Rosetta and Moffitt Partner to Develop microRNA Cancer Diagnostic
|
By LabMedica International staff writers Posted on 16 Jun 2014 |
Rosetta Genomics, Ltd. (Princeton, NJ, USA; Rehovot, Israel), a leading developer and provider of microRNA-based molecular diagnostics and therapeutics, has entered into an agreement with Moffitt Cancer Center (Tampa, FL, USA) to advance the development and commercialization of Rosetta's thyroid neoplasia assay. Rosetta will work with Marino Leon, MD, associate member of the department of anatomic pathology at Moffitt.
Kenneth A. Berlin, president and CEO of Rosetta Genomics commented, “Results from our initial studies demonstrated that microRNA expression levels can differentiate malignant nodules from benign nodules, and also demonstrated our ability to extract and profile microRNAs from thyroid fine-needle aspirations [FNAs] in various sample types and in a way consistent with common clinical practices. This agreement with Moffitt will help us further advance the development of this important assay, and should allow us to achieve our goal of launching this important assay prior to the end of 2015.”
Dr. Marino Leon noted, “Many patients with indeterminate thyroid FNA results are sent to surgery as a precaution, despite the fact that the majority of these cases are benign. This exposes patients to unnecessary surgical risk and costs the system hundreds of millions of dollars.”
Rosetta Genomics cancer tests are a series of microRNA-based diagnostic testing services. The Rosetta Cancer Origin test can accurately identify the primary tumor type in primary and metastatic cancer including cancer of unknown or uncertain primary (CUP). The company’s assays are offered directly by Rosetta Genomics in the United States, and through distributors around the world. Rosetta’s cancer testing services are commercially available through its Philadelphia (PA, USA)-based College of American Pathologists (CAP)-accredited, Clinical Laboratory Improvement Amendments (CLIA)-certified lab.
Rosetta’s research platform combining bioinformatics and state-of-the-art laboratory processes has led to the discovery of hundreds of biologically validated novel human microRNAs. Rosetta is working on the application of these technologies in the development and commercialization of a full range of microRNA-based diagnostic tools and therapeutics.
Moffitt is one of only 41 US National Cancer Institute-designated comprehensive cancer centers, a distinction that recognizes Moffitt's excellence in research, its contributions to clinical trials, prevention and cancer control. Moffitt is the No. 1 cancer hospital in Florida and has been listed in US News & World Report as one of “America's Best Hospitals” for cancer since 1999.
Related Links:
Rosetta Genomics
Moffitt Cancer Center
Kenneth A. Berlin, president and CEO of Rosetta Genomics commented, “Results from our initial studies demonstrated that microRNA expression levels can differentiate malignant nodules from benign nodules, and also demonstrated our ability to extract and profile microRNAs from thyroid fine-needle aspirations [FNAs] in various sample types and in a way consistent with common clinical practices. This agreement with Moffitt will help us further advance the development of this important assay, and should allow us to achieve our goal of launching this important assay prior to the end of 2015.”
Dr. Marino Leon noted, “Many patients with indeterminate thyroid FNA results are sent to surgery as a precaution, despite the fact that the majority of these cases are benign. This exposes patients to unnecessary surgical risk and costs the system hundreds of millions of dollars.”
Rosetta Genomics cancer tests are a series of microRNA-based diagnostic testing services. The Rosetta Cancer Origin test can accurately identify the primary tumor type in primary and metastatic cancer including cancer of unknown or uncertain primary (CUP). The company’s assays are offered directly by Rosetta Genomics in the United States, and through distributors around the world. Rosetta’s cancer testing services are commercially available through its Philadelphia (PA, USA)-based College of American Pathologists (CAP)-accredited, Clinical Laboratory Improvement Amendments (CLIA)-certified lab.
Rosetta’s research platform combining bioinformatics and state-of-the-art laboratory processes has led to the discovery of hundreds of biologically validated novel human microRNAs. Rosetta is working on the application of these technologies in the development and commercialization of a full range of microRNA-based diagnostic tools and therapeutics.
Moffitt is one of only 41 US National Cancer Institute-designated comprehensive cancer centers, a distinction that recognizes Moffitt's excellence in research, its contributions to clinical trials, prevention and cancer control. Moffitt is the No. 1 cancer hospital in Florida and has been listed in US News & World Report as one of “America's Best Hospitals” for cancer since 1999.
Related Links:
Rosetta Genomics
Moffitt Cancer Center
Latest Industry News
- Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
- FDA Clears Blood-Based Companion Diagnostic for Advanced Breast Cancer Therapy
- CareDx Expands Precision Oncology Portfolio with Naveris Acquisition
- Thermo Fisher Scientific to Sell Microbiology Business to Astorg
- Collaboration Expands Access to Rapid Metagenomic Diagnostics for Complex Infections
- Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
- QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
- QIAGEN Showcases Integrated Sample-to-Insight Oncology Solutions at AACR 2026
- Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
- Partnership Expands Ultrasensitive WGS Assay for for Hematologic Malignancies and MRD Monitoring
- Beckman Coulter Gains CE Mark for Rapid Assay Distinguishing Bacterial vs Viral Infections
- Junction, GRAIL Enable Scalable Access to Multi-Cancer Detection Test
- GRAIL Partners with Epic to Integrate Multi-Cancer Test into EHR
- Global Partnership Aims to Streamline NGS Tumor Profiling in Oncology Trials
- Takara Bio USA and Hamilton Partner to Automate NGS Library Preparation
- Lunit and CellCarta Collaborate to Expand AI Pathology in CDx Development
Channels
Clinical Chemistry
view channel
FDA-Cleared Assay Enables Comprehensive Automated Testosterone Testing
Accurate evaluation of androgen status often requires concordant measurement of total testosterone, free testosterone, and sex hormone‑binding globulin. Reference methods such as equilibrium dialysis with... Read more
CE-Marked Blood Biomarker Test Advances Automated Alzheimer’s Diagnostics
Specialized care settings frequently evaluate patients aged 50 years and older who present with signs and symptoms of cognitive decline to determine whether amyloid pathology linked to Alzheimer’s disease... Read moreMolecular Diagnostics
view channel
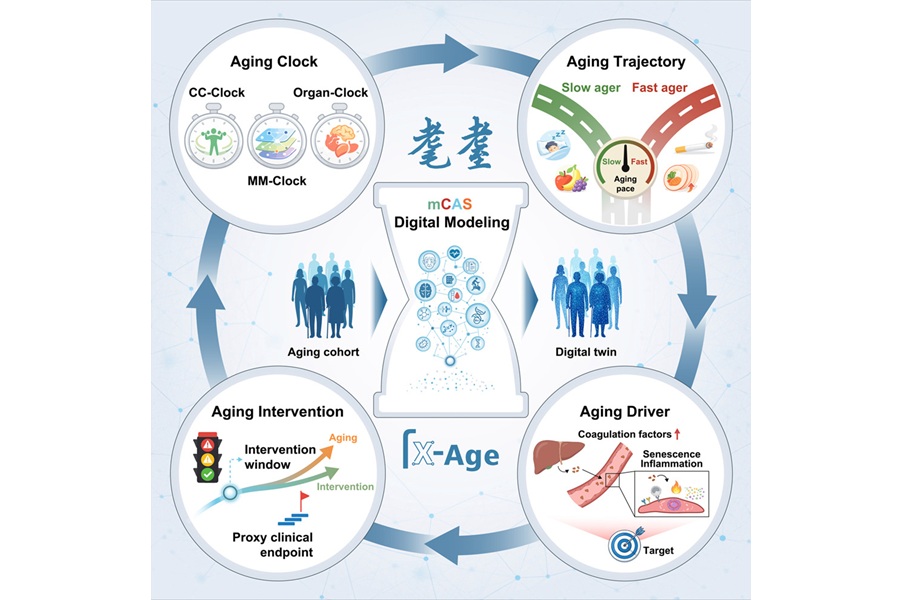
Blood-Based “Ageing Clock” Helps Predict Dementia Risk and Earlier Onset
Dementia imposes a growing health burden, affecting an estimated 982,000 people in the UK, with cases projected to reach 1.4 million by 2040. Earlier identification of those most likely to develop disease... Read more
Blood Test Refines Biopsy Decisions in Prostate Cancer
Prostate biopsy decisions remain challenging, with many men undergoing invasive procedures that ultimately yield negative results. In the U.S., more than a million prostate biopsies are performed each... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read more
Point-of-Care Tests Could Expand Access to Mpox Diagnosis
Mpox outbreaks in non-endemic regions have underscored the need for rapid, accessible diagnostics to limit transmission. Polymerase chain reaction (PCR) remains the clinical reference, yet it depends on... Read moreMicrobiology
view channel
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read more
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read more