Glioma Risk Lower with Elevated Blood Sugar
|
By LabMedica International staff writers Posted on 16 May 2017 |
Image: New research suggests that there may be a link between blood sugar and glioma (Photo courtesy of MNT).
While many cancers are more common among those with diabetes, cancerous brain tumors called gliomas are less common among those with elevated blood sugar and diabetes.
Glioma is a heterogeneous primary brain tumor for which there is no treatment that ensures long-term survival and patients diagnosed with the most common adult form of this tumor, glioblastoma, survive on average only 14 months.
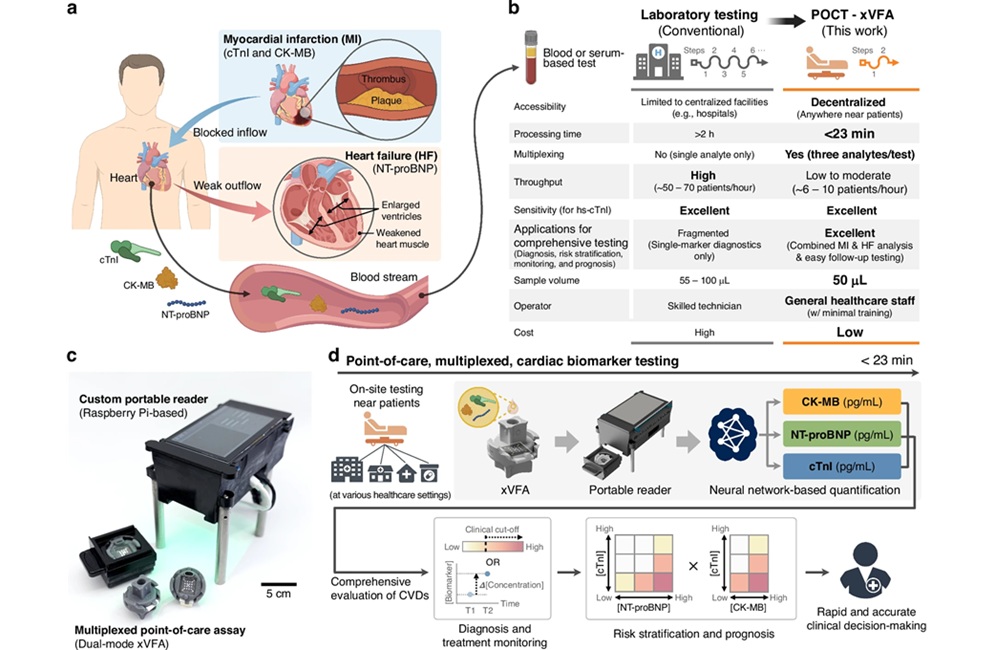
An international team of scientists led by those at the Ohio State University evaluated blood sugar and diabetes data and its relationship to subsequent development of brain cancer and found that those with elevated blood sugar and diabetes had a lower risk of developing glioma. They used data from the Apolipoprotein MOrtality RISk (AMORIS) (n = 528,580) and the Metabolic syndrome and Cancer project (Me-Can) cohorts (n = 269,365). They identified individuals who were followed for a maximum of 15 years after their first blood glucose test until glioma diagnosis, death, emigration or the end of follow-up.
They conducted an exploratory analysis of the 51 glioma cases using the Me-Can data set with multiple measurements to determine whether glucose values equal to or less than 7.0 mmol/L varied with the number of measurements or time between the blood glucose test and glioma diagnosis. They found that fasting and non-fasting glucose dose-response relationships were similar allowing them to combine data from these two groups.
They found that the relative proportions of male and female total glioma cases differ between the cohorts (in AMORIS 65% are men; in Me-Can 52%), as does fasting status (in AMORIS 61% of blood was drawn when total glioma cases were fasting; in Me-Can all blood samples were collected from fasting participants). Pre-diagnostic blood glucose levels were inversely related to glioma risk and pre-diagnostic diabetes. During the year before diagnosis, blood glucose was inversely associated with glioma in the AMORIS but not the Me-Can cohort. This AMORIS result is consistent with their hypothesis that excess glucose consumption by the preclinical tumor accounts for the inverse association between blood glucose and glioma.
Judith Schwartzbaum, PhD, a professor and lead author of the study said, “Diabetes and elevated blood sugar increase the risk of cancer at several sites including the colon, breast and bladder. But in this case, these rare malignant brain tumors are more common among people who have normal levels of blood glucose than those with high blood sugar or diabetes.” The study was published on May 3, 2017, in the journal Scientific Reports.
Glioma is a heterogeneous primary brain tumor for which there is no treatment that ensures long-term survival and patients diagnosed with the most common adult form of this tumor, glioblastoma, survive on average only 14 months.
An international team of scientists led by those at the Ohio State University evaluated blood sugar and diabetes data and its relationship to subsequent development of brain cancer and found that those with elevated blood sugar and diabetes had a lower risk of developing glioma. They used data from the Apolipoprotein MOrtality RISk (AMORIS) (n = 528,580) and the Metabolic syndrome and Cancer project (Me-Can) cohorts (n = 269,365). They identified individuals who were followed for a maximum of 15 years after their first blood glucose test until glioma diagnosis, death, emigration or the end of follow-up.
They conducted an exploratory analysis of the 51 glioma cases using the Me-Can data set with multiple measurements to determine whether glucose values equal to or less than 7.0 mmol/L varied with the number of measurements or time between the blood glucose test and glioma diagnosis. They found that fasting and non-fasting glucose dose-response relationships were similar allowing them to combine data from these two groups.
They found that the relative proportions of male and female total glioma cases differ between the cohorts (in AMORIS 65% are men; in Me-Can 52%), as does fasting status (in AMORIS 61% of blood was drawn when total glioma cases were fasting; in Me-Can all blood samples were collected from fasting participants). Pre-diagnostic blood glucose levels were inversely related to glioma risk and pre-diagnostic diabetes. During the year before diagnosis, blood glucose was inversely associated with glioma in the AMORIS but not the Me-Can cohort. This AMORIS result is consistent with their hypothesis that excess glucose consumption by the preclinical tumor accounts for the inverse association between blood glucose and glioma.
Judith Schwartzbaum, PhD, a professor and lead author of the study said, “Diabetes and elevated blood sugar increase the risk of cancer at several sites including the colon, breast and bladder. But in this case, these rare malignant brain tumors are more common among people who have normal levels of blood glucose than those with high blood sugar or diabetes.” The study was published on May 3, 2017, in the journal Scientific Reports.
Latest Clinical Chem. News
- Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
- Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
- Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
- AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
- Next Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
- Blood Metabolite Test Detects Early Cognitive Decline
- AI-Based Blood Test Diagnose Multiple Brain Disorders from Blood Sample
- Automated NfL Assay Supports Monitoring of Neurological Disorders
- Blood-Based Screening Test Targets Early Detection of Colorectal Cancer
- New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
- CSF Biomarker Improves Diagnosis of Parkinson’s Disease and Lewy Body Dementia
- Simple Urine Home Test Kit Could Detect Early-Stage Breast Cancer
- Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
- New Tool Tracks Biomarker Changes to Predict Myeloma Progression
- New Plasma Tau Assay Improves Prediction of Alzheimer’s Progression
- First IVD Immunoassay to Detect Alzheimer’s Risk Gene Variant Receives CE Mark
Channels
Clinical Chemistry
view channel
Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
Early detection across multiple cancers remains a major unmet need in population screening. Non-invasive approaches that can be delivered at scale may broaden access and shift diagnoses to earlier stages.... Read more
Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Alzheimer's disease often advances silently for years, making timely risk stratification difficult in routine practice. Current approaches to detect pathology can involve lumbar puncture or positron emission... Read moreMolecular Diagnostics
view channel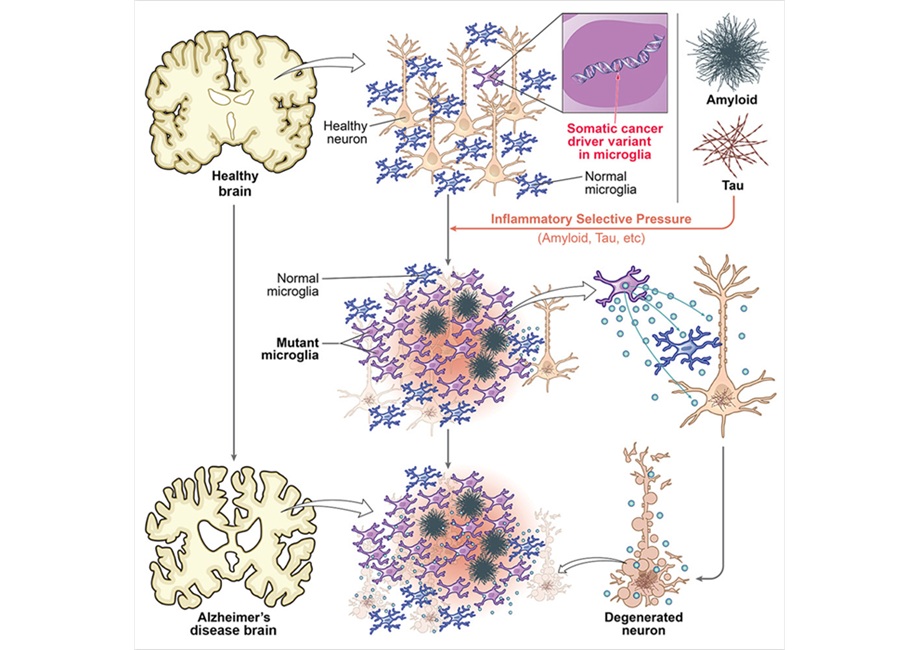
Cancer-Related Mutations in Immune Cells Linked to Alzheimer’s
Alzheimer’s disease is marked by protein aggregation and inflammatory changes in the brain’s immune system, yet its molecular drivers remain incompletely understood. With aging, human cells accumulate... Read more
Composite Blood Biomarkers Enable Early Detection of Common Cancers
Early diagnosis of colorectal, lung, and ovarian cancers remains challenging, with many patients identified only after tumors have begun to spread. A scalable blood test could expand access to screening,... Read more
Machine Learning Model Uses DNA Methylation to Predict Tumor Origin in Cancers of Unknown Primary
Cancers of unknown primary (CUP) are metastatic malignancies in which the primary site cannot be identified, complicating treatment selection. Many patients consequently receive broad, nonspecific chemotherapy... Read moreHematology
view channel
Single Assay Enables Rapid HLA and ABO Genotyping for Transplant Matching
CareDx (Brisbane, CA, USA) has introduced AlloSeq Nano, a nanopore‑based HLA (human leukocyte antigen) and ABO genotyping solution unveiled at the European Federation for Immunogenetics (EFI) Conference 2026.... Read more
Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
Diffuse large B-cell lymphoma (DLBCL) is the most common form of non-Hodgkin lymphoma and often presents with aggressive clinical behavior. Although many patients respond to standard chemotherapy with... Read moreImmunology
view channel
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read more
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreMicrobiology
view channel
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read more
Label-Free Microscopy Methodd Enables Faster, Quantitative Detection of Malaria
Microscopy of blood smears remains a cornerstone for malaria diagnosis but can be slow, stain-dependent, and operator intensive. With more than 200 million infections and over 600,000 deaths annually,... Read more
Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
Melanoma remains prone to relapse even after surgery and adjuvant immunotherapy, with 25% to 40% of patients experiencing recurrence. Clinicians lack reliable pre-treatment indicators to identify those... Read more
Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
Gram-negative bloodstream infections and sepsis demand fast, precise antimicrobial therapy, yet conventional susceptibility workflows can delay targeted treatment. Clinical laboratories need platforms... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
QuidelOrtho Corporation has completed the acquisition of LEX Diagnostics for approximately USD 100 million in cash. The transaction adds the LEX VELO System to QuidelOrtho’s portfolio. The platform received U.... Read more
Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
Seegene introduced STAgora, a real-time data analytics platform built on aggregated statistical testing data, at ESCMID Global 2026 in Munich, where it also presented an enhanced model of its automated... Read more
Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
Foundation Medicine, Inc., an independent affiliate of Roche, announced plans to expand its monitoring portfolio with SAGA Diagnostics’ Pathlight, a personalized, tumor-informed molecular residual disease... Read more






.jpg)