Blood Culture Diagnostic Techniques Compared for Human Brucellosis
|
By LabMedica International staff writers Posted on 16 Apr 2014 |
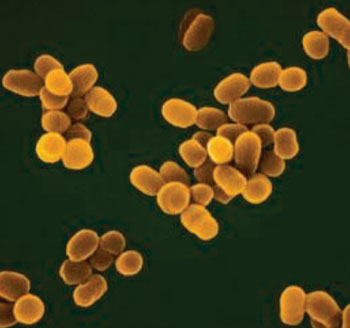
Image: Scanning electron micrograph of Brucella abortus (Photo courtesy of the Czech University of Life Science).
Isolation of the bacteria Brucella is the gold standard in the laboratory diagnosis of brucellosis and as the organism is intracellular, the number of circulating bacteria is usually low.
Three different blood culture methods, the lysis concentration (LC), clot culture and conventional Castaneda blood culture techniques, have been compared for the isolation rate and recovery time in the diagnosis of human brucellosis.
Microbiologists at the Shri B. M. Patil Medical College (Bijapur, Karnataka, India) performed blood cultures by LC, clot culture and conventional method in 169 patients who had antibody titers equal to or greater than 160 international units by the serum agglutination test (SAT).
For the conventional culture technique, the blood specimen was inoculated aseptically into the broth phase of Castaneda's biphasic medium consisting of brain heart infusion agar and broth with Brucella selective supplement (Hi-Media; Mumbai, India). For the LC technique, the mixtures centrifuged and the supernatant was discarded and the sediment was inoculated in the Castaneda's medium, instead of culture plate.
For the clot culture technique, the blood clot was preserved in the sterile screw capped plastic tube with glass beads after removal of serum was used for this method. The clot was disrupted by shaking the tubes on a shaker for 15 minutes and the disrupted clot was then inoculated in Castaneda's medium. The media were incubated at 37 °C with 10% carbon dioxide for a maximum of 30 days. Provisional confirmation and biotyping of the isolate was done by performing slide agglutination test using B. abortus and B. melitensis monospecific antisera (Murex Biotech; Dartford, UK).
Overall blood culture positivity was found to be 24.8% by conventional culture, 34.9% by clot method, and 43.1% by the lysis technique. The mean recovery time by lysis and clot culture techniques was significantly less than conventional method, resulting in an overall difference of nearly six and four days respectively.
The authors concluded that for the isolation of Brucella from blood specimen, LC method is better than conventional Castaneda's method as the isolation rate is high and the recovery time is less. Clot culture is a better option when a second blood sample cannot be obtained for culture. As lysis and clot culture techniques are sensitive, simple and inexpensive and yield earlier results, they can be adapted in the technically and economically deprived areas where automated systems are not feasible. The study was published on March 19, 2014, in the Journal of Laboratory Physicians.
Related Links:
Shri B. M. Patil Medical College
Hi-Media
Murex Biotech
Three different blood culture methods, the lysis concentration (LC), clot culture and conventional Castaneda blood culture techniques, have been compared for the isolation rate and recovery time in the diagnosis of human brucellosis.
Microbiologists at the Shri B. M. Patil Medical College (Bijapur, Karnataka, India) performed blood cultures by LC, clot culture and conventional method in 169 patients who had antibody titers equal to or greater than 160 international units by the serum agglutination test (SAT).
For the conventional culture technique, the blood specimen was inoculated aseptically into the broth phase of Castaneda's biphasic medium consisting of brain heart infusion agar and broth with Brucella selective supplement (Hi-Media; Mumbai, India). For the LC technique, the mixtures centrifuged and the supernatant was discarded and the sediment was inoculated in the Castaneda's medium, instead of culture plate.
For the clot culture technique, the blood clot was preserved in the sterile screw capped plastic tube with glass beads after removal of serum was used for this method. The clot was disrupted by shaking the tubes on a shaker for 15 minutes and the disrupted clot was then inoculated in Castaneda's medium. The media were incubated at 37 °C with 10% carbon dioxide for a maximum of 30 days. Provisional confirmation and biotyping of the isolate was done by performing slide agglutination test using B. abortus and B. melitensis monospecific antisera (Murex Biotech; Dartford, UK).
Overall blood culture positivity was found to be 24.8% by conventional culture, 34.9% by clot method, and 43.1% by the lysis technique. The mean recovery time by lysis and clot culture techniques was significantly less than conventional method, resulting in an overall difference of nearly six and four days respectively.
The authors concluded that for the isolation of Brucella from blood specimen, LC method is better than conventional Castaneda's method as the isolation rate is high and the recovery time is less. Clot culture is a better option when a second blood sample cannot be obtained for culture. As lysis and clot culture techniques are sensitive, simple and inexpensive and yield earlier results, they can be adapted in the technically and economically deprived areas where automated systems are not feasible. The study was published on March 19, 2014, in the Journal of Laboratory Physicians.
Related Links:
Shri B. M. Patil Medical College
Hi-Media
Murex Biotech
Latest Microbiology News
- Automated Blood Culture System Speeds Detection of Bloodstream Infections
- New Culture Medium Speeds C. difficile Resistance Detection and Reduces Costs
- Gut Microbiome Signatures Help Identify Risk of IBD Progression
- FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
- New AMR Assay Supports Rapid Infection Control Screening in Hospitals
- Diagnostic Gaps Complicate Bundibugyo Ebola Outbreak Response in Congo
- Study Finds Hidden Mpox Infections May Drive Ongoing Spread
- Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
- Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
- Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
- Label-Free Microscopy Method Enables Faster, Quantitative Detection of Malaria
- Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
Channels
Clinical Chemistry
view channel
Urinary Biomarker Assay Predicts Kidney Disease Progression Beyond Standard Measures
Many patients with type 2 diabetes and chronic kidney disease continue to experience progressive renal decline, yet conventional markers such as albuminuria and estimated glomerular filtration rate (eGFR)... Read more
Saliva-Based Test Detects Biochemical Signs of Sleep Loss
Acute sleep loss impairs cognition and motor skills, raising safety risks that resemble alcohol intoxication. Clinicians currently lack an objective biochemical test to determine when someone is dangerously... Read more
Simple Dual-Tau Blood Test Detects and Stages Alzheimer’s Disease
Alzheimer’s disease is typically confirmed and staged with positron emission tomography scans and cerebrospinal fluid testing, procedures that are costly and invasive. Broader access to minimally invasive... Read more
Alzheimer’s Blood Biomarkers Linked to Early Cognitive Differences Before Dementia
Blood-based screening for Alzheimer’s disease offers a noninvasive, lower-cost alternative to brain imaging or spinal fluid testing, yet its ability to flag the earliest cognitive changes has been unclear.... Read moreMolecular Diagnostics
view channel
Blood-Based RNA Test May Predict Chemotherapy Sensitivity in Lung Cancer
Lung cancer care increasingly relies on biomarker-guided patient stratification, but tissue biopsy can be impractical and treatment selection remains difficult for many patients. Blood-based assays that... Read more
Blood Test Predicts Immunotherapy Response in Head and Neck Cancer
Head and neck squamous cell carcinoma affects hundreds of thousands of people worldwide each year, yet response rates to immunotherapy remain low. Clinicians lack reliable, minimally invasive tools to... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channelAptamer-Based Biosensor Enables Mutation-Resilient SARS-CoV-2 Detection
Rapid evolution of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can undermine existing molecular diagnostics, especially when assays target small viral components. Double-antibody sandwich... Read more
Study Points to Autoimmune Pathway Behind Long COVID Symptoms
Long COVID leaves many SARS-CoV-2 survivors with persistent fatigue, cognitive issues, palpitations, and musculoskeletal pain for months or years. Estimates cited in new research suggest 4%–20% of infected... Read more
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read morePathology
view channel
AI Pathology Tool Predicts Meningioma Recurrence from Routine Slides
Meningiomas are the most common primary brain tumors in adults, yet their course ranges from indolent to highly recurrent disease. Estimating an individual patient’s recurrence risk often requires advanced... Read more
3D Spatial Multi-Omics Maps Intra-Tumor Diversity in Colorectal Cancer
Colorectal cancer remains a leading cause of cancer death, and clinical decision-making is complicated by marked intra-tumor heterogeneity. Conventional bulk sequencing averages molecular signals across... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channel
Genetic Testing Program Expands Detection of Alpha-1 Antitrypsin Deficiency
Alpha-1 Antitrypsin Deficiency (AATD) is a progressive genetic condition, the leading known genetic risk factor for chronic obstructive pulmonary disease (COPD), and a cause of liver disease in both children... Read more





















