Clinical Performance Supports New Diagnostic Assay for Multiple Sclerosis
By LabMedica International staff writers
Posted on 03 Feb 2014
New clinical data from a validation study demonstrates positive performance with a novel molecular test developed to facilitate identification of patients with multiple sclerosis.Posted on 03 Feb 2014
DioGenix (Gaithersburg, MD, USA) has announced the new data supporting the clinical validation of "MSPrecise,” its proprietary next-generation sequencing (NGS) assay for the identification of patients with multiple sclerosis (MS) at first clinical presentation. The test performed consistently with previous clinical studies, having a specificity of 82% while maintaining sensitivity comparable to what has been published for the current standard-of-care (p=0.0027).
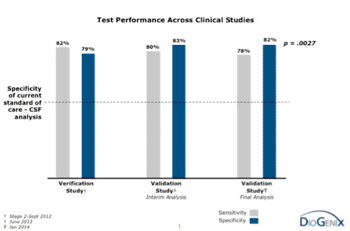
Image: Graph summarizing results from clinical studies of MSPrecise demonstrating positive performance in identifying patients with multiple sclerosis (Photo courtesy of DioGenix).
Over 200 subjects being evaluated for nonspecific neurological symptoms that could be MS, were enrolled in the prospective, blinded clinical trial that evaluated and thus validated the performance of MSPrecise. These subjects were undergoing a comprehensive evaluation using the current standard-of-care for imaging of the central nervous system (CNS) and analysis of their cerebral spinal fluid and blood.
This study compared the results of MSPrecise DNA mutational analysis with a consensus diagnosis made by a panel of independent neurologists chosen for their significant clinical experience in diagnosing and treating MS. The MSPrecise interpretive scoring system provides a simple scaled score to the neurologist who differentiates patients with MS from those with other similarly presenting neurological diseases. Thirteen MS clinical centers of excellence participated in the trial—believed to be the largest prospective diagnostic study of its kind in MS—with over 20 thought-leading clinicians consenting subjects. Results from this study will now be submitted for peer review.
The results are consistent with two prior DioGenix studies that compared MSPrecise to published performance data for the oligoclonal banding (OCB) test and experimental controls. In a previous, mainly retrospective verification study, MSPrecise demonstrated a clear improvement in the ability to classify early-stage MS patients from those with other similarly presenting neurological diseases in comparison to OCB analysis. Individuals who present with clinical symptoms and evidence of nonspecific neurological disease undergo a battery of tests in a diagnostic process that can take months or even years to complete. The diagnostic standard-of-care for MS includes CSF analysis using the OCB test alongside a comprehensive set of clinical tests to rule-out other neurological diseases. Unfortunately, the OCB test yields a high rate of false-positive results, which can unnecessarily expose patients who do not have MS to chronic and expensive therapy that, in some cases, actually exacerbates their underlying disease.
“MSPrecise should offer neurologists greater insight into early disease events by exploiting the incredible biological resolution provided by next-generation sequencing. As we are able to now more accurately measure these key early biological changes, we believe we can help inform more appropriate courses of treatment for individuals who suffer from these types of immune-mediated diseases,” said Larry Tiffany, President and CEO, DioGenix.
“MSPrecise interrogates key genes involved in the immune system of patients being evaluated for MS. The growing body of evidence indicates this NGS assay may advance our efforts to more accurately diagnose patients with MS or other immune-mediated neurological disease,” said Elliot M. Frohman, MD, PhD, FAAN, Professor of Neurology & Ophthalmology, and Director, MS Program and Clinical Center for MS at The University of Texas Southwestern Medical Center. MSPrecise utilizes NGS to measure mutations found in rearranged immunoglobulin genes in immune cells initially isolated from cerebrospinal fluid. MSPrecise would augment the current standard-of-care for MS diagnosis by providing a more accurate measurement of a patient’s immune response to a challenge within the CNS. This novel method of measuring changes in adaptive human immunity may also be able to discern individuals whose disease is more progressive and requires more aggressive treatment.
DioGenix continues its sponsored research to determine if the same DNA mutation signature in patients with MS found in cerebral spinal fluid can be readily detected in blood. This research is supported by Fast Forward, a subsidiary of the [US] National MS Society.
Related Links:
DioGenix














