Basophil Activation Test Predicts Pediatric Wheezing Prognosis
|
By LabMedica International staff writers Posted on 20 Jan 2021 |

Image: The CytoFLEX benchtop flow cytometer features up to three lasers for 13-color flow cytometry (Photo courtesy of Beckman Coulter Life Sciences).
Although asthma is a common chronic airway inflammatory disease in children, the natural history of asthma remains unknown. It is difficult to diagnose asthma in children aged under five years because of the complexity of wheezing phenotypes at that age.
Basophils are important immune cells that are involved in allergic diseases including asthma. CD63-based basophil activation tests (BATs) are a useful tool for not only identifying specific allergens, but also monitoring immunologic homeostasis. BATs are used for diagnosing food and drug allergies with high specificity and sensitivity.
A team of Pediatricians at the Shanghai Jiao Tong University School of Medicine (Shanghai, China) conducted a prospective cohort observation trial from July 2017 to March 2018 and enrolled children aged under five years with a convincing history of wheezing episodes (confirmed by the team or documented as “expiratory wheeze” in previous medical records). All enrolled subjects participated in specific immunoglobulin E (sIgE) testing. Serum samples were measured via the DX-Blot 45 Automatic Western Blotting instrument (Hangzhou Zhejiang Dixun Biological Genetic Engineering Co., Ltd. Hangzhou, Zhejiang, China).
The scientists collected anti-coagulated peripheral blood and stored at 2–8 °C for up to 6 hours. The Basophil Activation Test Set (Bühlmann Laboratories, Schönenbuch, Switzerland) was used for the BAT. In vitro stimulation was performed using an inhalant mix (Bühlmann Laboratories) that included common inhalation allergens (GX1, G12, T3, T4, W6, W9, M6, D1, D2, E1, and E2) as specific antigens. After processing and the red blood cells were lysed, the cells were resuspended in phosphate-buffered saline and assessed with a CytoFLEX flow cytometer (Beckman Coulter, Brea, CA, USA). The test result was considered positive when the level of CD63 expression in stimulated cells was >15% over the baseline level.
The investigators reported that of the 45 originally enrolled children, 38 completed both the follow-up and a BAT. After stimulation with the inhalant mixture, the CD63 expression on basophils and the rate of positive CD63-based BAT results in children diagnosed with asthma were both significantly higher than those in children who were not diagnosed with asthma. For the prediction of asthma, the positive predictive value and negative predictive value of CD63-based BAT was 71.8 and 69.2%, respectively. The positive likelihood ratio and negative likelihood ratio of CD63-based BAT were 1.70 and 0.3, respectively.
The authors concluded that their study indicates that CD63-based BAT has potential clinical value for predicting asthma outcome in young children with wheezing episodes. The study was published on January 6, 2021 in the journal BMC Immunology.
Related Links:
Shanghai Jiao Tong University School of Medicine
Hangzhou Zhejiang Dixun Biological Genetic Engineering Co
Bühlmann Laboratories
Beckman Coulter
Basophils are important immune cells that are involved in allergic diseases including asthma. CD63-based basophil activation tests (BATs) are a useful tool for not only identifying specific allergens, but also monitoring immunologic homeostasis. BATs are used for diagnosing food and drug allergies with high specificity and sensitivity.
A team of Pediatricians at the Shanghai Jiao Tong University School of Medicine (Shanghai, China) conducted a prospective cohort observation trial from July 2017 to March 2018 and enrolled children aged under five years with a convincing history of wheezing episodes (confirmed by the team or documented as “expiratory wheeze” in previous medical records). All enrolled subjects participated in specific immunoglobulin E (sIgE) testing. Serum samples were measured via the DX-Blot 45 Automatic Western Blotting instrument (Hangzhou Zhejiang Dixun Biological Genetic Engineering Co., Ltd. Hangzhou, Zhejiang, China).
The scientists collected anti-coagulated peripheral blood and stored at 2–8 °C for up to 6 hours. The Basophil Activation Test Set (Bühlmann Laboratories, Schönenbuch, Switzerland) was used for the BAT. In vitro stimulation was performed using an inhalant mix (Bühlmann Laboratories) that included common inhalation allergens (GX1, G12, T3, T4, W6, W9, M6, D1, D2, E1, and E2) as specific antigens. After processing and the red blood cells were lysed, the cells were resuspended in phosphate-buffered saline and assessed with a CytoFLEX flow cytometer (Beckman Coulter, Brea, CA, USA). The test result was considered positive when the level of CD63 expression in stimulated cells was >15% over the baseline level.
The investigators reported that of the 45 originally enrolled children, 38 completed both the follow-up and a BAT. After stimulation with the inhalant mixture, the CD63 expression on basophils and the rate of positive CD63-based BAT results in children diagnosed with asthma were both significantly higher than those in children who were not diagnosed with asthma. For the prediction of asthma, the positive predictive value and negative predictive value of CD63-based BAT was 71.8 and 69.2%, respectively. The positive likelihood ratio and negative likelihood ratio of CD63-based BAT were 1.70 and 0.3, respectively.
The authors concluded that their study indicates that CD63-based BAT has potential clinical value for predicting asthma outcome in young children with wheezing episodes. The study was published on January 6, 2021 in the journal BMC Immunology.
Related Links:
Shanghai Jiao Tong University School of Medicine
Hangzhou Zhejiang Dixun Biological Genetic Engineering Co
Bühlmann Laboratories
Beckman Coulter
Latest Immunology News
- Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
- AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
- Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
- Testing Method Could Help More Patients Receive Right Cancer Treatment
- Groundbreaking Test Monitors Radiation Therapy Toxicity in Cancer Patients
- State-Of-The Art Techniques to Investigate Immune Response in Deadly Strep A Infections
- Novel Immunoassays Enable Early Diagnosis of Antiphospholipid Syndrome
- New Test Could Predict Immunotherapy Success for Broader Range Of Cancers
- Simple Blood Protein Tests Predict CAR T Outcomes for Lymphoma Patients
- Cell Sorter Chip Technology to Pave Way for Immune Profiling at POC
- Chip Monitors Cancer Cells in Blood Samples to Assess Treatment Effectiveness
- Automated Immunohematology Approaches Can Resolve Transplant Incompatibility
- AI Leverages Tumor Genetics to Predict Patient Response to Chemotherapy
- World’s First Portable, Non-Invasive WBC Monitoring Device to Eliminate Need for Blood Draw
- Predictive T-Cell Test Detects Immune Response to Viruses Even Before Antibodies Form
- Single Blood Draw to Detect Immune Cells Present Months before Flu Infection Can Predict Symptoms
Channels
Clinical Chemistry
view channel
3D Printed Point-Of-Care Mass Spectrometer Outperforms State-Of-The-Art Models
Mass spectrometry is a precise technique for identifying the chemical components of a sample and has significant potential for monitoring chronic illness health states, such as measuring hormone levels... Read more.jpg)
POC Biomedical Test Spins Water Droplet Using Sound Waves for Cancer Detection
Exosomes, tiny cellular bioparticles carrying a specific set of proteins, lipids, and genetic materials, play a crucial role in cell communication and hold promise for non-invasive diagnostics.... Read more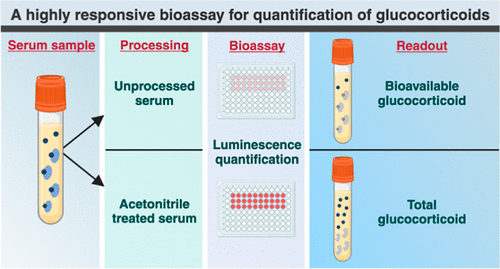
Highly Reliable Cell-Based Assay Enables Accurate Diagnosis of Endocrine Diseases
The conventional methods for measuring free cortisol, the body's stress hormone, from blood or saliva are quite demanding and require sample processing. The most common method, therefore, involves collecting... Read moreMolecular Diagnostics
view channel
Blood Test Accurately Predicts Lung Cancer Risk and Reduces Need for Scans
Lung cancer is extremely hard to detect early due to the limitations of current screening technologies, which are costly, sometimes inaccurate, and less commonly endorsed by healthcare professionals compared... Read more
Unique Autoantibody Signature to Help Diagnose Multiple Sclerosis Years before Symptom Onset
Autoimmune diseases such as multiple sclerosis (MS) are thought to occur partly due to unusual immune responses to common infections. Early MS symptoms, including dizziness, spasms, and fatigue, often... Read more
Blood Test Could Detect HPV-Associated Cancers 10 Years before Clinical Diagnosis
Human papilloma virus (HPV) is known to cause various cancers, including those of the genitals, anus, mouth, throat, and cervix. HPV-associated oropharyngeal cancer (HPV+OPSCC) is the most common HPV-associated... Read moreHematology
view channel
Next Generation Instrument Screens for Hemoglobin Disorders in Newborns
Hemoglobinopathies, the most widespread inherited conditions globally, affect about 7% of the population as carriers, with 2.7% of newborns being born with these conditions. The spectrum of clinical manifestations... Read more
First 4-in-1 Nucleic Acid Test for Arbovirus Screening to Reduce Risk of Transfusion-Transmitted Infections
Arboviruses represent an emerging global health threat, exacerbated by climate change and increased international travel that is facilitating their spread across new regions. Chikungunya, dengue, West... Read more
POC Finger-Prick Blood Test Determines Risk of Neutropenic Sepsis in Patients Undergoing Chemotherapy
Neutropenia, a decrease in neutrophils (a type of white blood cell crucial for fighting infections), is a frequent side effect of certain cancer treatments. This condition elevates the risk of infections,... Read more
First Affordable and Rapid Test for Beta Thalassemia Demonstrates 99% Diagnostic Accuracy
Hemoglobin disorders rank as some of the most prevalent monogenic diseases globally. Among various hemoglobin disorders, beta thalassemia, a hereditary blood disorder, affects about 1.5% of the world's... Read moreMicrobiology
view channel
New CE-Marked Hepatitis Assays to Help Diagnose Infections Earlier
According to the World Health Organization (WHO), an estimated 354 million individuals globally are afflicted with chronic hepatitis B or C. These viruses are the leading causes of liver cirrhosis, liver... Read more
1 Hour, Direct-From-Blood Multiplex PCR Test Identifies 95% of Sepsis-Causing Pathogens
Sepsis contributes to one in every three hospital deaths in the US, and globally, septic shock carries a mortality rate of 30-40%. Diagnosing sepsis early is challenging due to its non-specific symptoms... Read morePathology
view channelAI-Powered Digital Imaging System to Revolutionize Cancer Diagnosis
The process of biopsy is important for confirming the presence of cancer. In the conventional histopathology technique, tissue is excised, sliced, stained, mounted on slides, and examined under a microscope... Read more
New Mycobacterium Tuberculosis Panel to Support Real-Time Surveillance and Combat Antimicrobial Resistance
Tuberculosis (TB), the leading cause of death from an infectious disease globally, is a contagious bacterial infection that primarily spreads through the coughing of patients with active pulmonary TB.... Read moreTechnology
view channel
New Diagnostic System Achieves PCR Testing Accuracy
While PCR tests are the gold standard of accuracy for virology testing, they come with limitations such as complexity, the need for skilled lab operators, and longer result times. They also require complex... Read more
DNA Biosensor Enables Early Diagnosis of Cervical Cancer
Molybdenum disulfide (MoS2), recognized for its potential to form two-dimensional nanosheets like graphene, is a material that's increasingly catching the eye of the scientific community.... Read more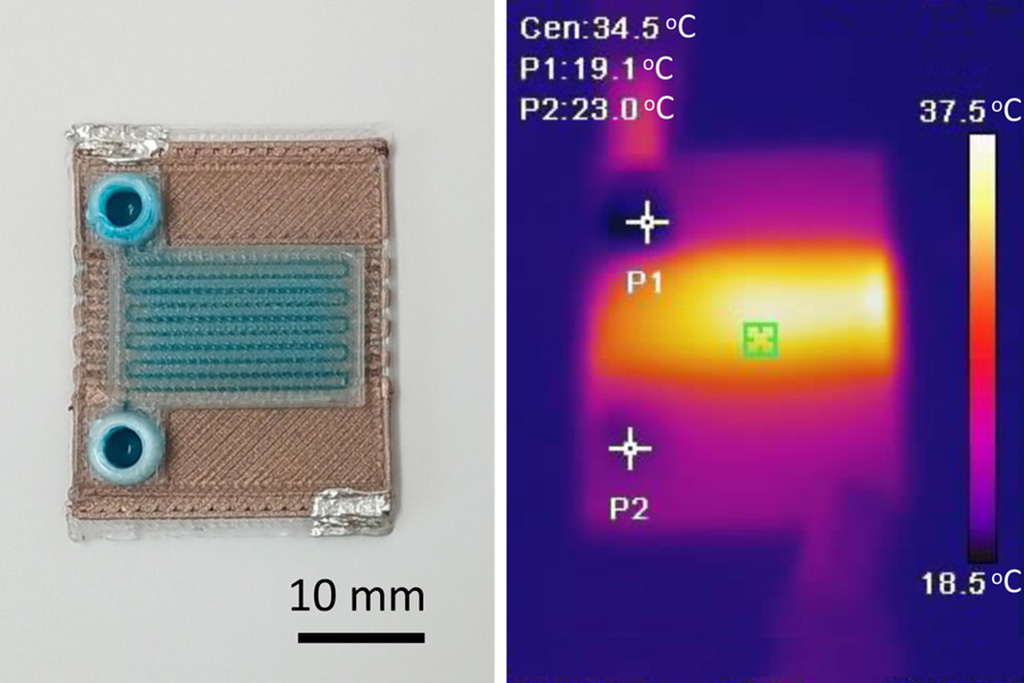
Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
Microfluidics, which are miniature devices that control the flow of liquids and facilitate chemical reactions, play a key role in disease detection from small samples of blood or other fluids.... Read more
Breakthrough in Diagnostic Technology Could Make On-The-Spot Testing Widely Accessible
Home testing gained significant importance during the COVID-19 pandemic, yet the availability of rapid tests is limited, and most of them can only drive one liquid across the strip, leading to continued... Read moreIndustry
view channel
ECCMID Congress Name Changes to ESCMID Global
Over the last few years, the European Society of Clinical Microbiology and Infectious Diseases (ESCMID, Basel, Switzerland) has evolved remarkably. The society is now stronger and broader than ever before... Read more
Bosch and Randox Partner to Make Strategic Investment in Vivalytic Analysis Platform
Given the presence of so many diseases, determining whether a patient is presenting the symptoms of a simple cold, the flu, or something as severe as life-threatening meningitis is usually only possible... Read more
Siemens to Close Fast Track Diagnostics Business
Siemens Healthineers (Erlangen, Germany) has announced its intention to close its Fast Track Diagnostics unit, a small collection of polymerase chain reaction (PCR) testing products that is part of the... Read more













.jpg)