Qiagen Launches Novel Tuberculosis Blood Test That Marks Quantum Leap from Traditional TB Skin Test
|
By LabMedica International staff writers Posted on 21 Oct 2021 |

QIAGEN N.V. (Venlo, Netherlands) has announced the launch and CE marking of QIAreach QuantiFERON-TB test for tuberculosis (TB) infection, in order to help achieve global TB elimination targets by increasing access to easy and reliable TB testing, especially in high burden countries, low-resource regions.
Built on the proven QuantiFERON-TB Gold Plus technology, QIAreach QuantiFERON-TB is a portable device that enables the ultrasensitive digital detection of TB infection with an end-to-end workflow that is simple and cost efficient, and increases access to reliable Interferon Gamma Release Assay (IGRA) testing. A quantum leap from the traditional TB skin test, QuantiFERON-TB uses blood samples to test for interferon-gamma molecules released from T-cells that have come into contact with TB bacteria. QIAreach QuantiFERON-TB runs these tests on the digital eHub and eStick system, a true walkaway solution with random access and no calibration or maintenance.
QIAreach QuantiFERON TB testing requires one patient visit and produces an easy-to-read result in 20 minutes after sample incubation, with the ability to record and send results for improved data management, making it ideal for TB screening programs. QIAGEN will focus on regions that face a high burden of the disease, and where getting access to lab infrastructure and resources for testing are limited. QIAGEN will work with commercial distribution partners as well as the Stop TB Partnership’s Global Drug Facility to ensure affordable access across all high burden, low and middle-income countries. In parallel, QIAGEN will continue its close collaboration with a variety of international organizations and public health institutions to help drive scale-up through innovative implementation projects.
“With QIAreach QuantiFERON-TB we have taken a great step towards leaving no one behind when facing a deadly disease like tuberculosis that is preventable and curable. With this groundbreaking new way to detect TB infections we can aid to prevent patients, especially those in high burden countries, from developing the active form of TB,” said Thierry Bernard, CEO of QIAGEN. “It combines QIAGEN’s proven QuantiFERON-TB technology with a very portable and field-friendly digital device to deliver extremely reliable results without the need for complex laboratory infrastructure at effective cost.”
Related Links:
QIAGEN N.V.
Latest Microbiology News
- Rapid Urine Test Speeds Antibiotic Selection for UTIs
- WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
- Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
- WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
- New Imaging Approach Could Help Predict Dangerous Gut Infection
- Rapid Sequencing Could Transform Tuberculosis Care
- Blood-Based Viral Signature Identified in Crohn’s Disease
- Hidden Gut Viruses Linked to Colorectal Cancer Risk
- Three-Test Panel Launched for Detection of Liver Fluke Infections
- Rapid Test Promises Faster Answers for Drug-Resistant Infections
- CRISPR-Based Technology Neutralizes Antibiotic-Resistant Bacteria
- Comprehensive Review Identifies Gut Microbiome Signatures Associated With Alzheimer’s Disease
- AI-Powered Platform Enables Rapid Detection of Drug-Resistant C. Auris Pathogens
- New Test Measures How Effectively Antibiotics Kill Bacteria
- New Antimicrobial Stewardship Standards for TB Care to Optimize Diagnostics
Channels
Clinical Chemistry
view channel
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read more
Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
Alzheimer’s disease develops slowly, and biological changes can appear in blood many years before symptoms. While plasma assays for phosphorylated tau offer earlier detection, discerning whether these... Read moreMolecular Diagnostics
view channel
New Molecular Test Boosts Accuracy of Bile Duct Cancer Diagnosis
Bile duct strictures can arise from cancer or benign disease, but their location within ducts connecting the liver, gallbladder, and intestines complicates evaluation. Standard biopsy and cytology may... Read moreAdaptive PCR Platform Improves Consistency in Small-Batch NGS Workflows
PCR amplification during next-generation sequencing (NGS) library preparation can introduce variability, often requiring manual quantification and risking over-cycling artifacts. The issue is especially... Read more
First IVDR‑Certified IGH Clonality Assay Supports Diagnosis of B-Cell Malignancies
Accurate identification of clonal immunoglobulin heavy chain (IGH) gene rearrangements is central to evaluating suspected B-cell lymphoproliferative disorders, where a single B-cell clone yields a defining... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel
Rapid Urine Test Speeds Antibiotic Selection for UTIs
Urinary tract infections are a common reason for antibiotic prescribing and have led to more than 800,000 hospital admissions across England in the past five years, according to National Health Service data.... Read more
WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
Tuberculosis (TB) remains a leading infectious killer, with more than 3,300 deaths and 29,000 new illnesses every day. Diagnostic delays and dependence on centralized laboratory networks continue to impede... Read morePathology
view channel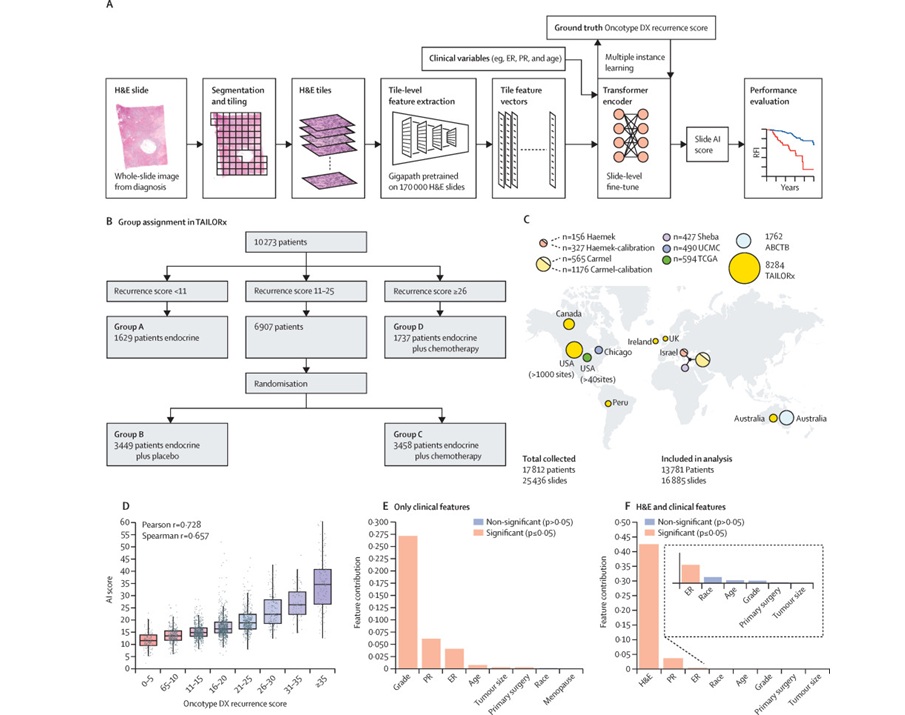
AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer remains a difficult decision because only a subset benefits and many undergo toxicity without gain. Genomic assays can help but are costly,... Read more
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read moreIndustry
view channel
Lunit and CellCarta Collaborate to Expand AI Pathology in CDx Development
Lunit (Seoul, South Korea), a leading provider of AI for cancer diagnostics and precision oncology, and CellCarta (Montreal, QC, Canada), a global contract research organization (CRO) laboratory serving... Read more




.jpg)