Biomarker Outperforms Current Gold Standard to Detect Brain Shunt Infections
|
By LabMedica International staff writers Posted on 15 Sep 2016 |
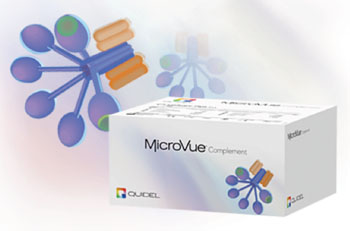
Image: The MicroVue complement sC5b-9 Plus enzyme immunoassay kit (Photo courtesy of Quidel Corporation).
Children treated with cerebrospinal fluid (CSF) shunts to manage hydrocephalus frequently develop shunt failure and/or infections, conditions that present with overlapping symptoms.
The potential life-threatening nature of shunt infections requires rapid diagnosis; however, traditional microbiology is time consuming, expensive, and potentially unreliable. A quick and accurate test for bacterial infection in brain shunts or meningitis would improve the quality and efficiency of patient care and patient outcomes. Such a test would also lower health care expenses by avoiding needless hospitalization and treatment.
Scientists at the University of Alabama (Birmingham, AL, USA) prospectively enrolled 198 consecutive undergoing evaluation and treatment for newly diagnosed hydrocephalus, shunt infection, and versus malfunction. Study samples were collected at the same time as routine lab specimens to provide corresponding laboratory results for each specimen (glucose, protein, hematology indices, gram stain, and culture). Samples were labeled and stored at –20 °C and/or –80 °C for a period of one week on average prior to analysis.
Cerebrospinal fluid (CSF) CSF was assayed for the soluble membrane attack complex (sMAC) by enzyme-linked immunosorbent assay (ELISA) in patients with suspected shunt failure or infection. CSF was obtained at the time of initial surgical intervention. Statistical analysis was performed to assess the diagnostic potential of sMAC in pyogenic-infected versus non-infected patients. Soluble MAC was quantitated using the MicroVue complement sC5b-9 Plus enzyme immunoassay (Quidel Corporation, Athens, OH, USA). The lower limit of detection for the assay was 3.7 ng/mL.
The team used appropriately adjusted cutoff values for maximum sensitivity and specificity, and the sMAC was able to detect 14 of 15 infections in the 248 patients, while the current diagnostic gold standard of bacterial culture was less accurate, detecting only 11 of the 15 infections. At the best cutoff value, the test had excellent diagnostic capability with a sensitivity of 93% and a specificity of 86%. Children with pyogenic shunt infection had significantly increased sMAC levels compared with non-infected patients (3,211 ± 1,111 ng/mL versus 26 ± 3.8 ng/mL).
In infected patients undergoing serial CSF draws, sMAC levels were prognostic for both positive and negative clinical outcomes. Children with delayed, broth-only growth of commensal organisms such as Propionibacterium acnes, Staphylococcus epidermidis had the lowest sMAC levels (7.96 ± 1.7 ng/mL), suggesting contamination rather than shunt infection. The study was first published online on July 7, 2016, in the Journal of Clinical Investigation Insight.
Related Links:
University of Alabama
Quidel
The potential life-threatening nature of shunt infections requires rapid diagnosis; however, traditional microbiology is time consuming, expensive, and potentially unreliable. A quick and accurate test for bacterial infection in brain shunts or meningitis would improve the quality and efficiency of patient care and patient outcomes. Such a test would also lower health care expenses by avoiding needless hospitalization and treatment.
Scientists at the University of Alabama (Birmingham, AL, USA) prospectively enrolled 198 consecutive undergoing evaluation and treatment for newly diagnosed hydrocephalus, shunt infection, and versus malfunction. Study samples were collected at the same time as routine lab specimens to provide corresponding laboratory results for each specimen (glucose, protein, hematology indices, gram stain, and culture). Samples were labeled and stored at –20 °C and/or –80 °C for a period of one week on average prior to analysis.
Cerebrospinal fluid (CSF) CSF was assayed for the soluble membrane attack complex (sMAC) by enzyme-linked immunosorbent assay (ELISA) in patients with suspected shunt failure or infection. CSF was obtained at the time of initial surgical intervention. Statistical analysis was performed to assess the diagnostic potential of sMAC in pyogenic-infected versus non-infected patients. Soluble MAC was quantitated using the MicroVue complement sC5b-9 Plus enzyme immunoassay (Quidel Corporation, Athens, OH, USA). The lower limit of detection for the assay was 3.7 ng/mL.
The team used appropriately adjusted cutoff values for maximum sensitivity and specificity, and the sMAC was able to detect 14 of 15 infections in the 248 patients, while the current diagnostic gold standard of bacterial culture was less accurate, detecting only 11 of the 15 infections. At the best cutoff value, the test had excellent diagnostic capability with a sensitivity of 93% and a specificity of 86%. Children with pyogenic shunt infection had significantly increased sMAC levels compared with non-infected patients (3,211 ± 1,111 ng/mL versus 26 ± 3.8 ng/mL).
In infected patients undergoing serial CSF draws, sMAC levels were prognostic for both positive and negative clinical outcomes. Children with delayed, broth-only growth of commensal organisms such as Propionibacterium acnes, Staphylococcus epidermidis had the lowest sMAC levels (7.96 ± 1.7 ng/mL), suggesting contamination rather than shunt infection. The study was first published online on July 7, 2016, in the Journal of Clinical Investigation Insight.
Related Links:
University of Alabama
Quidel
Latest Immunology News
- Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
- Study Points to Autoimmune Pathway Behind Long COVID Symptoms
- Immune Enzyme Linked to Treatment-Resistant Inflammatory Bowel Disease
- Simple Blood Test Could Replace Biopsies for Lung Transplant Rejection Monitoring
- Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
- Biomarkers and Molecular Testing Advance Precision Allergy Care
- Point-of-Care Tests Could Expand Access to Mpox Diagnosis
- T-Cell Senescence Profiling May Predict CAR T Responses
- Finger-Prick Lateral Flow Test Detects Sepsis Biomarkers at Point of Care
- Study Highlights Low Sensitivity of Current Lyme Tests in Early Infection
- Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
- Study Finds Influenza Often Undiagnosed in Winter Deaths
- Combined Screening Approach Identifies Early Leprosy Cases
- Antibody Blood Test Identifies Active TB and Distinguishes Latent Infection
- FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
- Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Channels
Clinical Chemistry
view channel
Urine-Based Test Shows Promise for Autism Screening in Children
Autism spectrum disorder (ASD) is commonly diagnosed through behavioral assessments, which can involve long waits that delay intervention. Earlier identification is linked to better developmental outcomes,... Read more
Liquid Biopsy Biomarkers May Improve Childhood Epilepsy Diagnosis
Childhood epilepsy remains a major neurological disorder with unmet needs for accurate, non-invasive biomarkers, as conventional tests such as electroencephalography and neuroimaging can have limited sensitivity... Read moreMolecular Diagnostics
view channel
Blood-Based MRD Monitoring Supports Relapse Prevention in Leukemia
In myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML), molecular blood testing for minimal residual disease (MRD) can detect signs of impending relapse before clinical symptoms emerge.... Read more
Genomic Test Predicts Chemotherapy Benefit in Metastatic Prostate Cancer
Metastatic prostate cancer remains a major treatment challenge, particularly when deciding whether to add chemotherapy to hormonal therapy. In the United States, about 334,000 men are diagnosed with prostate... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channel
Study Points to Autoimmune Pathway Behind Long COVID Symptoms
Long COVID leaves many SARS-CoV-2 survivors with persistent fatigue, cognitive issues, palpitations, and musculoskeletal pain for months or years. Estimates cited in new research suggest 4%–20% of infected... Read more
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read morePathology
view channel
AI-Powered Atlas Maps Immune Structures Linked to Cancer Outcomes
Tertiary lymphoid structures are emerging as important indicators of antitumor immunity, but their heterogeneity and spatial context within tumors remain difficult to capture through routine diagnostics.... Read more
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channel