Urine-Based Kidney Transplantation Rejection Risk Assay Launched
|
By LabMedica International staff writers Posted on 30 Mar 2020 |
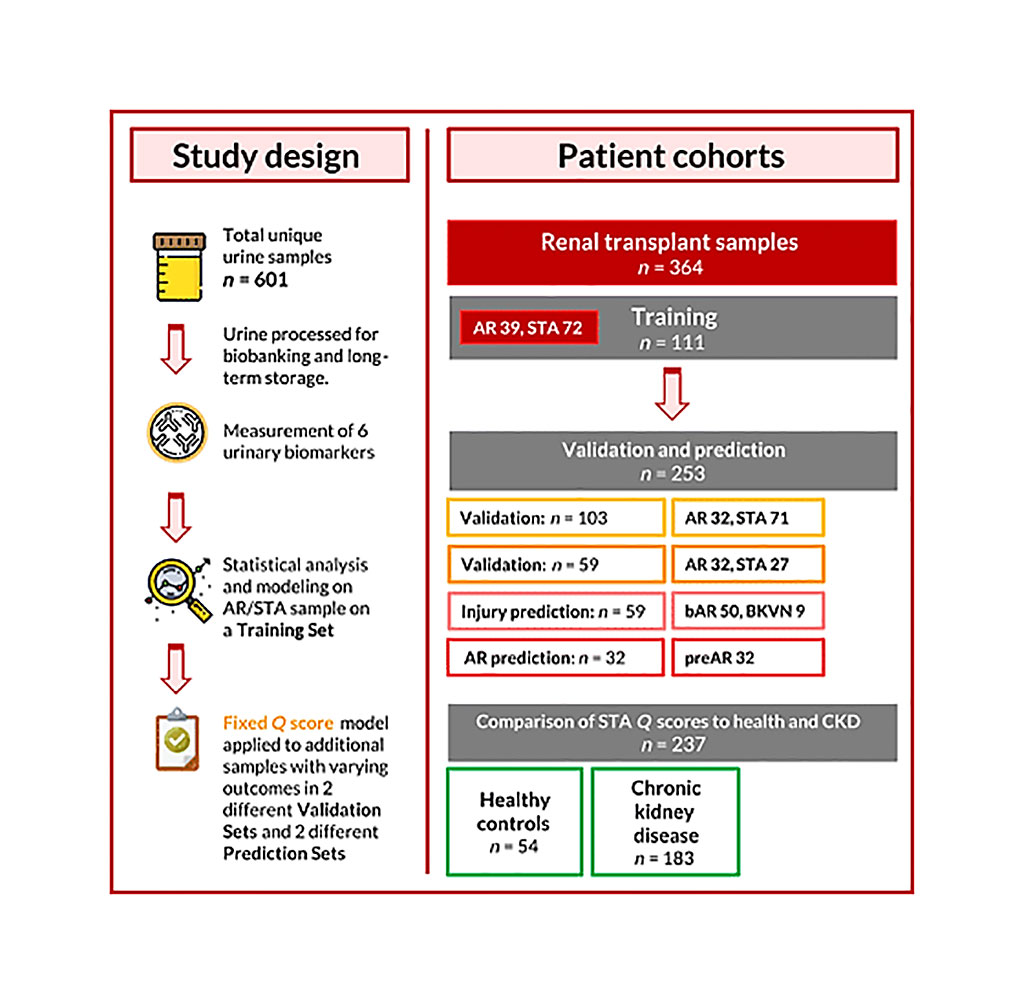
Image: Schematic diagram of a urine score for noninvasive accurate diagnosis and prediction of kidney transplant rejection study (Photo courtesy of the University of California, San Francisco).
For kidney transplant recipients, prompt and accurate detection of transplant rejection is vital for timely intervention. Unfortunately, the gold standard for diagnosis of rejection is kidney biopsy, an invasive procedure.
To diagnose acute rejection in kidney transplant patients, clinicians usually extract several small transplanted kidney samples every few months post-transplant, in addition to measuring the patient's serum creatinine levels. However, kidney biopsies can be invasive and expensive, while serum creatinine levels are often inaccurate and not sensitive enough to detect transplant rejection.
Scientists from the University of California, San Francisco (San Francisco, CA, USA) and their colleagues collected a total of 601 prospective urine samples from both pediatric and adult renal allograft recipients immediately before a renal allograft biopsy. Each sample was then paired with a renal transplant biopsy and classified into the following diagnoses: stable (170); acute rejection (AR, 103); borderline AR (bAR, 50); and BK virus nephropathy (9). The team also collected additional urine samples from 32 patients with AR before the rejection episode and paired them with biopsies.
The team evaluated a noninvasive, spot urine–based diagnostic assay based on measurements of six urinary DNA, protein, and metabolic biomarkers. The team used the QiSant assay (Nephrosant, San Francisco, CA, USA) that analyzes six biomarkers from 4 mL of urine sample: the amount of cell-free DNA (cfDNA); the fraction of methylated cfDNA; the proteins clusterin and creatinine; the inflammation marker CXCL10; and total protein amount in the urine. The assay uses a proprietary enzyme-linked immunosorbent (ELISA)-based tool, including a 5' biotinylated oligonucleated immunoprobe to target cfDNA fragments, and artificial intelligence to estimate the likelihood of acute kidney rejection. After collecting patient samples, the scientists developed a composite Q score — ranging from 0 to 100 — on all six biomarkers in a training set of 39 AR and 72 stable patients (STA).
In the first validation set, which had 32 AR patients and 71 STA patients, the group found that the score between the patient types had about a 91% clinical sensitivity and a 92% clinical specificity. Meanwhile, in a second validation set of 32 AR patients and 27 STA patients, the team found that the scaled score had 100% sensitivity and 96% specificity. Most patients with samples (159) with scores above the AR threshold had a clinical diagnosis of active AR, early AR, or went on to develop biopsy-confirmed AR up to 200 days after using the QiSant assay.
The authors concluded that they had demonstrated the clinical utility of this assay for predicting AR before a rise in the serum creatinine, enabling earlier detection of rejection than currently possible by standard of care tests. This noninvasive, sensitive, and quantitative approach is a robust and informative method for the rapid and routine monitoring of renal allografts. The study was published on March 18, 2020 in the journal Science Translational Medicine.
Related Links:
University of California, San Francisco
Nephrosant
To diagnose acute rejection in kidney transplant patients, clinicians usually extract several small transplanted kidney samples every few months post-transplant, in addition to measuring the patient's serum creatinine levels. However, kidney biopsies can be invasive and expensive, while serum creatinine levels are often inaccurate and not sensitive enough to detect transplant rejection.
Scientists from the University of California, San Francisco (San Francisco, CA, USA) and their colleagues collected a total of 601 prospective urine samples from both pediatric and adult renal allograft recipients immediately before a renal allograft biopsy. Each sample was then paired with a renal transplant biopsy and classified into the following diagnoses: stable (170); acute rejection (AR, 103); borderline AR (bAR, 50); and BK virus nephropathy (9). The team also collected additional urine samples from 32 patients with AR before the rejection episode and paired them with biopsies.
The team evaluated a noninvasive, spot urine–based diagnostic assay based on measurements of six urinary DNA, protein, and metabolic biomarkers. The team used the QiSant assay (Nephrosant, San Francisco, CA, USA) that analyzes six biomarkers from 4 mL of urine sample: the amount of cell-free DNA (cfDNA); the fraction of methylated cfDNA; the proteins clusterin and creatinine; the inflammation marker CXCL10; and total protein amount in the urine. The assay uses a proprietary enzyme-linked immunosorbent (ELISA)-based tool, including a 5' biotinylated oligonucleated immunoprobe to target cfDNA fragments, and artificial intelligence to estimate the likelihood of acute kidney rejection. After collecting patient samples, the scientists developed a composite Q score — ranging from 0 to 100 — on all six biomarkers in a training set of 39 AR and 72 stable patients (STA).
In the first validation set, which had 32 AR patients and 71 STA patients, the group found that the score between the patient types had about a 91% clinical sensitivity and a 92% clinical specificity. Meanwhile, in a second validation set of 32 AR patients and 27 STA patients, the team found that the scaled score had 100% sensitivity and 96% specificity. Most patients with samples (159) with scores above the AR threshold had a clinical diagnosis of active AR, early AR, or went on to develop biopsy-confirmed AR up to 200 days after using the QiSant assay.
The authors concluded that they had demonstrated the clinical utility of this assay for predicting AR before a rise in the serum creatinine, enabling earlier detection of rejection than currently possible by standard of care tests. This noninvasive, sensitive, and quantitative approach is a robust and informative method for the rapid and routine monitoring of renal allografts. The study was published on March 18, 2020 in the journal Science Translational Medicine.
Related Links:
University of California, San Francisco
Nephrosant
Latest Molecular Diagnostics News
- Simple Cytogenetic Method Could Improve Classification of ALL Subtypes
- Blood-Based Assay Enables Noninvasive Monitoring of Sarcoma Immunotherapy Response
- Genomic Test Guides Chemotherapy Decisions in Early-Stage Breast Cancer
- Tumor Mutation Marker Helps Refine Lung Cancer Prognosis and Guide Therapy Selection
- Multi-Cancer Test Boosts Detection When Added to Standard Screening
- Blood-Based MRD Monitoring Supports Relapse Prevention in Leukemia
- Genomic Test Predicts Chemotherapy Benefit in Metastatic Prostate Cancer
- Blood Protein Markers Flag Multiple Sclerosis Risk Years Before Diagnosis
- Digital PCR Assays Support Surveillance of Bundibugyo Ebolavirus Outbreak
- Updated Guidance Prioritizes Stool-Based Colorectal Cancer Screening Tests
- Blood-Based Proteomic Test May Predict Treatment Response in Non-Small Cell Lung Cancer
- Position Statements Outline Evidence Standards for Multi-Cancer Detection Tests
- Ultrasensitive MRD Blood Test Detects Early Breast Cancer Recurrence
- Gene Fusion Patterns May Flag High Risk Solitary Fibrous Tumors
- New RNA Origami Method Supports Faster Targeted Testing for Repeat Expansion Disorders
- FDA Approves Expanded Liquid Biopsy Panel for Advanced Cancer Profiling
Channels
Clinical Chemistry
view channel
Simple Dual-Tau Blood Test Detects and Stages Alzheimer’s Disease
Alzheimer’s disease is typically confirmed and staged with positron emission tomography scans and cerebrospinal fluid testing, procedures that are costly and invasive. Broader access to minimally invasive... Read more
Alzheimer’s Blood Biomarkers Linked to Early Cognitive Differences Before Dementia
Blood-based screening for Alzheimer’s disease offers a noninvasive, lower-cost alternative to brain imaging or spinal fluid testing, yet its ability to flag the earliest cognitive changes has been unclear.... Read moreMolecular Diagnostics
view channel
Genomic Test Guides Chemotherapy Decisions in Early-Stage Breast Cancer
Selecting adjuvant therapy for early-stage hormone receptor-positive breast cancer depends on accurately assessing long-term risk of distant recurrence. Clinical features alone can leave uncertainty about... Read more
Simple Cytogenetic Method Could Improve Classification of ALL Subtypes
Many cancers deviate from the normal chromosome number, but the clinical impact of extreme chromosome loss remains unclear. This widespread genomic disruption is associated with aggressive disease and... Read more
Blood-Based Assay Enables Noninvasive Monitoring of Sarcoma Immunotherapy Response
Sarcomas remain difficult to monitor during immunotherapy, as low tumor mutation burden can limit traditional circulating tumor DNA approaches and repeat tissue biopsies are often impractical in advanced disease.... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channel
Study Points to Autoimmune Pathway Behind Long COVID Symptoms
Long COVID leaves many SARS-CoV-2 survivors with persistent fatigue, cognitive issues, palpitations, and musculoskeletal pain for months or years. Estimates cited in new research suggest 4%–20% of infected... Read more
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channel