostics
view channel
Ultrasensitive MRD Blood Test Detects Early Breast Cancer Recurrence
SAGA Diagnostics (Morrisville, NC, USA), a company specializing in tumor-informed, blood-based cancer detection and precision medicine, announced the publication of a new study evaluating its Pathlight... Read more
Position Statements Outline Evidence Standards for Multi-Cancer Detection Tests
Cancer screening is intended to reduce mortality, but policy decisions often depend on early indicators that may not fully reflect true survival benefit. The emergence of blood-based tests capable of detecting... Read moreHematology
view channel
Higher Ferritin Threshold May Improve Iron Deficiency Detection in Children
Iron deficiency in school-age children can affect brain development, learning, growth, and physical performance, yet early deficiency may be missed when screening focuses mainly on anemia.... Read more
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read moreImmunology
view channel
Immune Enzyme Linked to Treatment-Resistant Inflammatory Bowel Disease
Inflammatory bowel disease (IBD) affects nearly 3 million people in the United States and its prevalence continues to rise. Medications that target tumor necrosis factor (TNF)-alpha are widely used, but... Read more
Simple Blood Test Could Replace Biopsies for Lung Transplant Rejection Monitoring
Lung transplant recipients face some of the highest rates of acute cellular rejection, and routine surveillance often relies on repeated surgical biopsies. These procedures can cause complications such... Read moreMicrobiology
view channel
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read more
New AMR Assay Supports Rapid Infection Control Screening in Hospitals
As antimicrobial resistance spreads worldwide, healthcare-associated infections are placing a growing burden on hospitals, increasing the need for faster and broader diagnostic solutions.... Read moreTechnology
view channel
AI-Enabled Assistant Unifies Molecular Workflow Planning and Support
Clinical laboratories and research groups face increasingly complex molecular workflows and expanding technical documentation spread across multiple systems. Fragmented digital tools can slow experiment... Read more
AI Tool Automates Validation of Laboratory Software Configuration Changes
Regulated laboratories face heavy documentation and requalification demands when software configurations change, slowing improvements and discouraging beneficial updates. A new capability now automates... Read moreIndustry
view channel
New Distribution Agreement Expands Access to CE-Marked Precision Oncology Assays
Eurobio Scientific (Les Ulis, France) has signed a distribution agreement with Canhelp Genomics (Hangzhou, China) to broaden availability of the Canhelp‑UCa and Canhelp‑Origin assays. The agreement extends... Read more