Consensus Guidelines for Thyroid Cancer Molecular Tests Revised
By LabMedica International staff writers
Posted on 21 Jul 2015
National guidelines for thyroid cancer testing have been revised to reflect newly available tests that better incorporate personalized medicine into diagnosing the condition.Posted on 21 Jul 2015
The specific utility of these novel, clinically available molecular tests is becoming widely appreciated, especially in perioperative decision making by the surgeon regarding the need for surgery and the extent of initial resection.
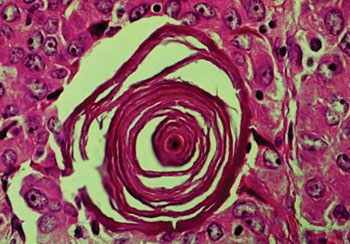
Image: Histopathology of a psammoma body seen in thyroid gland papillary carcinoma (Photo courtesy of The Armed Forces Institute of Pathology).
Scientists at the University of Pittsburgh Schools of the Health Sciences (PA, USA) led a panel of experts in revising national guidelines for thyroid cancer testing to reflect newly available tests that better integrate tailored personal treatment in differentiating thyroid diseases. Standardized interpretation of fine-needle aspiration (FNA) cytology for thyroid nodules has improved since the advent of a tiered Bethesda classification scheme. While concordance among cytopathologists is high (90%) for benign and malignant cytologic diagnoses, there is significant intra- and inter-observer variability for any type of indeterminate cytologic diagnosis.
The gene expression classifier (GEC) and seven-gene molecular panel (7-gene MT) are the two widely available tests at the present time. The Afirma GEC (Veracyte; South San Francisco, CA, USA) was developed as a proprietary method for molecular analysis of thyroid FNA specimens diagnosed as atypia of uncertain significance/follicular lesion of undetermined significance (AUS/FLUS) and follicular neoplasm/suspicious for follicular neoplasm (FN), with the intent of predicting benign pathology and thus avoiding unnecessary thyroidectomy for asymptomatic nodules. With an initially reported high negative predictive value, GEC combines an assay signature of 167 genes with commercial cytologic examination. A low-cost 7-gene MT test called ThyroSeq (CBLPATH; Rye Brook, NY, USA) allows pathologists to simultaneously test for multiple genetic markers of thyroid cancer using just a few cells collected from the nodule.
Other available tests use different technology to serve as accurate “rule-out” tools, but do not have the high sensitivity needed to also reliably “rule-in” cancer. In some cases, the accuracy of the “rule-out” tests depends on the prevalence of cancer in the patients seen by each individual cancer institute. This is critical because clinicians must know this rate at their institution to correctly calculate the accuracy of “rule-out” test results for each patient. The study was published online on May 18, 2015, in the journal Thyroid.
Related Links:
University of Pittsburgh Schools of the Health Sciences
Veracyte
CBLPATH