Solid Media Used to Isolate Leptospira
By LabMedica International staff writers
Posted on 25 Feb 2013
Isolation of Leptospira, from clinical samples and testing of antimicrobial susceptibility are difficult and time-consuming.Posted on 25 Feb 2013
Pathogenic Leptospira are slow-growing Gram-negative spirochetes and a new solid medium that facilitates more-rapid growth of these organisms has been developed
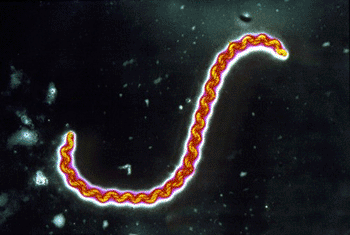
Image: Leptospira interrogans (Photo courtesy of Science Photo Library).
Microbiologists at the Mahidol University (Bangkok, Thailand) developed the new solid medium, called LVW agar, by testing the effects of combinations of ingredients and incubation conditions. All recipes contained three core ingredients and a variable concentration of two additional ingredients. A total of 109 pathogenic Leptospira isolates were used during the development and evaluation of the new solid medium and Etest, which is well-established method for antimicrobial resistance testing, antibiotic susceptibility testing in microbiology laboratories around the world.
The medium was developed by evaluating the effects of numerous factors on the growth rate of a strain of Leptospira interrogans. These included the type of base agar, the concentration of rabbit serum (RS), and the concentration and duration of carbon dioxide (CO2) incubation during the initial period of culture. The highest growth rate of this strain was achieved using a Noble agar base supplemented with 10% RS and named LVW agar, with an initial incubation at 30 °C in 5% CO2 for two days prior to continuous culture in air at 30 °C.
These conditions were used to develop the Etest for three species, L. interrogans, L. kirschnerii, and L. borgpetersenii. The minimum inhibitory concentrations (MICs) were read on day seven for all samples. The validated Etest methodology was performed for 109 Leptospira isolates with five antimicrobial drugs, penicillin G, doxycycline, ceftriaxone, cefotaxime, and chloramphenicol. The MIC90 values were as follows: penicillin G, 0.064 units/mL; doxycycline, 0.19 µg/mL; cefotaxime, 0.047 µg/mL; ceftriaxone, 0.5 µg/mL; and chloramphenicol, 2 µg/mL.
Leptospirosis is a zoonotic disease that has a worldwide distribution but has the greatest impact on health in developing countries, where it is often grossly under recognized. The authors concluded that LVW agar that enables rapid growth, isolation of single colonies, and simple antimicrobial susceptibility testing for Leptospira species will become widely used in diagnostic microbiology laboratories, but especially in resource-limited settings. The study was published in the January 2013 issue of the journal Antimicrobial Agents and Chemotherapy.
Related Links:
Mahidol University














