Cytokine Target Identified for Treatment-Resistant Hemophilia A
|
By LabMedica International staff writers Posted on 04 May 2021 |
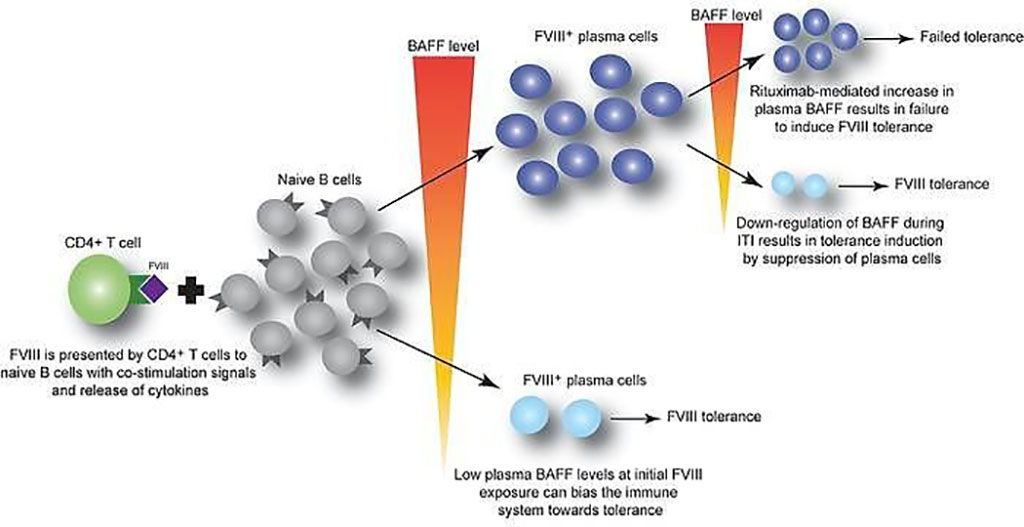
Image: B cell–activating factor modulates the factor VIII immune response in hemophilia A (Photo courtesy of Children’s Hospital of Philadelphia)
Hemophilia A, also called factor VIII (8) deficiency or classic hemophilia, is a genetic disorder caused by missing or defective factor VIII (FVIII), a clotting protein. Although it is passed down from parents to children, about 1/3 of cases found have no previous family history.
Currently, hemophilia is the most common inherited bleeding disorder, which affects 1 in 10,000 men across the globe. Since patients lack coagulation factor VIII (FVIII), therapies are typically aimed at infusions to replace the missing protein. Some patients, however, become treatment resistant due to the development of neutralizing antibodies, or FVIII inhibitors.
An international team of hematologists and their associates led by those at the Children’s Hospital of Philadelphia (Philadelphia, PA, USA) used plasma samples from 69 pediatric patients undergoing recombinant FVIII therapy, in addition to samples from mouse models, to determine whether cytokine B cell activating factor (BAFF) may help generate and sustain FVIII antibodies. Of the pediatric patients, 24 had FVIII inhibitors.
BAFF, proliferation-inducing ligand (APRIL), and B cell maturation antigen (BCMA) levels from the patient samples were measured by a multiplex ELISA Because of limitations on sample transport, BAFF levels from rituximab-exposed patients were measured by ELISA (R&D Systems, Minneapolis, MN, USA). The frequency of FVIII-specific immunoglobulin-secreting B cells was quantified by a B cell ELISPOT assay. Plates were analyzed using the ImmunoSpot system (Cellular Technology Limited, Shaker Heights, OH, USA).
The team reported that BAFF levels to be higher in patients with persistent FVIII inhibitors compared to those without inhibitors (1.30 versus 0.99 ng/mL, respectively). Moreover, BAFF levels decreased from baseline in hemophilia A inhibitor patients who underwent immune tolerance induction (ITI) and achieved FVIII tolerance from 1.43 ± 0.63 to 0.81 ± 0.32 ng/mL. In comparison, those who failed ITI had steady levels at 1.33 to 1.23 ng/mL. A similar pattern and trend in BAFF levels was also noted for 46 adults, although non-inhibitor patients saw a generally higher level of BAFF. Furthermore, a receiver-operating characteristic (ROC) analysis of the pooled pediatric and adult data showed that BAFF levels greater than 1.03 ng/mL had 68.3% sensitivity, 63.8% specificity, and likelihood ratio of 1.89 for the presence of FVIII inhibitors.
Valder R. Arruda, MD, PhD, a Hematologist and senior author of the study, said, “Our data suggest that BAFF may regulate the generation and maintenance of FVIII inhibitors, as well as anti-FVIII B cells. Given that an FDA-approved anti-BAFF antibody is currently used to suppress the immune response in autoimmune diseases, future studies should explore the use of this treatment in combination with rituximab to achieve better outcomes for hemophilia A patients resistant to FVIII protein replacement therapy.”
The authors concluded that their data suggest that BAFF may regulate the generation and maintenance of FVIII inhibitors and/or anti-FVIII B cells. Finally, antiCD20/anti-BAFF combination therapy may be clinically useful for ITI. The study was published on April 15, 2021 in The Journal of Clinical Investigation.
Related Links:
Children’s Hospital of Philadelphia
R&D Systems
Cellular Technology Limited
Currently, hemophilia is the most common inherited bleeding disorder, which affects 1 in 10,000 men across the globe. Since patients lack coagulation factor VIII (FVIII), therapies are typically aimed at infusions to replace the missing protein. Some patients, however, become treatment resistant due to the development of neutralizing antibodies, or FVIII inhibitors.
An international team of hematologists and their associates led by those at the Children’s Hospital of Philadelphia (Philadelphia, PA, USA) used plasma samples from 69 pediatric patients undergoing recombinant FVIII therapy, in addition to samples from mouse models, to determine whether cytokine B cell activating factor (BAFF) may help generate and sustain FVIII antibodies. Of the pediatric patients, 24 had FVIII inhibitors.
BAFF, proliferation-inducing ligand (APRIL), and B cell maturation antigen (BCMA) levels from the patient samples were measured by a multiplex ELISA Because of limitations on sample transport, BAFF levels from rituximab-exposed patients were measured by ELISA (R&D Systems, Minneapolis, MN, USA). The frequency of FVIII-specific immunoglobulin-secreting B cells was quantified by a B cell ELISPOT assay. Plates were analyzed using the ImmunoSpot system (Cellular Technology Limited, Shaker Heights, OH, USA).
The team reported that BAFF levels to be higher in patients with persistent FVIII inhibitors compared to those without inhibitors (1.30 versus 0.99 ng/mL, respectively). Moreover, BAFF levels decreased from baseline in hemophilia A inhibitor patients who underwent immune tolerance induction (ITI) and achieved FVIII tolerance from 1.43 ± 0.63 to 0.81 ± 0.32 ng/mL. In comparison, those who failed ITI had steady levels at 1.33 to 1.23 ng/mL. A similar pattern and trend in BAFF levels was also noted for 46 adults, although non-inhibitor patients saw a generally higher level of BAFF. Furthermore, a receiver-operating characteristic (ROC) analysis of the pooled pediatric and adult data showed that BAFF levels greater than 1.03 ng/mL had 68.3% sensitivity, 63.8% specificity, and likelihood ratio of 1.89 for the presence of FVIII inhibitors.
Valder R. Arruda, MD, PhD, a Hematologist and senior author of the study, said, “Our data suggest that BAFF may regulate the generation and maintenance of FVIII inhibitors, as well as anti-FVIII B cells. Given that an FDA-approved anti-BAFF antibody is currently used to suppress the immune response in autoimmune diseases, future studies should explore the use of this treatment in combination with rituximab to achieve better outcomes for hemophilia A patients resistant to FVIII protein replacement therapy.”
The authors concluded that their data suggest that BAFF may regulate the generation and maintenance of FVIII inhibitors and/or anti-FVIII B cells. Finally, antiCD20/anti-BAFF combination therapy may be clinically useful for ITI. The study was published on April 15, 2021 in The Journal of Clinical Investigation.
Related Links:
Children’s Hospital of Philadelphia
R&D Systems
Cellular Technology Limited
Latest Hematology News
- Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
- Advanced CBC-Derived Indices Integrated into Hematology Platforms
- Blood Test Enables Early Detection of Multiple Myeloma Relapse
- Single Assay Enables Rapid HLA and ABO Genotyping for Transplant Matching
- Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
- Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
- Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
- New Platelet Function Assay Enables Monitoring of Antiplatelet Therapy
- Open Multi-Omics Platform Identifies Prognostic Subtypes in Blood Cancers
- AI-Powered Digital Workflow Standardizes Bone Marrow Aspirate Morphology
- Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
- New Guidelines Aim to Improve AL Amyloidosis Diagnosis
- Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
- Fast and Easy Test Could Revolutionize Blood Transfusions
- High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
- AI Algorithm Effectively Distinguishes Alpha Thalassemia Subtypes
Channels
Clinical Chemistry
view channel
FDA-Cleared Assay Enables Comprehensive Automated Testosterone Testing
Accurate evaluation of androgen status often requires concordant measurement of total testosterone, free testosterone, and sex hormone‑binding globulin. Reference methods such as equilibrium dialysis with... Read more
CE-Marked Blood Biomarker Test Advances Automated Alzheimer’s Diagnostics
Specialized care settings frequently evaluate patients aged 50 years and older who present with signs and symptoms of cognitive decline to determine whether amyloid pathology linked to Alzheimer’s disease... Read moreMolecular Diagnostics
view channel
Blood-Based “Ageing Clock” Helps Predict Dementia Risk and Earlier Onset
Dementia imposes a growing health burden, affecting an estimated 982,000 people in the UK, with cases projected to reach 1.4 million by 2040. Earlier identification of those most likely to develop disease... Read more
Blood Test Refines Biopsy Decisions in Prostate Cancer
Prostate biopsy decisions remain challenging, with many men undergoing invasive procedures that ultimately yield negative results. In the U.S., more than a million prostate biopsies are performed each... Read moreImmunology
view channel
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read more
Point-of-Care Tests Could Expand Access to Mpox Diagnosis
Mpox outbreaks in non-endemic regions have underscored the need for rapid, accessible diagnostics to limit transmission. Polymerase chain reaction (PCR) remains the clinical reference, yet it depends on... Read moreMicrobiology
view channel
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read more
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read more
Tumor-on-a-Chip Platform Models Pancreatic Cancer Treatment Response
Pancreatic cancer remains one of the hardest malignancies to treat because tumors are embedded within a dense microenvironment that shapes growth and therapy response. Standard laboratory models often... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more




.jpg)