New Immunoassay Evaluated for Toxoplasmosis
By LabMedica International staff writers
Posted on 25 Apr 2017
The infection by Toxoplasma gondii, an obligate intracellular parasite, is generally benign but can be particularly severe during pregnancy, leading to malformations of the fetus or a series of complications in the newborn.Posted on 25 Apr 2017
Currently, there are several analytical systems for the detection of antibodies, with different level of automation, such as Enzyme-Linked Immunosorbent Assay (ELISA), Chemiluminescence Immunoassay (CLIA) and Enzyme Linked Fluorescent Assay (ELFA) able, depending on the instrument used, to process a large number of samples in a short time.
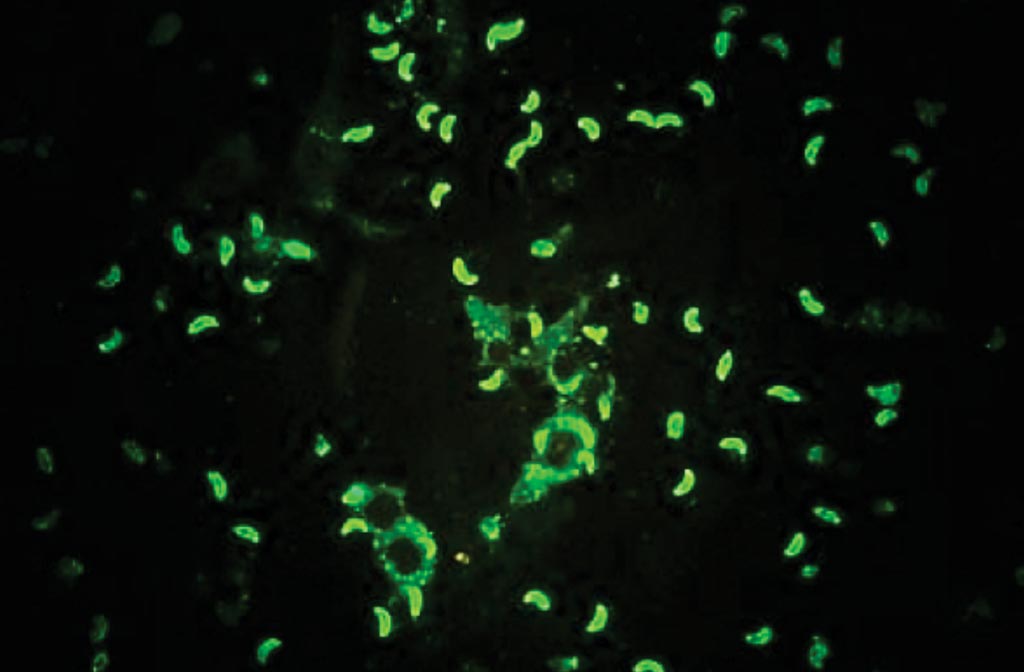
Image: Indirect fluorescent antibody test for Toxoplasma gondii, diffuse staining is considered positive (Photo courtesy of SPL).
Microbiologists at the Legnano Hospital examined 301 sera samples from women who came to their center for the routine follow-up in pregnancy and were divided into three groups: Group 1:106 samples from non-immune women; Group 2:100 samples from women with past infection; and Group 3: 95 samples from women with positive or equivocal immunoglobulin M (IgM) (82 with positive IgG and 13 with negative IgG).
The scientists routinely tested samples using chemiluminescence LIAISON XL system for IgG and IgM. In positive subjects for IgM, samples are tested using an ELFA test. All samples were also tested with TGS TA system for detecting IgG and IgM and, for Group 3, IgG avidity (TGS TA Toxoplasma IgG, Toxoplasma IgM, Toxoplasma IgG Avidity.
The overall concordance of the IgG results between LIAISON XL and TGS TA was 99.3%: 100% in Group 1, 98% in Group 2 and 100% in Group 3. The overall concordance of the IgM results between LIAISON XL and TGS TA was 93.9%: 100% in Group 1, 94% in Group 2 and 82.8% in Group 3. In Group 3, the concordance between the results of the IgG avidity with the ELFA and TGS TA tests was 81.7%. Comparing the clinical diagnosis obtained with the protocol and that of the TGS TA system, the overall concordance was 92.7%: 100% in Group 1, 92.0% in Group 2 and 78.9% in Group 3.
The authors concluded that the overall concordance of IgG antibodies is excellent for both protocols. The TGS TA avidity test seems to predict earlier the maturation of the IgG compared to the ELFA test since many samples with low avidity with the ELFA were seen with moderate avidity with TGS TA and all those with borderline avidity with the ELFA were seen with high avidity with TGS TA. This system is shown to be a valuable tool with overall good clinical correlation and able to clearly identify nonspecific subjects, those with a non-recent infection.














