Tripeptide Drug Effectively Controls Metabolic Syndrome in Rodent Model
By LabMedica International staff writers
Posted on 22 Dec 2014
Promising results in reducing obesity and normalizing glucose metabolism obtained with a synthetic dipeptide drug have been enhanced by the addition of a molecule of a third hormone, glucagon.Posted on 22 Dec 2014
Investigators at Indiana University (Bloomington, USA) had reported previously that a "unimolecular dual incretin" derived from an intermixed peptide sequence from the hormones GLP-1 and GIP corrected two causal mechanisms of diabetes-linked obesity, adiposity-induced insulin resistance, and pancreatic insulin deficiency more effectively than did selective mono-agonists. This superior efficacy translated across rodent models of obesity and diabetes, including db/db mice and ZDF rats, to primates (cynomolgus monkeys and humans).
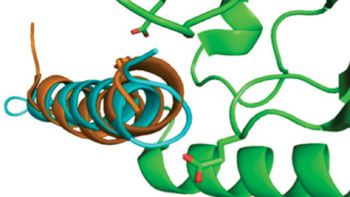
Image: The new peptide offers a triple hormone effect in a single-cell molecule (Photo courtesy of Indiana University).
Incretins are a group of gastrointestinal hormones that cause an increase in the amount of insulin released from the beta cells of the islets of Langerhans after eating, even before blood glucose levels become elevated. They also slow the rate of absorption of nutrients into the blood stream by reducing gastric emptying and may directly reduce food intake. Incretins also inhibit glucagon release from the alpha cells of the Islets of Langerhans. The two main candidate molecules that fulfill criteria for an incretin are glucagon-like peptide-1 (GLP-1) and gastric inhibitory peptide (also known as: glucose-dependent insulinotropic polypeptide or GIP). Both GLP-1 and GIP are rapidly inactivated by the enzyme dipeptidyl peptidase-4 (DPP-4).
In a paper published in the December 8, 2014, online edition of the journal Nature Medicine, the investigators and their colleagues at the German Research Center for Environmental Health (Neuherberg, Germany) discussed results obtained with a new tripeptide drug that comprised the previous GLP-1/GIP combination with the addition of the hormone glucagon. Glucagon enhanced the effects of the other two hormones by increasing energy expenditure.
Results obtained by treating a rodent model of metabolic syndrome with the tripeptide drug showed that the new compound specifically and equally targeted three receptors of GLP-1, GIP, and glucagon, and reduced the animals' body weight by about 30%, almost twice as much as the GLP-1/GIP double hormone.
"This peptide represents the first rationally designed, fully potent, and balanced triple agonist ever achieved in the treatment of any disease," said contributing author Dr. Richard DiMarchi, professor of chemistry at Indiana University. "The benefits of the previously reported individual co-agonists have been integrated to a single molecule of triple action that provides unprecedented efficacy to lower body weight and control metabolism."
Human clinical trials of the tripeptide drug are being managed by Roche (Basel, Switzerland).
Related Links:
Indiana University
German Research Center for Environmental Health
Roche




.jpg)