Prenatal Test Reduces Time of Detecting Chromosomal Abnormalities
|
By LabMedica International staff writers Posted on 24 Aug 2022 |
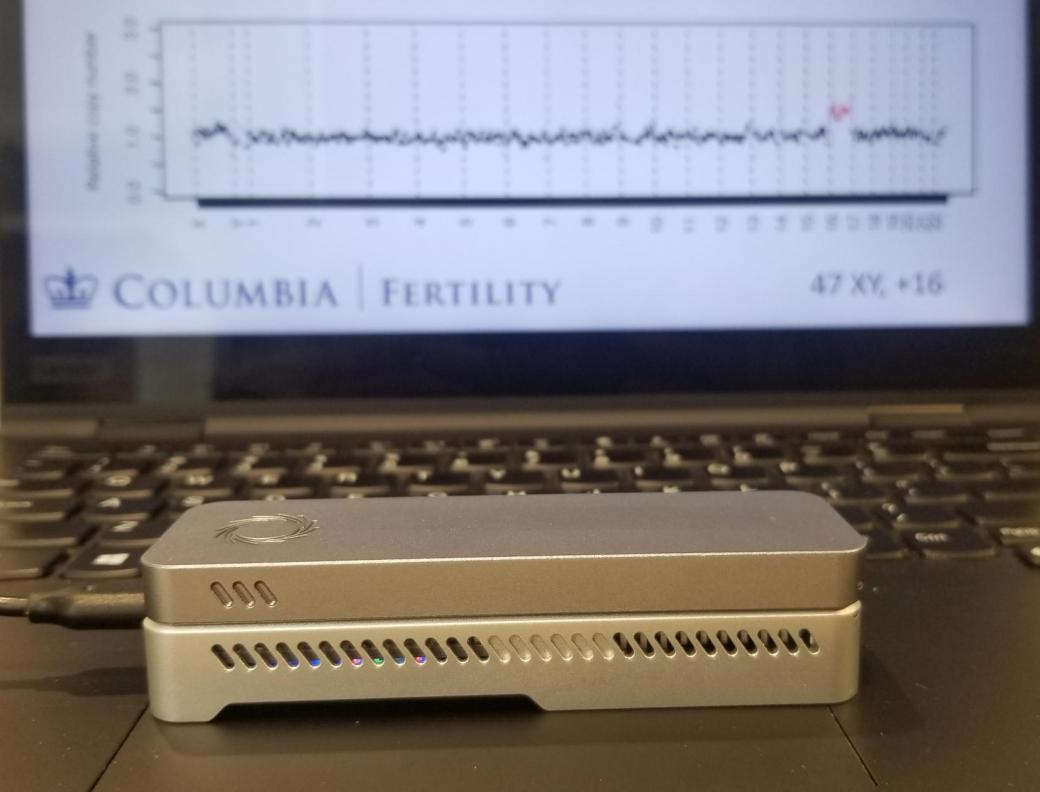
A newly developed prenatal test can determine if a fetus or embryo has the right number of chromosomes at a fraction of the time and cost of currently available clinical genetic tests.
Currently available prenatal genetic tests cost thousands of dollars and take days to weeks to deliver results, adding to the emotional and financial stress of fertility treatment and pregnancy and impacting treatment options.
Clinical Scientists from the Columbia University Irving Medical Center (New York, NY, USA) and their colleagues developed the new test, called STORK (Short-read Transpore Rapid Karyotyping), that can be used in the doctor's office at the point of care, delivers results in under two hours, and is about 10 times less expensive to process per sample than current tests. The nanopore-based sequencing technology was used to analyze tiny fragments of DNA 15,000 times faster than currently available chromosomal testing methods, significantly reducing the amount of time to get results. The test also uses much smaller equipment, about the size of a harmonica and weighing just 450 grams, making it accessible for use in physician offices.
The chromosomal abnormalities that this test can detect are, by far, the most common causes of miscarriage, structural anomalies, and developmental delays. Prenatal genetic testing is recommended for pregnant women who are age 35 or older, have a family history of genetic disorders, or have had one or more miscarriages. It is also used increasingly during in vitro fertilization (IVF) to test embryos prior to implantation to improve the chances of pregnancy and reduce the risk of miscarriage.
The team tested STORK in 218 blinded samples from miscarriages, pregnancies, via amniotic fluid or chorionic villi, and biopsies from embryos prior to implantation. Results were compared with those obtained using standard clinical testing. STORK results agreed with standard clinical testing in all of the pregnancy-related samples and in 98% of the embryos tested. For miscarriage samples, STORK was more accurate than standard testing and was determined to have correctly identified chromosome numbers in the 10 cases for which the two tests disagreed. An additional 60 pregnancy samples were tested with STORK at an independent certified laboratory, and those results were identical to results obtained with standard clinical testing.
Zev Williams, MD, PhD, Associate Professor of Women's Health and a senior author of the study, said, “The affordability of this test also means that individuals who have suffered a miscarriage do not have to wait until a second or third loss before insurance will cover expensive lab tests, leaving many women in the dark and often blaming themselves. Our study also shows that our rapid test was better than the gold standard for testing miscarriage samples, giving women who have suffered a pregnancy loss a sense of closure and the ability to take steps to prevent another loss.” The study was published on August 18, 2022 in the New England Journal of Medicine.
Latest Molecular Diagnostics News
- Plasma Protein Signature Predicts Lung Cancer Risk Up to Five Years Ahead
- Circulating Tumor DNA Testing Guides Chemotherapy, Reduces Relapse in Colon Cancer
- Researchers Uncover Distinct Chromosome Signature in Aggresive ALT Cancers
- Simple Cytogenetic Method Could Improve Classification of ALL Subtypes
- Blood-Based Assay Enables Noninvasive Monitoring of Sarcoma Immunotherapy Response
- Genomic Test Guides Chemotherapy Decisions in Early-Stage Breast Cancer
- Tumor Mutation Marker Helps Refine Lung Cancer Prognosis and Guide Therapy Selection
- Multi-Cancer Test Boosts Detection When Added to Standard Screening
- Blood-Based MRD Monitoring Supports Relapse Prevention in Leukemia
- Genomic Test Predicts Chemotherapy Benefit in Metastatic Prostate Cancer
- Blood Protein Markers Flag Multiple Sclerosis Risk Years Before Diagnosis
- Digital PCR Assays Support Surveillance of Bundibugyo Ebolavirus Outbreak
- Updated Guidance Prioritizes Stool-Based Colorectal Cancer Screening Tests
- Blood-Based Proteomic Test May Predict Treatment Response in Non-Small Cell Lung Cancer
- Position Statements Outline Evidence Standards for Multi-Cancer Detection Tests
- Ultrasensitive MRD Blood Test Detects Early Breast Cancer Recurrence
Channels
Molecular Diagnostics
view channel
Plasma Protein Signature Predicts Lung Cancer Risk Up to Five Years Ahead
Lung cancer remains a leading cause of cancer death, and many cases are detected only after symptoms appear. Current screening programs largely target people with a history of smoking, leaving other at-risk... Read more
Circulating Tumor DNA Testing Guides Chemotherapy, Reduces Relapse in Colon Cancer
Adjuvant therapy decisions after curative surgery for colon cancer remain difficult, as conventional clinicopathologic factors often fail to capture residual disease risk. Liquid biopsy approaches that... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channelAptamer-Based Biosensor Enables Mutation-Resilient SARS-CoV-2 Detection
Rapid evolution of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can undermine existing molecular diagnostics, especially when assays target small viral components. Double-antibody sandwich... Read more
Study Points to Autoimmune Pathway Behind Long COVID Symptoms
Long COVID leaves many SARS-CoV-2 survivors with persistent fatigue, cognitive issues, palpitations, and musculoskeletal pain for months or years. Estimates cited in new research suggest 4%–20% of infected... Read more
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read morePathology
view channel
Blood-Based Method Tracks Gene Activity in the Living Brain
Real-time measurement of gene activity in the brain has been limited by assays requiring destructive tissue sampling. Tracking active genes could reveal how the body responds to environmental factors,... Read more
FDA Approval Expands Automated PD-L1 Testing Across Solid Tumors
Clinical laboratories play a central role in guiding immunotherapy by reporting programmed death ligand-1 (PD‑L1) status across multiple solid tumors. Many sites are standardizing this work on fully automated... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channel























