Novel Test May Enable Quick Reliable Detection of Sepsis
|
By LabMedica International staff writers Posted on 20 Jun 2016 |
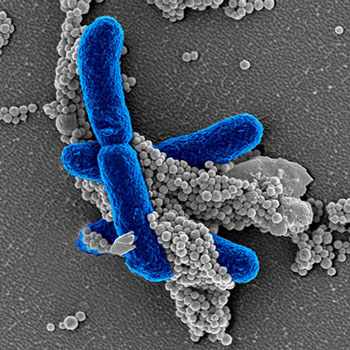
Image: In the new pathogen-detection technology, engineered FcMBL proteins coupled to magnetic beads (grey) specifically bind to carbohydrate molecules on the surface of pathogens, like infectious E. coli (blue) in this electron micrograph, or on fragments of dead pathogens circulating in the bloodstream. After isolation in a magnetic field, the total pathogenic material is quantified with a second FcMBL protein linked to a color-producing enzyme (Photo courtesy of Wyss Institute at Harvard University).
Researchers have developed a rapid specific diagnostic assay that could help physicians decide within an hour whether a patient has a systemic infection. Its potential to detect pathogen materials was demonstrated in animal studies and a prospective human clinical study, whose results also suggested that it could also serve as a companion diagnostic to monitor antibiotic and dialysis-like sepsis therapies.
The current standard-of-care for detecting blood-borne infections is blood culture, but this takes days, only identifies pathogens in less than 30% of patients with fulminant infections, and is not able to detect toxic fragments of dead pathogens that also drive the exaggerated inflammatory reactions leading to sepsis. Biomarkers that report elevated inflammation are used to monitor treatment of patients with sepsis, but they fail to distinguish inflammation triggered by infectious pathogens from that induced by non-infectious causes (e.g. burns, traumas, surgeries).
The assay was developed and tested by a research team from Wyss Institute for Biologically Inspired Engineering at Harvard University (Boston, MA, USA) led by Donald Ingber, MD, PhD, Wyss Institute’s founding director, and professor at Harvard Medical School and Boston Children’s Hospital: "Our pathogen detection technology solves both dilemmas: it quickly reports whether infectious pathogens are present in the body, even at early stages of infection before sepsis develops; And it can more specifically identify patients who have excessive inflammation due to systemic infection, rather than other causes," said Prof. Ingber, "This assay could become a real game changer in this clinical area, and it also should lead to more judicious use of antibiotics."
"In a cohort of emergency room patients with suspected sepsis, we saw that the assay picked up infection within an hour in 85% of patients who exhibited clinical symptoms of sepsis, and equally importantly it did not falsely predict infection in healthy subjects or patients with inflammation triggered by other causes, such as trauma. On the other hand, blood cultures that we performed in parallel using the same samples only detected pathogens in 18% of the cases," said Nathan Shapiro, MD, PhD, director, Translational Research, Center for Vascular Biology Research at BIDMC, who worked with the team.
The assay technology is based on magnetic beads to FcMBL, a genetically engineered pathogen-binding protein previously developed by Prof. Ingber and Michael Super, a Wyss senior staff scientist who co-leads the pathogen-detection effort. By recognizing surface carbohydrate molecules, FcMBL binds to pathogens and pathogen-released fragments – pathogen-associated molecular patterns (PAMPs). The team previously established FcMBL as a key component of an advanced dialysis-like, pathogen-extracting therapeutic device, and of a method for fast retrieval of infectious pathogens from complex clinical samples to enable identification and antibody susceptibilities.
"In our latest work, we show that the FcMBL-based pathogen-detecting assay is considerably faster and more accurate than any other available assay for systemic infection. We are currently working to ready it for high-throughput use in clinical and point-of-care situations," said co-lead-author Mark Cartwright, PhD, staff scientist at Wyss.
As a prerequisite to their clinical study, the team had successfully tested the assay in rat and pig models infected with pathogenic E. coli. "The animal models clearly told us that the assay can sensitively trace spikes of PAMPs released during antibiotic therapy, or residual infectious PAMP materials, even when no living bacteria circulate anymore in blood but they remain hidden inside internal organs. Thus, this assay could be an excellent tool for monitoring ongoing infection and responses to antibiotics and dialysis-like therapies for severe infections and sepsis," said Mike Super, PhD.
The findings suggest that this technology, with its rapid handling time, high sensitivity and broad specificity, could provide a real advance for diagnosing infections in clinical microbiology laboratories and point-of-care settings.
The study, by Cartwright M et al, was published online June 12, 2016, in the journal eBioMedicine.
Related Links:
Wyss Institute
The current standard-of-care for detecting blood-borne infections is blood culture, but this takes days, only identifies pathogens in less than 30% of patients with fulminant infections, and is not able to detect toxic fragments of dead pathogens that also drive the exaggerated inflammatory reactions leading to sepsis. Biomarkers that report elevated inflammation are used to monitor treatment of patients with sepsis, but they fail to distinguish inflammation triggered by infectious pathogens from that induced by non-infectious causes (e.g. burns, traumas, surgeries).
The assay was developed and tested by a research team from Wyss Institute for Biologically Inspired Engineering at Harvard University (Boston, MA, USA) led by Donald Ingber, MD, PhD, Wyss Institute’s founding director, and professor at Harvard Medical School and Boston Children’s Hospital: "Our pathogen detection technology solves both dilemmas: it quickly reports whether infectious pathogens are present in the body, even at early stages of infection before sepsis develops; And it can more specifically identify patients who have excessive inflammation due to systemic infection, rather than other causes," said Prof. Ingber, "This assay could become a real game changer in this clinical area, and it also should lead to more judicious use of antibiotics."
"In a cohort of emergency room patients with suspected sepsis, we saw that the assay picked up infection within an hour in 85% of patients who exhibited clinical symptoms of sepsis, and equally importantly it did not falsely predict infection in healthy subjects or patients with inflammation triggered by other causes, such as trauma. On the other hand, blood cultures that we performed in parallel using the same samples only detected pathogens in 18% of the cases," said Nathan Shapiro, MD, PhD, director, Translational Research, Center for Vascular Biology Research at BIDMC, who worked with the team.
The assay technology is based on magnetic beads to FcMBL, a genetically engineered pathogen-binding protein previously developed by Prof. Ingber and Michael Super, a Wyss senior staff scientist who co-leads the pathogen-detection effort. By recognizing surface carbohydrate molecules, FcMBL binds to pathogens and pathogen-released fragments – pathogen-associated molecular patterns (PAMPs). The team previously established FcMBL as a key component of an advanced dialysis-like, pathogen-extracting therapeutic device, and of a method for fast retrieval of infectious pathogens from complex clinical samples to enable identification and antibody susceptibilities.
"In our latest work, we show that the FcMBL-based pathogen-detecting assay is considerably faster and more accurate than any other available assay for systemic infection. We are currently working to ready it for high-throughput use in clinical and point-of-care situations," said co-lead-author Mark Cartwright, PhD, staff scientist at Wyss.
As a prerequisite to their clinical study, the team had successfully tested the assay in rat and pig models infected with pathogenic E. coli. "The animal models clearly told us that the assay can sensitively trace spikes of PAMPs released during antibiotic therapy, or residual infectious PAMP materials, even when no living bacteria circulate anymore in blood but they remain hidden inside internal organs. Thus, this assay could be an excellent tool for monitoring ongoing infection and responses to antibiotics and dialysis-like therapies for severe infections and sepsis," said Mike Super, PhD.
The findings suggest that this technology, with its rapid handling time, high sensitivity and broad specificity, could provide a real advance for diagnosing infections in clinical microbiology laboratories and point-of-care settings.
The study, by Cartwright M et al, was published online June 12, 2016, in the journal eBioMedicine.
Related Links:
Wyss Institute
Latest Microbiology News
- FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
- New AMR Assay Supports Rapid Infection Control Screening in Hospitals
- Diagnostic Gaps Complicate Bundibugyo Ebola Outbreak Response in Congo
- Study Finds Hidden Mpox Infections May Drive Ongoing Spread
- Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
- Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
- Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
- Label-Free Microscopy Method Enables Faster, Quantitative Detection of Malaria
- Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
- RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
Channels
Clinical Chemistry
view channel
Fluid Biomarker Improves Diagnosis and Monitoring of Primary CNS Lymphoma
Primary central nervous system lymphoma (PCNSL) is a rare malignancy of the brain, spinal cord, and eyes with delayed diagnosis and poor outcomes. Current fluid-based testing using interleukin measurements... Read more
New CA19-9 Cutoff Value Helps Identify High-Risk Pancreatic Cancer Patients
Pancreatic ductal adenocarcinoma (PDAC) is frequently diagnosed at an advanced stage and remains one of the most lethal solid tumors. Clinicians commonly use serum carbohydrate antigen 19-9 (CA19-9) to... Read moreMolecular Diagnostics
view channel
Gene Fusion Patterns May Flag High Risk Solitary Fibrous Tumors
Solitary fibrous tumor (SFT) is a rare subtype of sarcoma, a cancer that develops in connective tissues such as fat, muscle, blood vessels, and fibrous tissue. SFTs most commonly arise in the chest cavity... Read more
New RNA Origami Method Supports Faster Targeted Testing for Repeat Expansion Disorders
Repeat expansion disorders drive conditions such as myotonic dystrophy, Huntington’s disease, and amyotrophic lateral sclerosis (ALS), yet accurately sizing the mutated sequences remains difficult.... Read moreHematology
view channel
Higher Ferritin Threshold May Improve Iron Deficiency Detection in Children
Iron deficiency in school-age children can affect brain development, learning, growth, and physical performance, yet early deficiency may be missed when screening focuses mainly on anemia.... Read more
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read moreImmunology
view channel
Immune Enzyme Linked to Treatment-Resistant Inflammatory Bowel Disease
Inflammatory bowel disease (IBD) affects nearly 3 million people in the United States and its prevalence continues to rise. Medications that target tumor necrosis factor (TNF)-alpha are widely used, but... Read more
Simple Blood Test Could Replace Biopsies for Lung Transplant Rejection Monitoring
Lung transplant recipients face some of the highest rates of acute cellular rejection, and routine surveillance often relies on repeated surgical biopsies. These procedures can cause complications such... Read morePathology
view channel
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read more
Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
Gene expression profiling can inform tumor biology and treatment selection, but spatial assays remain costly and time-consuming. Results can take weeks and cost thousands of dollars, limiting large-scale... Read moreTechnology
view channel
AI-Enabled Assistant Unifies Molecular Workflow Planning and Support
Clinical laboratories and research groups face increasingly complex molecular workflows and expanding technical documentation spread across multiple systems. Fragmented digital tools can slow experiment... Read more
AI Tool Automates Validation of Laboratory Software Configuration Changes
Regulated laboratories face heavy documentation and requalification demands when software configurations change, slowing improvements and discouraging beneficial updates. A new capability now automates... Read moreIndustry
view channel
Natera to Present Data on MRD-Guided Cancer Care at ASCO 2026
Natera, Inc. (Austin, TX, USA), a company focused on cell-free DNA testing and precision medicine, announced an oncology data program for the 2026 American Society of Clinical Oncology (ASCO) Annual Meeting,... Read more