Histone Deacetylase Inhibitors Boost Parvovirus Cancer Killing Action
By LabMedica International staff writers
Posted on 28 Oct 2013
The anticancer action of parvoviruses can be dramatically improved by co-treating cancer cells with parvovirus and histone deacetylase inhibitors (HDACIs) such as valproic acid.Posted on 28 Oct 2013
The rat parvovirus H-1PV has oncolytic and tumor-suppressive properties potentially exploitable in cancer therapy. This possibility is being explored by cancer researchers, and while results have been encouraging, they have found that it is necessary to improve the cancer-killing capability of the virus.
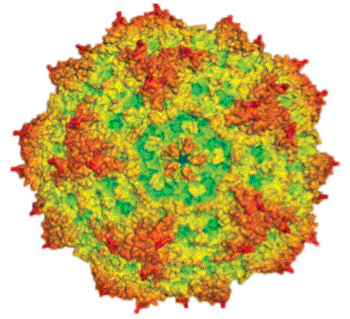
Image: Computer-generated representation of parvovirus H-1 (H-1PV) (Photo courtesy of Dr. Antonio Marchini, German Cancer Research Center).
Towards this end, investigators at the German Cancer Research Center (Heidelberg) have sought drugs or drug combinations that would improve the ability of parvoviruses to kill cancer cells. In a paper published in the September 17, 2013, online edition of the journal EMBO Molecular Medicine they described the effect of the histone deacetylase inhibitor valproic acid (VPA) on the interaction between H-1PV and human cervical carcinoma and pancreatic carcinoma cell lines.
The investigators showed that co-treatment of cultures with the parvovirus and VPA boosted the ability of the virus to kill a range of human cervical carcinoma and pancreatic carcinoma cell lines by inducing oxidative stress, DNA damage, and apoptosis. Furthermore, in rat and mouse xenograft models, H-1PV/VPA co-treatment strongly inhibited tumor growth promoting complete tumor remission in all co-treated animals. In contrast, animals treated with the same virus dose without the drug displayed no regression, not even when a 20-times higher dose of viruses was administered.
At the molecular level, the investigators found that the parvovirus nonstructural protein NS1 modulated viral transcription and cytotoxicity, both of which were enhanced by VPA treatment. NS1 was acetylated at residues K85 and K257 and addition of VPA correlated with an enhanced rate of NS1 acetylation. In contrast, amino-acid substitution of the two acetylation sites strongly impaired NS1-mediated viral gene transcription, viral replication, and cytotoxicity. VPA induced hyper-acetylation of NS1, which converted the protein into a more active polypeptide.
"The synergistic effect of a combination of parvoviruses and valproic acid enables us to deliver both the viruses and the drug at low doses, which prevents severe side effects," said senior author Dr. Antonio Marchini, a principle investigator in virology at the German Cancer Research Center. "The results are encouraging us to carry out further tests of this combination therapy. We believe it has the potential to arrest tumor growth in severe cases of cancer. We obtained impressive results in preclinical trials with parvovirus H-1 in brain tumors. However, the oncolytic effect of the viruses is weaker in other cancers. Therefore, we are searching for ways to increase the therapeutic potential of the viruses."
Related Links:
German Cancer Research Center