Rac1 Levels Linked to Mechanics of Invadopodia Formation and Metastasis
By LabMedica International staff writers
Posted on 10 Jun 2014
Cancer researchers have linked increases and decreases in the level of Rac1 protein to the appearance and disappearance of invadopodia, amoeboid-like protrusions used by metastatic cancer cells to invade neighboring tissues.Posted on 10 Jun 2014
Invadopodia release enzymes that degrade the extracellular matrix (ECM) and allow cellular movement while propelling the cancer cell into neighboring tissues.
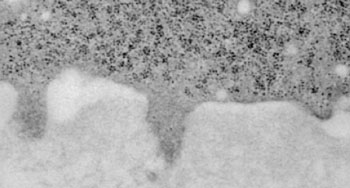
Image: Electron micrograph shows a cancer cell (upper darker area) that has formed three invadopodia that are penetrating the adjacent extracellular matrix (lower lighter area) (Photo courtesy of Yeshiva University).
Rac1, also known as Ras-related C3 botulinum toxin substrate 1, is a protein encoded by the RAC1 gene. This gene can produce a variety of alternatively spliced versions of the Rac1 protein, which appear to carry out different functions. Rac1 is thought to play a significant role in the development of various cancers, including melanoma and non-small-cell lung cancer. As a result, it is now considered a therapeutic target for these diseases. Rac1 is a small (approximately 21 kDa) signaling G-protein (more specifically a GTPase), and is a regulator of many cellular processes, including the cell cycle, cell-cell adhesion, motility (through the actin network), and of epithelial differentiation (proposed to be necessary for maintaining epidermal stem cells).
To study Rac1 involvement in invadopodia dynamics, investigators at Yeshiva University (New York, NY, USA) developed a genetically encoded, single-chain Rac1 fluorescence resonance energy (FRET) transfer biosensor technique that, combined with live-cell imaging, revealed exactly when and where Rac1 was activated inside cancer cells.
Results obtained by this methodology revealed that low levels of Rac1 were found during formation of invadopodia and while they were actively degrading the ECM. Elevated Rac1 levels coincided with disappearance of the invadopodia. These findings were confirmed by using siRNAs to silence the RAC1 gene. When the gene was inactivated, ECM degradation increased. Conversely, when Rac1 activity was enhanced - using light to activate a Rac1 protein analog - the invadopodia disappeared.
“We have known for some time that invadopodia are driven by protein filaments called actin,” said senior author Dr. Louis Hodgson, assistant professor of anatomy and structural biology at Yeshiva University. “But exactly what was regulating the actin in invadopodia was not clear. Rac1 levels in invadopodia of invasive tumor cells appear to surge and ebb at precisely timed intervals in order to maximize the cells’ invasive capabilities. So high levels of Rac1 induce the disappearance of ECM-degrading invadopodia, while low levels allow them to stay—which is the complete opposite of what Rac1 was thought to be doing in invadopodia.”
“Rac1 inhibitors have been developed,” said Dr. Hodgson, “but it would not be safe to use them indiscriminately. Rac1 is an important molecule in healthy cells, including immune cells. So we would need to find a way to shut off this signaling pathway specifically in cancer cells.”
Related Links:
Yeshiva University









 Analyzer.jpg)