Suppression of a Specific Kinase Blocks Growth of Dependent Tumors
By LabMedica International staff writers
Posted on 20 Dec 2018
A recent paper revealed that the enzyme glycogen synthase kinase 3 (GSK3) was required for the in vitro and in vivo growth and survival of human mutant KRas-dependent tumors but was dispensable for mutant KRas-independent tumors.Posted on 20 Dec 2018
Approximately 20% of all human cancers have mutations in the KRas (V-Ki-ras2 Kirsten rat sarcoma viral oncogene homolog) gene. These KRas-mutant cancers are among the most difficult to treat due to their resistance to chemotherapy.
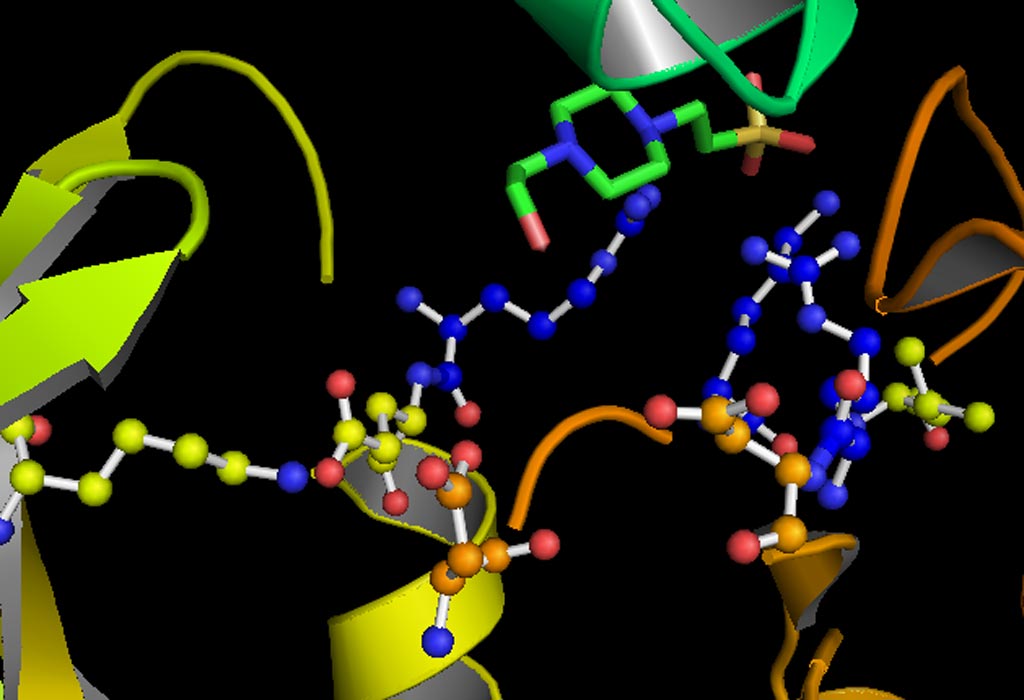
Image: A model of the active site of the kinase GSK-3 (glycogen synthase kinase 3) (Photo courtesy of Wikimedia Commons).
Since targeting KRas directly has proven difficult, identifying vulnerabilities specific for mutant KRas tumors is an important alternative approach. Toward this end, investigators at the Moffit Cancer Center (Tampa, FL, USA) sought to identify kinases and their corresponding pathways that were required by mutant KRas tumors in order to induce malignant transformation and to target such pathways for cancer therapy.
They reported in the December 4, 2018, online edition of the journal Nature Communications that the kinase GSK3 was necessary for survival of a KRas-dependent cancer cell line, and that suppression of GSK3 inhibited tumor growth by increasing the levels of beta-catenin, a dual function protein, involved in regulation and coordination of cell–cell adhesion and gene transcription, and c-Myc, a regulator of genes and proto-oncogenes that code for transcription factors. Inhibiting phosphorylation of the GSK3 substrates c-Myc and beta-catenin and their subsequent upregulation contributed to the antitumor activity of GSK3 inhibition.
The investigators showed that GSK3 chemical or genetic blockade inhibited the growth in mice of mutant KRas primary and metastatic patient-derived xenografts from pancreatic cancer patients who had progressed despite chemo- and radiation therapies. This discovery has opened new avenues to target mutant KRas-dependent cancers.
“This study discovered a novel vulnerability in mutant KRas-addicted human cancers. We have uncovered an essential link between GSK3 activity and mutant KRas dependency that is highly relevant in many aggressive and end-stage cancers. This discovery opens new avenues to target mutant KRas-addicted cancers,” said senior author Dr. Said Sebti, a senior member of the drug discovery department at Moffit Cancer Center.
Related Links:
Moffit Cancer Center