Immunoassay Detects Antibodies to American Trypanosomiasis
|
By LabMedica International staff writers Posted on 11 Jan 2011 |
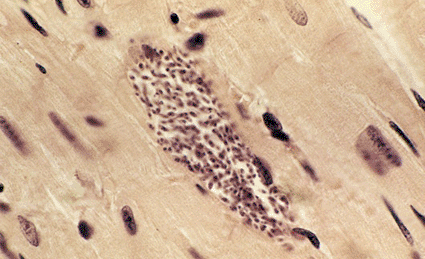
Image: Light micrograph of a section through a cluster of Trypanosoma cruzi protozoans (purple, center) in the heart muscle of a patient suffering from Chagas disease (Photo courtesy of Sinclair Stammers / Science Photo Library).
A fully automated immunoassay can determine the presence of antibodies to Trypanosoma cruzi, the causative agent of Chagas disease, in human serum and plasma.
The quantitative two-step chemiluminescent microparticle immunoassay (CMIA) has been compared to an enzyme linked immunoassays (ELISA) for specificity and sensitivity. In the first step, sample, assay diluent, and paramagnetic microparticles co-coated with four chimeric T. cruzi recombinant antigens, are combined. A murine acridinium-labeled antihuman immunoglobulin (IgG) conjugate is added. After another wash cycle, pre-trigger and trigger solutions are added to the reaction mixture.
The ARCHITECT Chagas prototype assay (Abbott GmbH & Co.; Wiesbaden-Delkenheim, Germany) was tested on total of 10, 284 specimens to determine the specificity and sensitivity of the assay. The tests were done in Abbott's laboratory in Germany and in external sites in Guatemala and Brazil.
Of 10,284 specimens tested, 9,629 had a final interpretation of T. cruzi antibody-negative based on the testing algorithm used, whereas 655 specimens were determined to be T. cruzi antibody-positive. In comparison with the ELISA cruzi assay (bioMérieux; Marcy l'Etoile, France; www.biomerieux.com), the ARCHITECT assay demonstrated superior specificity (99.99% vs. 99.93%) and sensitivity (99.85% vs. 98.38%), along with excellent precision. This suggests that this CMIA has the potential to serve as single assay to determine the T. cruzi status of a given blood unit or diagnostic specimen on a fully automated instrument platform.
The authors of the study concluded that the utility of the fully automated ARCHITECT Chagas assay can be used in the diagnostic setting and for blood donor testing in endemic as well as nonendemic regions. The assay, once it is commercially available, might be used as a single test for routine testing in high-prevalence areas such as South and Central America, which would enable an economic testing strategy being highly advantageous for the target countries. Chagas disease or American Trypanosomiasis represents the third largest tropical disease burden worldwide, after malaria and schistosomiasis. The study was published in January 2011, in Diagnostic Microbiology and Infectious Disease.
Related Links:
bioMérieux
Abbott
The quantitative two-step chemiluminescent microparticle immunoassay (CMIA) has been compared to an enzyme linked immunoassays (ELISA) for specificity and sensitivity. In the first step, sample, assay diluent, and paramagnetic microparticles co-coated with four chimeric T. cruzi recombinant antigens, are combined. A murine acridinium-labeled antihuman immunoglobulin (IgG) conjugate is added. After another wash cycle, pre-trigger and trigger solutions are added to the reaction mixture.
The ARCHITECT Chagas prototype assay (Abbott GmbH & Co.; Wiesbaden-Delkenheim, Germany) was tested on total of 10, 284 specimens to determine the specificity and sensitivity of the assay. The tests were done in Abbott's laboratory in Germany and in external sites in Guatemala and Brazil.
Of 10,284 specimens tested, 9,629 had a final interpretation of T. cruzi antibody-negative based on the testing algorithm used, whereas 655 specimens were determined to be T. cruzi antibody-positive. In comparison with the ELISA cruzi assay (bioMérieux; Marcy l'Etoile, France; www.biomerieux.com), the ARCHITECT assay demonstrated superior specificity (99.99% vs. 99.93%) and sensitivity (99.85% vs. 98.38%), along with excellent precision. This suggests that this CMIA has the potential to serve as single assay to determine the T. cruzi status of a given blood unit or diagnostic specimen on a fully automated instrument platform.
The authors of the study concluded that the utility of the fully automated ARCHITECT Chagas assay can be used in the diagnostic setting and for blood donor testing in endemic as well as nonendemic regions. The assay, once it is commercially available, might be used as a single test for routine testing in high-prevalence areas such as South and Central America, which would enable an economic testing strategy being highly advantageous for the target countries. Chagas disease or American Trypanosomiasis represents the third largest tropical disease burden worldwide, after malaria and schistosomiasis. The study was published in January 2011, in Diagnostic Microbiology and Infectious Disease.
Related Links:
bioMérieux
Abbott
Latest Hematology News
- Advanced CBC-Derived Indices Integrated into Hematology Platforms
- Blood Test Enables Early Detection of Multiple Myeloma Relapse
- Single Assay Enables Rapid HLA and ABO Genotyping for Transplant Matching
- Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
- Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
- Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
- New Platelet Function Assay Enables Monitoring of Antiplatelet Therapy
- Open Multi-Omics Platform Identifies Prognostic Subtypes in Blood Cancers
- AI-Powered Digital Workflow Standardizes Bone Marrow Aspirate Morphology
- Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
- New Guidelines Aim to Improve AL Amyloidosis Diagnosis
- Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
- Fast and Easy Test Could Revolutionize Blood Transfusions
- High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
- AI Algorithm Effectively Distinguishes Alpha Thalassemia Subtypes
- MRD Tests Could Predict Survival in Leukemia Patients
Channels
Clinical Chemistry
view channel
Ultrasensitive Test Detects Key Biomarker of Frontotemporal Dementia Subtype
Dementia affects more than 57 million people worldwide and is projected to nearly double within two decades, straining health systems and families. While biomarkers now enable accurate identification of... Read more
Routine Blood Tests Years Before Pregnancy Could Identify Preeclampsia Risk
High blood pressure during pregnancy is common and can progress to pre-eclampsia, making close monitoring at antenatal visits essential. However, most risk assessment begins only after pregnancy has started.... Read moreMolecular Diagnostics
view channel
Blood Test Maps Tumor Microenvironment to Predict Immunotherapy Response
Immunotherapy has transformed cancer care, yet durable benefit remains limited to a subset of patients, and clinicians still lack reliable tools to predict response before treatment begins.... Read more
Multiplex Respiratory Panel Integrates Automated Extraction to Streamline High-Volume Testing
Respiratory infections drive heavy testing volumes in clinical laboratories, where accurate, timely results across multiple pathogens are essential. Many labs are seeking to streamline workflows and increase... Read more
Whole-Blood RNA Test Predicts Disease Trajectory and Treatment Response
Clinicians often must predict whether acutely ill patients will recover or deteriorate despite limited time and clinical evidence. Earlier prognostic information could improve triage and guide treatment... Read more
Blood-Based Epigenetic Test Predicts GLP-1 Response and Tracks Treatment Effects
Prescriptions for GLP-1 medicines for weight loss are expanding rapidly, yet clinicians still lack scalable tools to predict biological response before treatment or monitor drug-driven changes beyond the scale.... Read moreImmunology
view channel
Point-of-Care Tests Could Expand Access to Mpox Diagnosis
Mpox outbreaks in non-endemic regions have underscored the need for rapid, accessible diagnostics to limit transmission. Polymerase chain reaction (PCR) remains the clinical reference, yet it depends on... Read more
T-Cell Senescence Profiling May Predict CAR T Responses
Chimeric antigen receptor (CAR) T-cell therapy can deliver striking, durable remissions, yet many patients experience minimal or no benefit. The quality of patient-derived cytotoxic T lymphocytes used... Read moreMicrobiology
view channel
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read more
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Tumor-on-a-Chip Platform Models Pancreatic Cancer Treatment Response
Pancreatic cancer remains one of the hardest malignancies to treat because tumors are embedded within a dense microenvironment that shapes growth and therapy response. Standard laboratory models often... Read more
New Platform Captures Extracellular Vesicles for Early Cancer Detection
Early diagnosis remains the most effective way to reduce cancer mortality, yet many screening tools miss disease at its earliest stages. Biomarkers shed by tumors into blood and other fluids can be scarce... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Expand Digital Pathology
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more