New Biorepository Facility Offers Wide Range of Storage Options
By LabMedica International staff writers
Posted on 06 Jun 2011
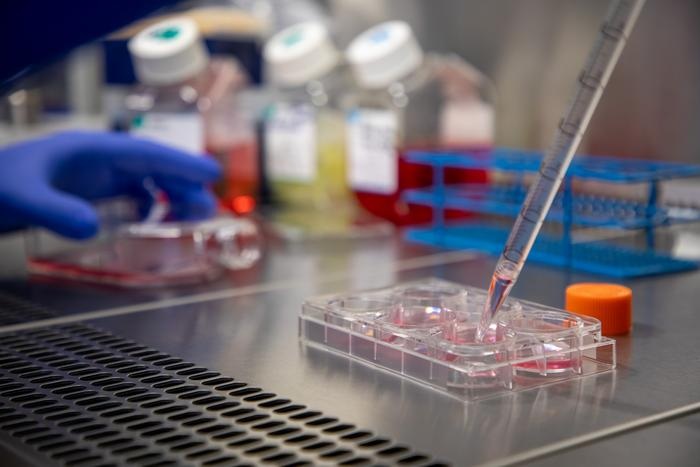
A provider of comprehensive sample management solutions has opened its new state-of-the-art biorepository facility in Indianapolis (IN, USA).Posted on 06 Jun 2011
The 60,000-square-foot facility is part of the company's strategic growth plan, and represents a US$4.6 million investment by BioStorage Technologies (Zionsville, IN, USA).
The new biorepository facility offers a wide range of storage options, including automated carousel controlled-room temperature storage at 15 °C to 27 °C, bulk sample storage at 15 °C to 27 °C, walk-in cold sample storage -20 °C to 5 °C, ultra-low temperature storage -70 °C to -80 °C, and -190 °C vapor phase liquid nitrogen.
The biorepository facility is strategically placed near the second largest FedEx hub in the United States. It is dedicated to the preparation, storage, and cold-chain transport of human biological samples for a wide variety of customers, including donor programs, academic centers, contract research organizations, and biotechnology companies.
Once samples are processed, aliquots of blood and/or DNA products can be retrieved and scheduled for transport through the company's proprietary tracking and inventory management system, ISISS. This process ensures samples are removed, transported, processed, and replaced to storage in a compliant, consistent, and efficient fashion.
"The future of drug discovery is in personalized medicine and genomic-based therapies, which will require an ability to store and analyze large quantities of tissue and blood samples," said Greg Swanberg, CEO of BioStorage Technologies. "As this trend continues, an increasing amount of pharma and biotech companies rely on our expertise in comprehensive sample management to ensure their valuable biological samples are managed in optimal conditions and not jeopardized by preanalytical variables that can have a negative impact on sample integrity."
Related Links:
BioStorage Technologies