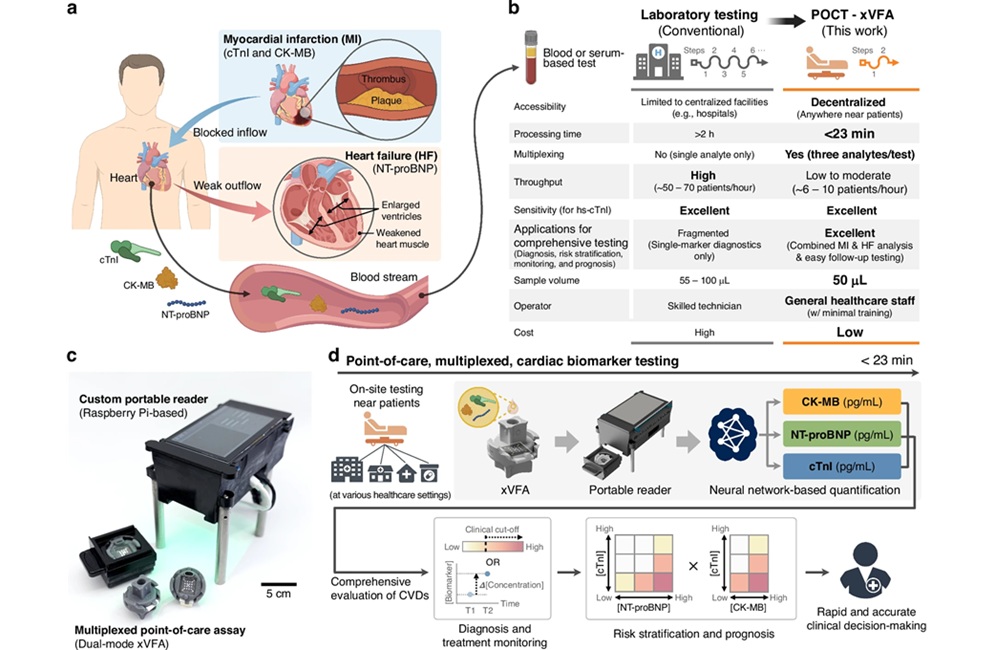
Urinary Detection Method Developed for Prostate Cancer
|
By LabMedica International staff writers Posted on 22 Jan 2019 |

Image: The Synergy H1 multi-mode microplate reader (Photo courtesy of BioTek).
Prostate cancer (PCa) is one of the most common types of malignancy worldwide and is the second leading cause of cancer death among men. This cancer tends to be asymptomatic and slow growing, often with onset in young men, but usually not detected until the age of 40 to 50 years.
The conventional methods for PCa screening recommended by the American Cancer Society are serum prostate specific antigen (PSA) testing and digital rectal examination (DRE). However, these methods have some drawbacks due to their sensitivity, specificity and accuracy. The PCA3 gene has shown promise as a non-invasive PCa biomarker.
Scientists at Mahidol University (Bangkok, Thailand) collected spot urine samples from five healthy male volunteers, first voided post-DRE urine from five benign prostate hyperplasia (BPH) patients and from five PCa patients. Diagnosis of patients was made by histopathological analysis after prostate biopsy subsequently. PCa patients were identified with positive biopsy.
Total RNA was isolated from the cell pellets of urine as well as from cell lines and total RNA was converted to cDNA using RevertAid First Strand cDNA synthesis kit. The team developed an assay based on interactions between unmodified gold nanoparticles (AuNPs) and thiolated polymerase chain reaction (PCR) products. Thiolated PCR products were amplified by RT-PCR using a thiol-labeled primer at the 5′ end. Thiolated products of PCA3 bound to the surface of AuNPs and led to the prevention of salt-induced aggregation (red color). In the absence of the PCR products, AuNPs changed their color from red to blue due to the salt-induced aggregation. These changes were detected by the naked eye and a microplate spectrophotometer.
The team reported that assay was specific for PCA3 in prostate cancer cell lines with a visual detection limit of 31.25 ng/reaction. The absorption ratio 520/640 nm was linear against PCR product concentration in the reaction. This method is promising for discrimination of prostate cancer patients from both healthy controls and benign prostatic hyperplasia patients according to their urinary PCA3 expression levels. The results indicated that the proposed colorimetric assay was more sensitive than gel electrophoresis.
The authors concluded that a sensitive and specific AuNP-based colorimetric method for visual detection of PCA3 in prostate cancer was successfully developed. This new method was based on interactions between thiolated PCR products and unmodified AuNPs. The positive and negative results were clearly distinguished by the naked eye, being red and blue color, respectively. The incubation time was short and results were obtained within 10 minutes of RT-PCR completion. Moreover, a large number of samples could be tested simultaneously in 96-well microtiter plates. The study was published in the January 2019 issue of the journal Clinica Chimica Acta.
Related Links:
Mahidol University
The conventional methods for PCa screening recommended by the American Cancer Society are serum prostate specific antigen (PSA) testing and digital rectal examination (DRE). However, these methods have some drawbacks due to their sensitivity, specificity and accuracy. The PCA3 gene has shown promise as a non-invasive PCa biomarker.
Scientists at Mahidol University (Bangkok, Thailand) collected spot urine samples from five healthy male volunteers, first voided post-DRE urine from five benign prostate hyperplasia (BPH) patients and from five PCa patients. Diagnosis of patients was made by histopathological analysis after prostate biopsy subsequently. PCa patients were identified with positive biopsy.
Total RNA was isolated from the cell pellets of urine as well as from cell lines and total RNA was converted to cDNA using RevertAid First Strand cDNA synthesis kit. The team developed an assay based on interactions between unmodified gold nanoparticles (AuNPs) and thiolated polymerase chain reaction (PCR) products. Thiolated PCR products were amplified by RT-PCR using a thiol-labeled primer at the 5′ end. Thiolated products of PCA3 bound to the surface of AuNPs and led to the prevention of salt-induced aggregation (red color). In the absence of the PCR products, AuNPs changed their color from red to blue due to the salt-induced aggregation. These changes were detected by the naked eye and a microplate spectrophotometer.
The team reported that assay was specific for PCA3 in prostate cancer cell lines with a visual detection limit of 31.25 ng/reaction. The absorption ratio 520/640 nm was linear against PCR product concentration in the reaction. This method is promising for discrimination of prostate cancer patients from both healthy controls and benign prostatic hyperplasia patients according to their urinary PCA3 expression levels. The results indicated that the proposed colorimetric assay was more sensitive than gel electrophoresis.
The authors concluded that a sensitive and specific AuNP-based colorimetric method for visual detection of PCA3 in prostate cancer was successfully developed. This new method was based on interactions between thiolated PCR products and unmodified AuNPs. The positive and negative results were clearly distinguished by the naked eye, being red and blue color, respectively. The incubation time was short and results were obtained within 10 minutes of RT-PCR completion. Moreover, a large number of samples could be tested simultaneously in 96-well microtiter plates. The study was published in the January 2019 issue of the journal Clinica Chimica Acta.
Related Links:
Mahidol University
Latest Molecular Diagnostics News
- Machine Learning Model Uses DNA Methylation to Predict Tumor Origin in Cancers of Unknown Primary
- Blood Test Enables Early Detection and Classification of Glioma
- Multi-Biomarker Blood Test Detects Early-Stage Cancers Across Types
- New Sample-to-Answer PCR System Supports High-Throughput Infectious Disease Testing
- Framework Guides Targeted Immunotherapy Selection in Liver Cancer
- Collaboration Brings Rapid At-Home STI Testing with Virtual Follow-Up
- Blood-Based Epigenetic Signals Enable Osteosarcoma Disease Monitoring
- Host–Virus Genetic Interactions Drive Nasopharyngeal Cancer Risk
- AI-Enabled Biochip Detects microRNA Biomarkers in Minutes
- Blood Test Detects Early Pancreatic Cancer in High-Risk Patients
- Long-Read RNA Sequencing Platform Improves Rare Disease Diagnosis
- Study Confirms Barrett’s Esophagus as Precursor to Esophageal Cancer
- Ultrasensitive Assay Reveals Previously Undetected Tuberculosis in Hospital Patients
- CE-Marked Blood Test Enables Monitoring of Neuroinflammation in Multiple Sclerosis
- Urine-Based Assay Predicts Severe Dengue Risk Early
- Ultrasensitive Assay Tracks Resistance Mutations MRD Monitoring
Channels
Clinical Chemistry
view channel
Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
Early detection across multiple cancers remains a major unmet need in population screening. Non-invasive approaches that can be delivered at scale may broaden access and shift diagnoses to earlier stages.... Read more
Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Alzheimer's disease often advances silently for years, making timely risk stratification difficult in routine practice. Current approaches to detect pathology can involve lumbar puncture or positron emission... Read moreMolecular Diagnostics
view channel
Machine Learning Model Uses DNA Methylation to Predict Tumor Origin in Cancers of Unknown Primary
Cancers of unknown primary (CUP) are metastatic malignancies in which the primary site cannot be identified, complicating treatment selection. Many patients consequently receive broad, nonspecific chemotherapy... Read more
Blood Test Enables Early Detection and Classification of Glioma
High-grade gliomas, particularly glioblastoma, are fast-growing brain tumors that are often diagnosed late and typically require invasive procedures for confirmation. Current pathways rely on symptoms,... Read more
Multi-Biomarker Blood Test Detects Early-Stage Cancers Across Types
Abbott is showcasing its Cancerguard multi-cancer early detection (MCED) test at the American Association for Cancer Research (AACR) Annual Meeting 2026, where new data highlight continued progress in... Read more
New Sample-to-Answer PCR System Supports High-Throughput Infectious Disease Testing
Clinical laboratories face mounting demand for rapid, high‑volume molecular testing for infectious diseases, including routine monitoring in immunocompromised patients. Consolidated, sample‑to‑answer workflows... Read moreHematology
view channel
Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
Diffuse large B-cell lymphoma (DLBCL) is the most common form of non-Hodgkin lymphoma and often presents with aggressive clinical behavior. Although many patients respond to standard chemotherapy with... Read more
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read moreImmunology
view channel
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read more
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreMicrobiology
view channel
Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
Melanoma remains prone to relapse even after surgery and adjuvant immunotherapy, with 25% to 40% of patients experiencing recurrence. Clinicians lack reliable pre-treatment indicators to identify those... Read more
Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
Gram-negative bloodstream infections and sepsis demand fast, precise antimicrobial therapy, yet conventional susceptibility workflows can delay targeted treatment. Clinical laboratories need platforms... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
QuidelOrtho Corporation has completed the acquisition of LEX Diagnostics for approximately USD 100 million in cash. The transaction adds the LEX VELO System to QuidelOrtho’s portfolio. The platform received U.... Read more
Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
Seegene introduced STAgora, a real-time data analytics platform built on aggregated statistical testing data, at ESCMID Global 2026 in Munich, where it also presented an enhanced model of its automated... Read more
Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
Foundation Medicine, Inc., an independent affiliate of Roche, announced plans to expand its monitoring portfolio with SAGA Diagnostics’ Pathlight, a personalized, tumor-informed molecular residual disease... Read more