FDA Clears Diagnostic Assay for Group B Strep
|
By LabMedica International staff writers Posted on 10 Aug 2018 |

Image: The NeuMoDx GBS assay is run on the 288 molecular system (Photo courtesy of NeuMoDx Molecular).
Group B Streptococcus (GBS) is a Gram-positive bacterium found in 10% to 35% of healthy adults and is a commonly found bacterium within the human body. A person who carries GBS and does not present symptoms of GBS disease is said to be “colonized” with GBS.
GBS colonization is not considered contagious, however, under certain circumstances, GBS can invade the body and cause serious infection; this is referred to as Group B Streptococcal disease. It is a leading cause of life-threatening bacterial infection in newborns who acquire the disease during birth by vertical (mother-to-baby) transmission. Approximately 2,000 cases of newborn infections occur per year, with estimates of a mortality rate of 0.27 per 1,000 live births.
The US Food and Drug Administration (FDA, Silver Springs, MD, USA) have granted 510(k) clearance to a high-throughput molecular diagnostics platform and Group B Strep assay from NeuMoDx Molecular (Ann Arbor, MI, USA). The 288 Molecular System is an automated sample-to-answer diagnostics platform that runs up to 288 patient samples in continuous random-access mode. The time to first results is approximately one hour, according to the firm, with a walkaway window of more than six hours, and NeuMoDx intends the system to be used in central laboratories of hospitals and clinical reference laboratories.
Testing for GBS colonization is usually performed in the third trimester of pregnancy to prevent complications of transmission to a newborn during labor and delivery. The NeuMoDx GBS Assay is a qualitative polymerase chain reaction (PCR)-based diagnostic test to detect an 88 base-pair region of the pcsB gene and incorporates sample lysis, automated DNA extraction, and real-time PCR. The firm said the assay showed a sensitivity of 97% and specificity of 96% compared to culture in a multicenter clinical performance study.
The revolutionary dry format reagents require no refrigeration and are extremely robust with an on-board stability of greater than 60 days, and ambient temperature storage shelf life of greater than one year. Additionally, the unitized format of the dry reagents significantly increases operating efficiency while minimizing the waste associated with systems requiring manual reconstitution and/or use of bulk format lyophilized reagents.
Related Links:
US Food and Drug Administration
NeuMoDx Molecular
GBS colonization is not considered contagious, however, under certain circumstances, GBS can invade the body and cause serious infection; this is referred to as Group B Streptococcal disease. It is a leading cause of life-threatening bacterial infection in newborns who acquire the disease during birth by vertical (mother-to-baby) transmission. Approximately 2,000 cases of newborn infections occur per year, with estimates of a mortality rate of 0.27 per 1,000 live births.
The US Food and Drug Administration (FDA, Silver Springs, MD, USA) have granted 510(k) clearance to a high-throughput molecular diagnostics platform and Group B Strep assay from NeuMoDx Molecular (Ann Arbor, MI, USA). The 288 Molecular System is an automated sample-to-answer diagnostics platform that runs up to 288 patient samples in continuous random-access mode. The time to first results is approximately one hour, according to the firm, with a walkaway window of more than six hours, and NeuMoDx intends the system to be used in central laboratories of hospitals and clinical reference laboratories.
Testing for GBS colonization is usually performed in the third trimester of pregnancy to prevent complications of transmission to a newborn during labor and delivery. The NeuMoDx GBS Assay is a qualitative polymerase chain reaction (PCR)-based diagnostic test to detect an 88 base-pair region of the pcsB gene and incorporates sample lysis, automated DNA extraction, and real-time PCR. The firm said the assay showed a sensitivity of 97% and specificity of 96% compared to culture in a multicenter clinical performance study.
The revolutionary dry format reagents require no refrigeration and are extremely robust with an on-board stability of greater than 60 days, and ambient temperature storage shelf life of greater than one year. Additionally, the unitized format of the dry reagents significantly increases operating efficiency while minimizing the waste associated with systems requiring manual reconstitution and/or use of bulk format lyophilized reagents.
Related Links:
US Food and Drug Administration
NeuMoDx Molecular
Latest Microbiology News
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
- RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
- Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
- New Bacterial Target Identified for Early Detection of Noma
- Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
- Rapid Urine Test Speeds Antibiotic Selection for UTIs
- WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
- Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
- WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
- New Imaging Approach Could Help Predict Dangerous Gut Infection
Channels
Clinical Chemistry
view channel
Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
Early detection across multiple cancers remains a major unmet need in population screening. Non-invasive approaches that can be delivered at scale may broaden access and shift diagnoses to earlier stages.... Read more
Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Alzheimer's disease often advances silently for years, making timely risk stratification difficult in routine practice. Current approaches to detect pathology can involve lumbar puncture or positron emission... Read moreMolecular Diagnostics
view channel
Machine Learning Model Uses DNA Methylation to Predict Tumor Origin in Cancers of Unknown Primary
Cancers of unknown primary (CUP) are metastatic malignancies in which the primary site cannot be identified, complicating treatment selection. Many patients consequently receive broad, nonspecific chemotherapy... Read more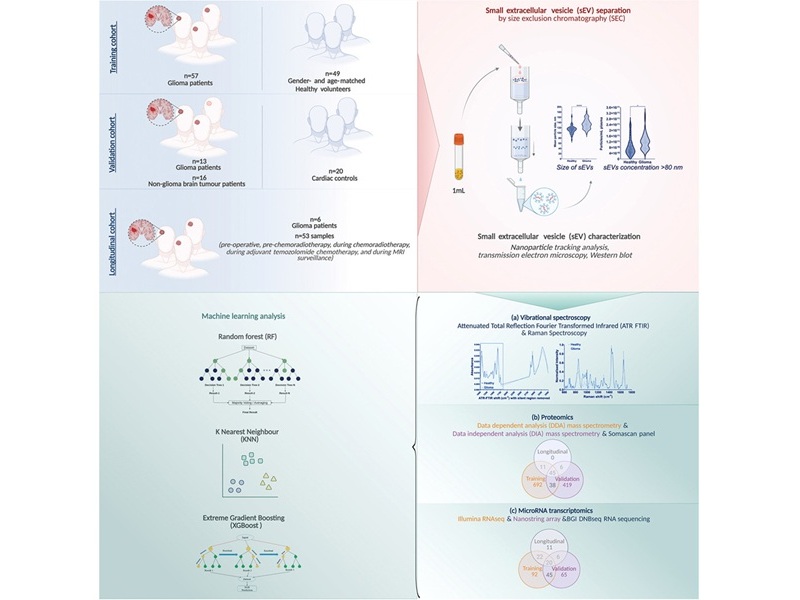
Blood Test Enables Early Detection and Classification of Glioma
High-grade gliomas, particularly glioblastoma, are fast-growing brain tumors that are often diagnosed late and typically require invasive procedures for confirmation. Current pathways rely on symptoms,... Read more
Multi-Biomarker Blood Test Detects Early-Stage Cancers Across Types
Abbott is showcasing its Cancerguard multi-cancer early detection (MCED) test at the American Association for Cancer Research (AACR) Annual Meeting 2026, where new data highlight continued progress in... Read more
New Sample-to-Answer PCR System Supports High-Throughput Infectious Disease Testing
Clinical laboratories face mounting demand for rapid, high‑volume molecular testing for infectious diseases, including routine monitoring in immunocompromised patients. Consolidated, sample‑to‑answer workflows... Read moreHematology
view channel
Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
Diffuse large B-cell lymphoma (DLBCL) is the most common form of non-Hodgkin lymphoma and often presents with aggressive clinical behavior. Although many patients respond to standard chemotherapy with... Read more
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read moreImmunology
view channel
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read more
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read morePathology
view channel
AI Pathology Model Predicts Immunotherapy Response in Lung Cancer
Clinicians face persistent challenges identifying which patients with metastatic non-small cell lung cancer will benefit from immunotherapy, even as these agents transform oncology care.... Read more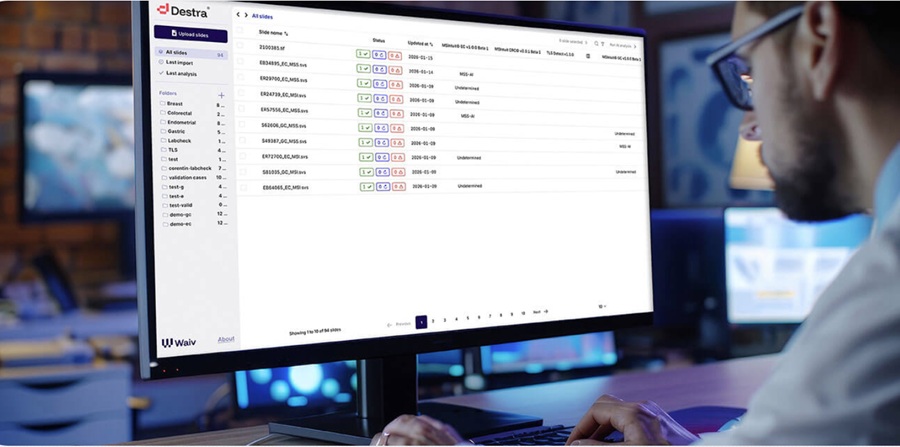
AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
Reliable prognostic profiling and biomarker screening are essential to guide oncology treatment decisions, while laboratories must balance speed and resource constraints. Earlier identification of high‑risk... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
QuidelOrtho Corporation has completed the acquisition of LEX Diagnostics for approximately USD 100 million in cash. The transaction adds the LEX VELO System to QuidelOrtho’s portfolio. The platform received U.... Read more
Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
Seegene introduced STAgora, a real-time data analytics platform built on aggregated statistical testing data, at ESCMID Global 2026 in Munich, where it also presented an enhanced model of its automated... Read more
Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
Foundation Medicine, Inc., an independent affiliate of Roche, announced plans to expand its monitoring portfolio with SAGA Diagnostics’ Pathlight, a personalized, tumor-informed molecular residual disease... Read more