Immune Signature Identified in Treatment-Resistant Myasthenia Gravis
Posted on 03 Mar 2026
Myasthenia gravis is a rare autoimmune disorder in which immune attack at the neuromuscular junction causes fluctuating weakness that can impair vision, movement, speech, swallowing, and breathing. Although many patients improve with therapy, a subset develops refractory disease that remains difficult to control. Clinicians also lack reliable biomarkers to anticipate treatment response. Researchers now report a distinct immune signature linked to treatment resistance and potential therapeutic selection.
At the University of Manchester (Manchester, UK), investigators delineated an immune signature of refractory myasthenia gravis by profiling peripheral blood from people living with the disease and comparing it with samples from healthy volunteers. The signature featured an overactive adaptive immune response marked by increased memory B cells, together with markedly reduced regulatory T cells (Tregs). This pattern indicates loss of immunoregulatory braking alongside heightened effector activity at the neuromuscular junction.
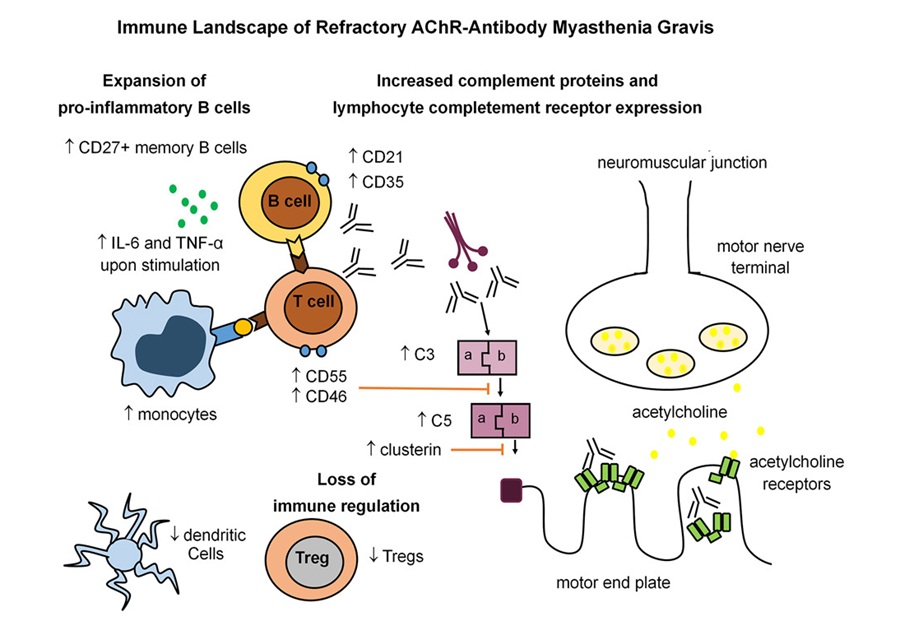
The team also described innate immune alterations, including reduced dendritic cells (DCs) and increased monocytes, together with heightened activity of the complement system. Taken together, these changes point to ongoing immune‑mediated damage and multifaceted dysregulation across adaptive and innate compartments. The findings were detailed in a study published in Med on February 6.
In a subset of refractory patients treated with rituximab, a B-cell–depleting therapy, peripheral B cells were successfully reduced in all cases, yet clinical improvement occurred only in some individuals. Non‑responders appeared to have disease driven by long‑lived plasma cells and particularly high complement activity. According to the findings, these patients may benefit more from therapies that target the complement pathway rather than B cells alone.
“For patients whose symptoms do not improve with existing treatments, the lack of clear answers can be incredibly frustrating,” said Dr. Katy Dodd, Neurology Consultant at Manchester Centre for Clinical Neuroscience. “Our findings help explain why some therapies work for certain patients but not others, and point toward more personalised approaches that could improve outcomes in the future.”
“Our study identifies a distinct immune signature associated with treatment-resistant myasthenia gravis,” said Dr. Madhvi Menon, UKRI Future Leaders Fellow at the Lydia Becker Institute of Immunology and Inflammation and lead author of the paper. “Understanding these immune differences brings us closer to predicting how patients will respond to therapy and to developing more targeted, personalised treatment approaches."
Related Links
University of Manchester














