New Monotest Diagnoses Risk of Invasive Aspergillosis
|
By LabMedica International staff writers Posted on 12 Nov 2020 |

Image: ASPERGILLUS GALACTOMANNAN Ag VIRCLIA® MONOTEST (Photo courtesy of Vircell S.L.)
A new monotest using chemiluminescence, meets the need for the immediate testing of individual samples for patients at risk of invasive aspergillosis, without resorting to batching, cumulating samples, or sending out to an external laboratory.
Aspergillus sp. is one of the most ubiquitous of the airborne saprophytic fungi. With the increase in the number of immune-suppressed patients, there has been a dramatic increase in severe and usually fatal invasive aspergillosis or invasive candidiasis, now one of the most common mold infections worldwide. Invasive Aspergillus Infection (IAI) is an important life-threatening infection in immune-compromised patients, especially those with prolonged neutropenia, allogeneic hematopoietic stem cell transplant (HSCT) and solid organ transplant (SOT). In immune-competent hosts, IAI is involved in chronic obstructive pulmonary, Large Burned patients, cirrhosis, corticosteroid treatment, most of them located in Intensive Care Units.
Diagnosis of IAI by conventional culture-based methods show clear deficiencies especially in sensitivity and speed: Confirmation with culture occurs in less than 50% of cases due to the frequent negative results of blood cultures and the difficulty of obtaining deep tissue samples or fluids ordinarily sterile. The availability of effective antifungal agents determines the need for rapid and accurate diagnosis. Therefore, it is essential to implement the diagnosis of invasive mycosis with non-culture-based techniques among which biomarkers are already an essential part.
Currently, the most commonly biomarker used in the case of Aspergillus is the Galactomannan. Galactomannan is a useful diagnostic test for the investigation of invasive aspergillosis in patients with a suggestive clinic. It can be done not only in blood but also in CSF, BAL or peritoneal fluid. Its systematic and periodic use is not recommended in non-symptomatic hematological patients who have been receiving antifungal prophylaxis. However, the format currently marketed by a microtiter plate capture ELISA makes it very difficult to use the test with only one or a few samples. The ELISA method is useful when studying a large number of patients at risk of invasive aspergillosis in a systematic way. New formats are now needed to facilitate its use with a single sample. These new systems must be able to quantify the fungal load and be easy to carry out. One model would be the new immunochromatography based on “Lateral Flow”, which is a very simple, but difficult to quantify technique.
Now, the VirClia Aspergillus Galactomannan AG, a new monotest using chemiluminescence developed by Vircell S.L. (Granada, Spain), could respond to these new diagnostic needs for testing individual samples immediately, without the need for batching, cumulating samples or sending out to an external laboratory. The VirClia Aspergillus Galactomannan AG has been successfully evaluated internally and externally against a commercially available ELISA reference assay and showed a 91% correlation.
Aspergillus sp. is one of the most ubiquitous of the airborne saprophytic fungi. With the increase in the number of immune-suppressed patients, there has been a dramatic increase in severe and usually fatal invasive aspergillosis or invasive candidiasis, now one of the most common mold infections worldwide. Invasive Aspergillus Infection (IAI) is an important life-threatening infection in immune-compromised patients, especially those with prolonged neutropenia, allogeneic hematopoietic stem cell transplant (HSCT) and solid organ transplant (SOT). In immune-competent hosts, IAI is involved in chronic obstructive pulmonary, Large Burned patients, cirrhosis, corticosteroid treatment, most of them located in Intensive Care Units.
Diagnosis of IAI by conventional culture-based methods show clear deficiencies especially in sensitivity and speed: Confirmation with culture occurs in less than 50% of cases due to the frequent negative results of blood cultures and the difficulty of obtaining deep tissue samples or fluids ordinarily sterile. The availability of effective antifungal agents determines the need for rapid and accurate diagnosis. Therefore, it is essential to implement the diagnosis of invasive mycosis with non-culture-based techniques among which biomarkers are already an essential part.
Currently, the most commonly biomarker used in the case of Aspergillus is the Galactomannan. Galactomannan is a useful diagnostic test for the investigation of invasive aspergillosis in patients with a suggestive clinic. It can be done not only in blood but also in CSF, BAL or peritoneal fluid. Its systematic and periodic use is not recommended in non-symptomatic hematological patients who have been receiving antifungal prophylaxis. However, the format currently marketed by a microtiter plate capture ELISA makes it very difficult to use the test with only one or a few samples. The ELISA method is useful when studying a large number of patients at risk of invasive aspergillosis in a systematic way. New formats are now needed to facilitate its use with a single sample. These new systems must be able to quantify the fungal load and be easy to carry out. One model would be the new immunochromatography based on “Lateral Flow”, which is a very simple, but difficult to quantify technique.
Now, the VirClia Aspergillus Galactomannan AG, a new monotest using chemiluminescence developed by Vircell S.L. (Granada, Spain), could respond to these new diagnostic needs for testing individual samples immediately, without the need for batching, cumulating samples or sending out to an external laboratory. The VirClia Aspergillus Galactomannan AG has been successfully evaluated internally and externally against a commercially available ELISA reference assay and showed a 91% correlation.
Latest Industry News
- Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
- QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
- QIAGEN Showcases Integrated Sample-to-Insight Oncology Solutions at AACR 2026
- Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
- Partnership Expands Ultrasensitive WGS Assay for for Hematologic Malignancies and MRD Monitoring
- Beckman Coulter Gains CE Mark for Rapid Assay Distinguishing Bacterial vs Viral Infections
- Junction, GRAIL Enable Scalable Access to Multi-Cancer Detection Test
- GRAIL Partners with Epic to Integrate Multi-Cancer Test into EHR
- Global Partnership Aims to Streamline NGS Tumor Profiling in Oncology Trials
- Takara Bio USA and Hamilton Partner to Automate NGS Library Preparation
- Lunit and CellCarta Collaborate to Expand AI Pathology in CDx Development
- Integrated DNA Technologies Expands into Clinical Diagnostics
- Co-Diagnostics Agreement Expands Commercial and Distribution Reach in South Asia
- Automated MSI Test Gains IVDR Certification to Guide CRC Therapy
- New Partnership Brings Alzheimer’s Blood Biomarker Test to Community Screening Network
- MGI Tech Strengthens Sequencing Portfolio with Dual Acquisition
Channels
Clinical Chemistry
view channel
Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Alzheimer's disease often advances silently for years, making timely risk stratification difficult in routine practice. Current approaches to detect pathology can involve lumbar puncture or positron emission... Read more
Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
Routine blood tests that measure low-density lipoprotein (LDL), commonly known as “bad” cholesterol, are widely used to guide lipid-lowering therapy, but they do not always provide a complete picture of... Read more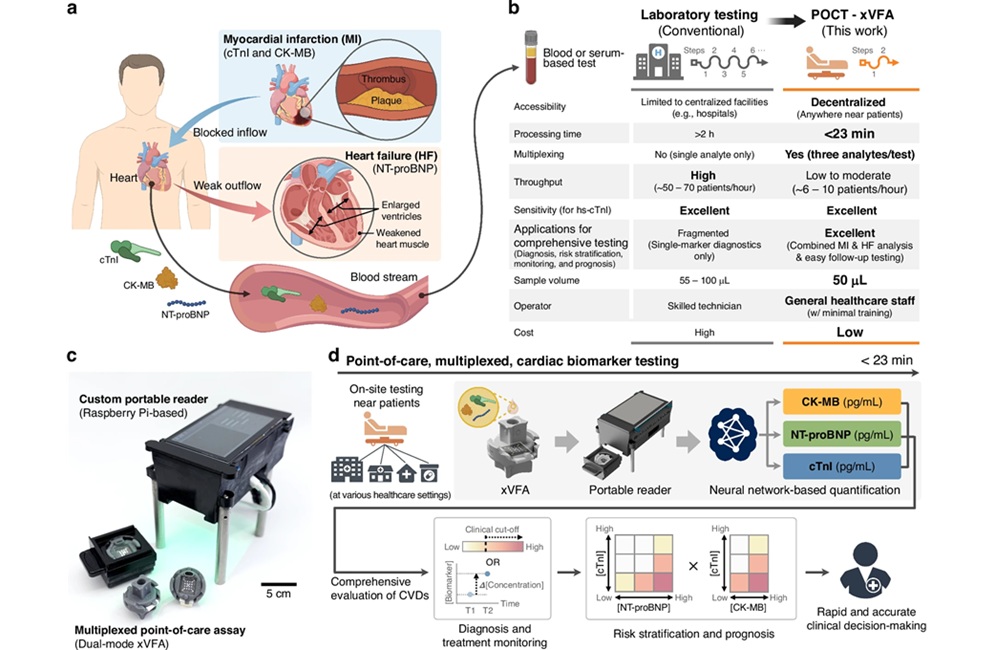
AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
Cardiovascular diseases are a leading cause of death, responsible for nearly 20 million deaths each year. Timely triage of myocardial infarction and heart failure hinges on rapid cardiac biomarker measurement,... Read moreNext Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
Clinical laboratories continue to face staffing shortages, limited space, and growing test volumes that pressure chemistry and electrolyte workflows. Maintaining rapid turnaround times increasingly depends... Read moreMolecular Diagnostics
view channel
Multi-Biomarker Blood Test Detects Early-Stage Cancers Across Types
Abbott is showcasing its Cancerguard multi-cancer early detection (MCED) test at the American Association for Cancer Research (AACR) Annual Meeting 2026, where new data highlight continued progress in... Read more
New Sample-to-Answer PCR System Supports High-Throughput Infectious Disease Testing
Clinical laboratories face mounting demand for rapid, high‑volume molecular testing for infectious diseases, including routine monitoring in immunocompromised patients. Consolidated, sample‑to‑answer workflows... Read moreHematology
view channel
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read more
Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
Chronic myelomonocytic leukemia (CMML) is a rare blood cancer in which acquired genetic mutations in bone marrow stem cells drive disease. Stem cell transplantation is the only curative option but carries... Read moreImmunology
view channel
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read more
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreMicrobiology
view channel
Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
Antibiotic resistance in early life is challenging to characterize, particularly around the timing and drivers of gene acquisition in newborns. Meconium, the first stool passed by infants, was long considered... Read more
mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
Central nervous system (CNS) infections such as encephalitis, ventriculitis, and brain abscess are among the most time-sensitive diagnostic challenges in neurology. Standard testing can return negative... Read morePathology
view channel
AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
Reliable prognostic profiling and biomarker screening are essential to guide oncology treatment decisions, while laboratories must balance speed and resource constraints. Earlier identification of high‑risk... Read more
Study Reveals Moleclar Mechanism Driving Aggressive Skin Cancer
Cutaneous squamous cell carcinoma (cSCC) is the world’s second most common skin cancer, and while many cases are treatable, a subset becomes highly aggressive and therapy‑resistant. Identifying molecular... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more