Screening Test Detects Tropical Disease in Blood Donors
By LabMedica International staff writers
Posted on 10 May 2010
A screening test that detects antibodies to a lethal tropical parasite will help safeguard blood for transfusions and tissue and organ recipients.Posted on 10 May 2010
The test is fully automated, and is highly sensitive and specific for the blood-borne parasite, Trypanosoma cruzi, the causative agent of Chagas diseases. It uses serum or plasma from donors of whole blood or blood components as well as from organ or tissue donors and detects antibodies to T. cruzi. The assay can be used to screen blood from the transplant donor whether the heart is still beating or even if it is not. It is not designed to be used with cord blood.
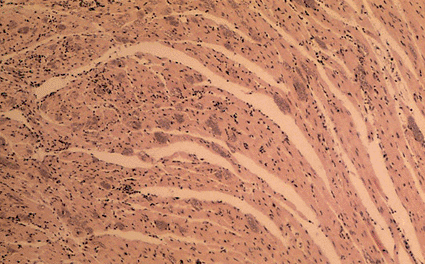
Image: Light micrograph of a section through clusters (small clumps of purple bodies) of Trypanosoma cruzi protozoans in the heart muscle of a patient suffering from Chagas disease (Photo courtesy of Sinclair Stammers / SPL).
Manufactured by Abbott Laboratories (Abbot Park, IL, USA), the Abbott Prism Chagas test is a two-step sandwich chemiluminescent assay that uses recombinant parasite antigens coated on microparticles. The test kit also contains anti-human conjugate and alkaline hydrogen peroxide for generating the photons. The test has detected antibodies to the parasite in over 1,000 donors since testing was instituted in 2007.
Reports indicate that 1 in every 27,500 blood donors in the United States will test positive for Chagas Disease. Blood donations in the United States exceed 15 million a year. The test is highly specific and does not react with serum of patients infected with other tropical or sexually transmitted diseases. An estimated 8-11 million people are known to be suffering from Chagas Disease in Mexico, Central and South America. The disease progresses from an acute phase to a chronic one, and is often undetected. The parasite has a predilection for the internal organs, especially the heart.
Karen Midthun, M.D., acting director of the US Food and Drug Administration's Center for Biologics Evaluation and Research, (FDA; Silver Spring, MD, USA), said that "Screening for T. cruzi is an important safety measure to help protect our blood supply and help prevent the spread of Chagas disease." This second-generation test has received FDA approval.
Related Links:
Abbott Laboratories
US Food and Drug Administration














