Molecular Assays Developed for Hantavirus Pulmonary Syndrome
|
By LabMedica International staff writers Posted on 16 Jan 2020 |
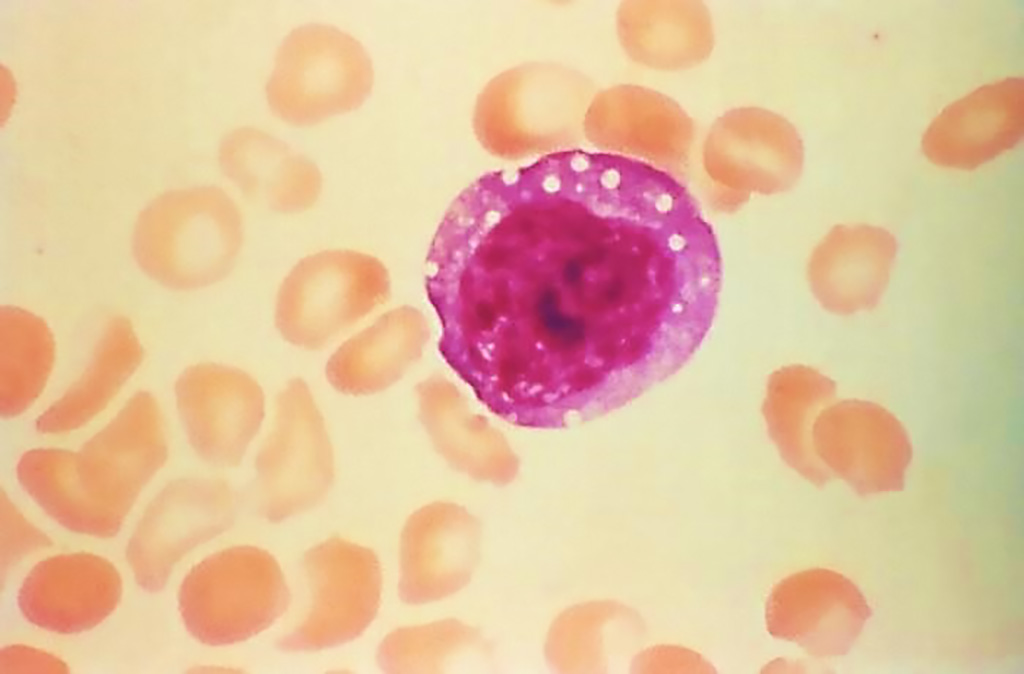
Image: This micrograph depicts an atypical, enlarged lymphocyte found in the blood smear from a hantavirus pulmonary syndrome (HPS) patient. This large atypical lymphocyte is an example of one of the laboratory findings, which when combined with a bandemia, i.e., immature white blood cells, and dropping platelet count, is characteristic of HPS (Photo courtesy of Centers for Disease Control and Prevention)
Hantavirus Pulmonary Syndrome (HPS) is a serious and often fatal disease caused by viruses known as hantaviruses. These anthropozoonotic diseases comprising of two clinical entities: Hemorrhagic Fever with Renal Syndrome (HFRS), which occurs in the Old World, and Hantavirus Pulmonary Syndrome (HPS) in the New World.
Laboratory diagnosis of HPS is often conducted by enzyme-linked immunosorbent assay (ELISA) for detection of immunoglobulin G (IgG) and IgM specific antibodies in serum or blood of suspected patients. Given that IgM antibodies are detectable early in the disease, IgM ELISA is considered the reference method for HPS diagnostics.
Scientists at the Evandro Chagas Institute (Ananindeua, Brazil) have developed real-time polymerase chain reaction (RT-qPCR) and semi-nested RT-qPCR assays for the diagnosis of HPS. The assays were developed for detection and quantification of four hantaviruses strains circulating in the Brazilian Amazon. assays were used to test two groups of samples: one comprising of 50 patients with disease and other containing samples from 50 healthy individuals according to IgM-ELISA results. A third group of 27 samples infected with other pathogens were tested for specificity analysis.
A consensus region in the N gene of these hantaviruses was used to design the primer sets and a hydrolysis probe. In vitro transcribed RNA was diluted in standards with known concentration. MS2 bacteriophage RNA was detected together with hantavirus RNA as an exogenous control in a duplex reaction. RT-qPCR efficiency was around 100% and the limit of detection was 0.9 copies/μL of RNA for RT-qPCR and 10 copies/μL of RNA for Semi-nested RT-PCR. There was no amplification of either negative samples or samples positive to other pathogens. Sample reactions were performed in triplicate in ViiA7 qPCR System (Applied Biosystems, Thermo-Fisher Scientific, Waltham, MA, USA).
The investigators reported that the RT-qPCR was more sensitive than semi-nested RT-PCR, being able to detect three samples undetected by conventional RT-PCR. RT-qPCR clinical sensitivity, specificity and general accuracy values were 92.5%, 100% and 97.63%, respectively. Taken together, these results indicate that both RT-qPCR and Semi-nested RT-PCR assays are efficient, sensitive and specific tools for genome detection of hantaviruses circulating in the Amazon region. In addition to being more sensitive, RT-qPCR offers other advantages such as agility in generating results, about three times faster than conventional assays and automation capability which leads to lower risk of contamination and greater reproducibility. The study was published on December 26, 2019 in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Evandro Chagas Institute
Applied Biosystems
Laboratory diagnosis of HPS is often conducted by enzyme-linked immunosorbent assay (ELISA) for detection of immunoglobulin G (IgG) and IgM specific antibodies in serum or blood of suspected patients. Given that IgM antibodies are detectable early in the disease, IgM ELISA is considered the reference method for HPS diagnostics.
Scientists at the Evandro Chagas Institute (Ananindeua, Brazil) have developed real-time polymerase chain reaction (RT-qPCR) and semi-nested RT-qPCR assays for the diagnosis of HPS. The assays were developed for detection and quantification of four hantaviruses strains circulating in the Brazilian Amazon. assays were used to test two groups of samples: one comprising of 50 patients with disease and other containing samples from 50 healthy individuals according to IgM-ELISA results. A third group of 27 samples infected with other pathogens were tested for specificity analysis.
A consensus region in the N gene of these hantaviruses was used to design the primer sets and a hydrolysis probe. In vitro transcribed RNA was diluted in standards with known concentration. MS2 bacteriophage RNA was detected together with hantavirus RNA as an exogenous control in a duplex reaction. RT-qPCR efficiency was around 100% and the limit of detection was 0.9 copies/μL of RNA for RT-qPCR and 10 copies/μL of RNA for Semi-nested RT-PCR. There was no amplification of either negative samples or samples positive to other pathogens. Sample reactions were performed in triplicate in ViiA7 qPCR System (Applied Biosystems, Thermo-Fisher Scientific, Waltham, MA, USA).
The investigators reported that the RT-qPCR was more sensitive than semi-nested RT-PCR, being able to detect three samples undetected by conventional RT-PCR. RT-qPCR clinical sensitivity, specificity and general accuracy values were 92.5%, 100% and 97.63%, respectively. Taken together, these results indicate that both RT-qPCR and Semi-nested RT-PCR assays are efficient, sensitive and specific tools for genome detection of hantaviruses circulating in the Amazon region. In addition to being more sensitive, RT-qPCR offers other advantages such as agility in generating results, about three times faster than conventional assays and automation capability which leads to lower risk of contamination and greater reproducibility. The study was published on December 26, 2019 in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Evandro Chagas Institute
Applied Biosystems
Latest Microbiology News
- Gut Microbiome Signatures Help Identify Risk of IBD Progression
- FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
- New AMR Assay Supports Rapid Infection Control Screening in Hospitals
- Diagnostic Gaps Complicate Bundibugyo Ebola Outbreak Response in Congo
- Study Finds Hidden Mpox Infections May Drive Ongoing Spread
- Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
- Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
- Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
- Label-Free Microscopy Method Enables Faster, Quantitative Detection of Malaria
- Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
Channels
Clinical Chemistry
view channel
Simple Dual-Tau Blood Test Detects and Stages Alzheimer’s Disease
Alzheimer’s disease is typically confirmed and staged with positron emission tomography scans and cerebrospinal fluid testing, procedures that are costly and invasive. Broader access to minimally invasive... Read more
Alzheimer’s Blood Biomarkers Linked to Early Cognitive Differences Before Dementia
Blood-based screening for Alzheimer’s disease offers a noninvasive, lower-cost alternative to brain imaging or spinal fluid testing, yet its ability to flag the earliest cognitive changes has been unclear.... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channel
Study Points to Autoimmune Pathway Behind Long COVID Symptoms
Long COVID leaves many SARS-CoV-2 survivors with persistent fatigue, cognitive issues, palpitations, and musculoskeletal pain for months or years. Estimates cited in new research suggest 4%–20% of infected... Read more
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read morePathology
view channel
Blood-Based Method Tracks Gene Activity in the Living Brain
Real-time measurement of gene activity in the brain has been limited by assays requiring destructive tissue sampling. Tracking active genes could reveal how the body responds to environmental factors,... Read more
FDA Approval Expands Automated PD-L1 Testing Across Solid Tumors
Clinical laboratories play a central role in guiding immunotherapy by reporting programmed death ligand-1 (PD‑L1) status across multiple solid tumors. Many sites are standardizing this work on fully automated... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channel
























