Inactivation of Enzyme Corrects Alzheimer's Disease Defects
By LabMedica International staff writers
Posted on 28 Feb 2019
A team of neurodegenerative disease researchers identified a molecular mechanism that impedes protein synthesis and may contribute to the loss of memory and synaptic plasticity that characterizes Alzheimer's disease (AD).Posted on 28 Feb 2019
Maintenance of memory and synaptic plasticity depend on de novo protein synthesis, dysregulation of which is implicated in AD. Recent studies have demonstrated the AD-associated hyperphosphorylation of the mRNA translation factor eukaryotic elongation factor 2 (EEF2), which results in inhibition of protein synthesis.
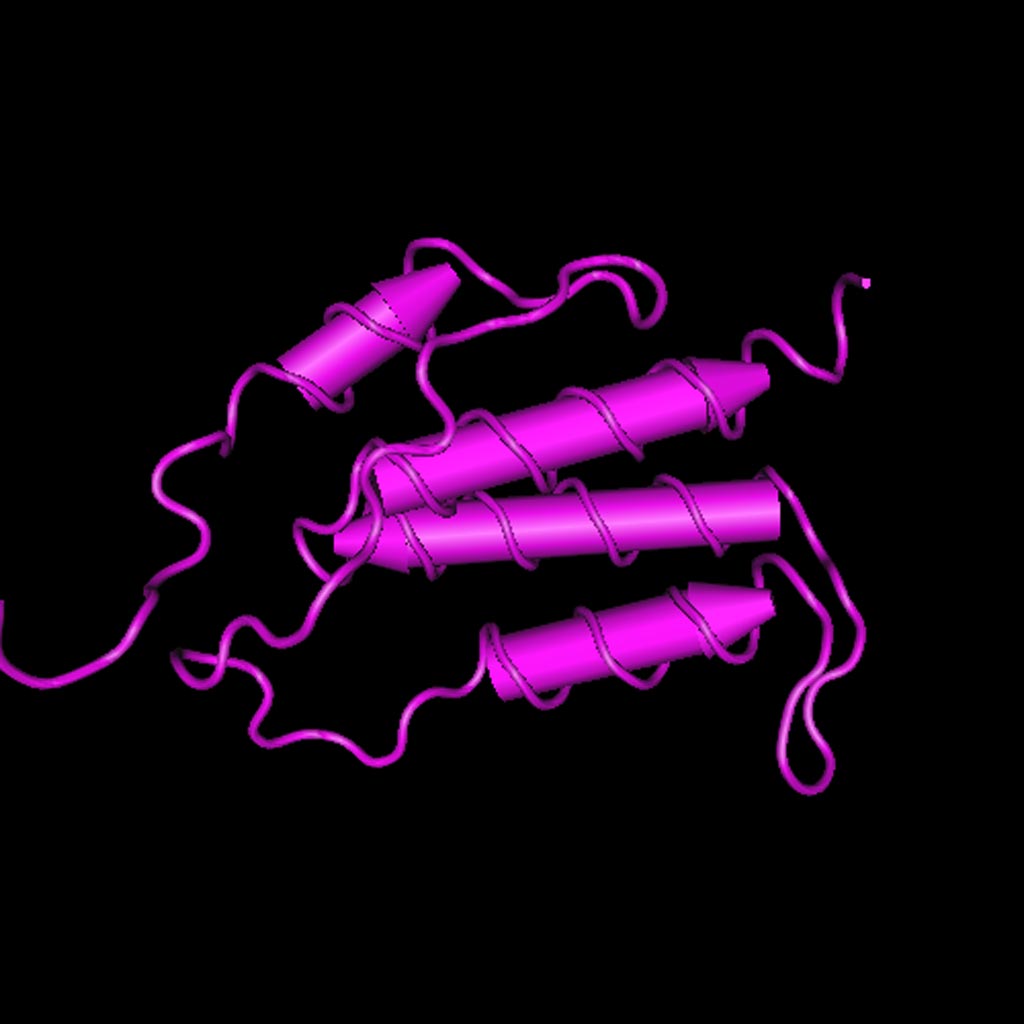
Image: Structure of the C-terminal helical repeat domain of eukaryotic elongation factor 2 kinase (EEF2K) (Photo courtesy of the U.S. National Center for Biotechnology Information).
The eukaryotic elongation factor 2 kinase (EEF2K) gene encodes a highly conserved protein kinase in the calmodulin-mediated signaling pathway that links activation of cell surface receptors to cell division. This kinase is involved in the regulation of protein synthesis by phosphorylating EEF2 and thus inhibiting its function. The activity of this kinase is increased in many cancers.
Investigators at Wake Forest Baptist Medical Center (Winston-Salem, NC, USA) examined whether suppression of EEF2 phosphorylation could improve protein synthesis capacity and AD-associated cognitive and synaptic impairments.
They reported in the February 1, 2019, issue of the Journal of Clinical Investigation that genetic inactivation of EEF2K in two AD mouse models suppressed AD-associated EEF2 hyperphosphorylation and improved memory deficits and hippocampal long-term potentiation (LTP) impairments without altering brain amyloid beta (Abeta) pathology. Furthermore, EEF2K reduction alleviated AD-associated defects in dendritic spine morphology, postsynaptic density formation, de novo protein synthesis, and dendritic polyribosome assembly. These results linked EEF2K/EEF2 signaling dysregulation to AD neurological defects and offer a feasible therapeutic target to correct them.
“Alzheimer’s is such a devastating disease and currently there is no cure or effective therapy,” said senior author Dr. Tao Ma, assistant professor of gerontology and geriatric medicine at Wake Forest Baptist Medical Center. “All completed clinical trials of new drugs have failed so there is clearly a need for novel therapeutic targets for potential treatments. These findings are encouraging and provide a new pathway for further research.”
Related Links:
Wake Forest Baptist Medical Center