Rapid UTI, Sepsis Tests Uses Digital PCR
|
By LabMedica International staff writers Posted on 30 Jan 2020 |
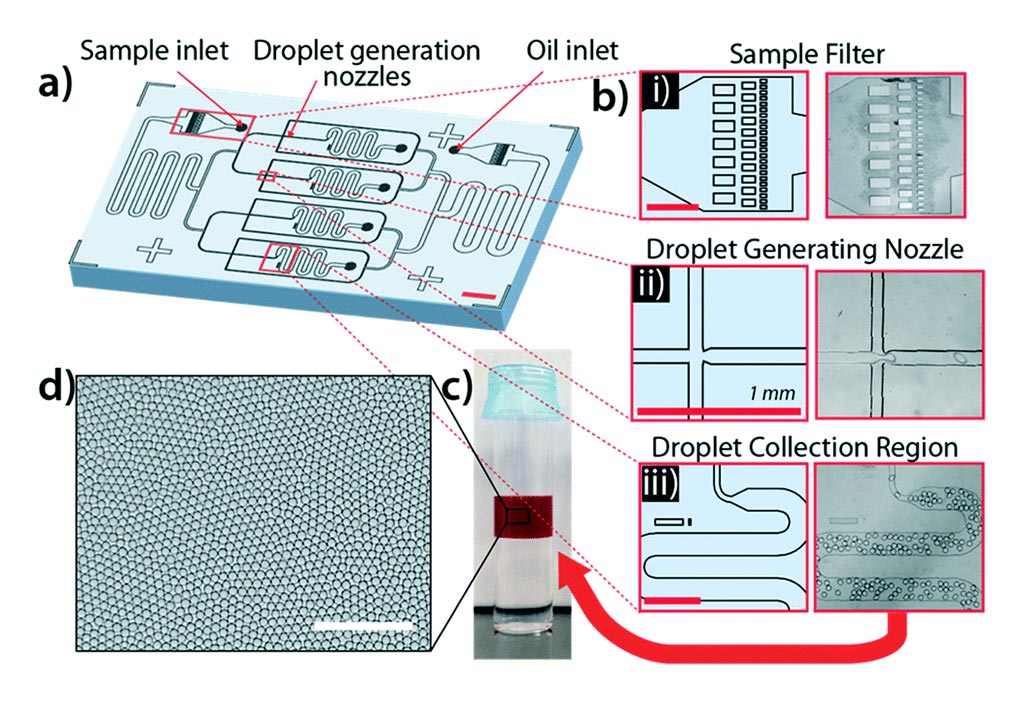
Image: The system for rapid bacterial detection and antibiotic susceptibility testing in whole blood using one-step, high throughput blood digital PCR (Photo courtesy of Velox Biosystems).
Sepsis due to antimicrobial resistant pathogens is a major health problem worldwide. The inability to rapidly detect and thus treat bacteria with appropriate agents in the early stages of infections leads to excess morbidity, mortality, and healthcare costs.
Sepsis can be caused by a very low abundance of pathogens in the bloodstream, and most diagnostic technologies require pathogen concentration, a days-long culture step, and pathogen extraction steps that have the potential to reduce sensitivity. Time to diagnosis is also critical in cases of suspected sepsis, as mortality increases 4% for every hour delay until treatment.
Scientists at the University of California, Irvine (Irvine, CA, USA) and their associates have developed a rapid diagnostic platform that integrates a novel one-step blood droplet digital polymerase chain reaction (PCR) assay and a high throughput 3D particle counter system with potential to perform bacterial identification and antibiotic susceptibility profiling directly from whole blood specimens, without requiring culture and sample processing steps.
The team used the cefotaximase-M-9 (CTX-M-9) family extended-spectrum β-lactamases (ESBLs) as a model system, and demonstrated that their technology developed by Velox Biosystems (Irvine, CA, USA) can simultaneously achieve unprecedented high sensitivity (10 CFU per mL) and rapid sample-to-answer assay time (one hour). In head-to-head studies, by contrast, real time PCR and Bio-Rad ddPCR (Bio-Rad, Hercules, CA, USA) only exhibited a limit of detection of 1,000 CFU per mL and 50–100 CFU per mL, respectively.
In the study, the team spiked whole-blood samples with a strain of Escherichia coli containing a synthetic resistance gene and was able to detect bacteria and resistance within one hour. The detection required no culture or sample processing, with a limit of detection of less than 10 colony-forming units per milliliter. The group was also able to demonstrate multiplexing ability for two targets per droplet. The study also detailed strategies to overcome the PCR inhibitory effects common to whole blood, a method to enhance the ability to partition blood into droplets by diluting it, and determined that the ideal fluorophore to get past the autofluorescence of blood was a dye called Quasar 670 from LGC Biosearch Technologies (Teddington, UK).
The team also demonstrated that the technology can be broadly applicable for targeted detection of a wide range of antibiotic resistant genes found in both Gram-positive (vanA, nuc, and mecA) and Gram-negative bacteria, including ESBLs (blaCTX-M-1 and blaCTX-M-2 families) and CREs (blaOXA-48 and blaKPC), as well as bacterial speciation (E. coli and Klebsiella spp.) and pan-bacterial detection, without requiring blood culture or sample processing. The study was first published on December 16, 2019 in the journal Lab on a Chip.
Related Links:
University of California, Irvine
Velox Biosystems
LGC Biosearch Technologies
Sepsis can be caused by a very low abundance of pathogens in the bloodstream, and most diagnostic technologies require pathogen concentration, a days-long culture step, and pathogen extraction steps that have the potential to reduce sensitivity. Time to diagnosis is also critical in cases of suspected sepsis, as mortality increases 4% for every hour delay until treatment.
Scientists at the University of California, Irvine (Irvine, CA, USA) and their associates have developed a rapid diagnostic platform that integrates a novel one-step blood droplet digital polymerase chain reaction (PCR) assay and a high throughput 3D particle counter system with potential to perform bacterial identification and antibiotic susceptibility profiling directly from whole blood specimens, without requiring culture and sample processing steps.
The team used the cefotaximase-M-9 (CTX-M-9) family extended-spectrum β-lactamases (ESBLs) as a model system, and demonstrated that their technology developed by Velox Biosystems (Irvine, CA, USA) can simultaneously achieve unprecedented high sensitivity (10 CFU per mL) and rapid sample-to-answer assay time (one hour). In head-to-head studies, by contrast, real time PCR and Bio-Rad ddPCR (Bio-Rad, Hercules, CA, USA) only exhibited a limit of detection of 1,000 CFU per mL and 50–100 CFU per mL, respectively.
In the study, the team spiked whole-blood samples with a strain of Escherichia coli containing a synthetic resistance gene and was able to detect bacteria and resistance within one hour. The detection required no culture or sample processing, with a limit of detection of less than 10 colony-forming units per milliliter. The group was also able to demonstrate multiplexing ability for two targets per droplet. The study also detailed strategies to overcome the PCR inhibitory effects common to whole blood, a method to enhance the ability to partition blood into droplets by diluting it, and determined that the ideal fluorophore to get past the autofluorescence of blood was a dye called Quasar 670 from LGC Biosearch Technologies (Teddington, UK).
The team also demonstrated that the technology can be broadly applicable for targeted detection of a wide range of antibiotic resistant genes found in both Gram-positive (vanA, nuc, and mecA) and Gram-negative bacteria, including ESBLs (blaCTX-M-1 and blaCTX-M-2 families) and CREs (blaOXA-48 and blaKPC), as well as bacterial speciation (E. coli and Klebsiella spp.) and pan-bacterial detection, without requiring blood culture or sample processing. The study was first published on December 16, 2019 in the journal Lab on a Chip.
Related Links:
University of California, Irvine
Velox Biosystems
LGC Biosearch Technologies
Latest Technology News
- New Diagnostic System Achieves PCR Testing Accuracy
- DNA Biosensor Enables Early Diagnosis of Cervical Cancer
- Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
- Breakthrough in Diagnostic Technology Could Make On-The-Spot Testing Widely Accessible
- First of Its Kind Technology Detects Glucose in Human Saliva
- Electrochemical Device Identifies People at Higher Risk for Osteoporosis Using Single Blood Drop
- Novel Noninvasive Test Detects Malaria Infection without Blood Sample
- Portable Optofluidic Sensing Devices Could Simultaneously Perform Variety of Medical Tests
- Point-of-Care Software Solution Helps Manage Disparate POCT Scenarios across Patient Testing Locations
- Electronic Biosensor Detects Biomarkers in Whole Blood Samples without Addition of Reagents
- Breakthrough Test Detects Biological Markers Related to Wider Variety of Cancers
- Rapid POC Sensing Kit to Determine Gut Health from Blood Serum and Stool Samples
- Device Converts Smartphone into Fluorescence Microscope for Just USD 50
- Wi-Fi Enabled Handheld Tube Reader Designed for Easy Portability
Channels
Clinical Chemistry
view channel
3D Printed Point-Of-Care Mass Spectrometer Outperforms State-Of-The-Art Models
Mass spectrometry is a precise technique for identifying the chemical components of a sample and has significant potential for monitoring chronic illness health states, such as measuring hormone levels... Read more.jpg)
POC Biomedical Test Spins Water Droplet Using Sound Waves for Cancer Detection
Exosomes, tiny cellular bioparticles carrying a specific set of proteins, lipids, and genetic materials, play a crucial role in cell communication and hold promise for non-invasive diagnostics.... Read more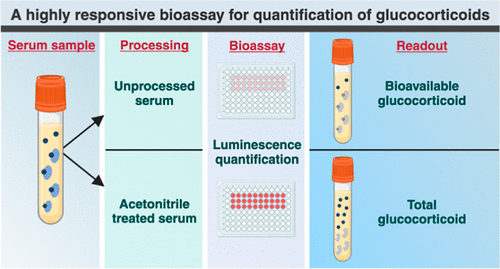
Highly Reliable Cell-Based Assay Enables Accurate Diagnosis of Endocrine Diseases
The conventional methods for measuring free cortisol, the body's stress hormone, from blood or saliva are quite demanding and require sample processing. The most common method, therefore, involves collecting... Read moreMolecular Diagnostics
view channel
New Genetic Testing Procedure Combined With Ultrasound Detects High Cardiovascular Risk
A key interest area in cardiovascular research today is the impact of clonal hematopoiesis on cardiovascular diseases. Clonal hematopoiesis results from mutations in hematopoietic stem cells and may lead... Read more
Blood Samples Enhance B-Cell Lymphoma Diagnostics and Prognosis
B-cell lymphoma is the predominant form of cancer affecting the lymphatic system, with about 30% of patients with aggressive forms of this disease experiencing relapse. Currently, the disease’s risk assessment... Read moreHematology
view channel
Next Generation Instrument Screens for Hemoglobin Disorders in Newborns
Hemoglobinopathies, the most widespread inherited conditions globally, affect about 7% of the population as carriers, with 2.7% of newborns being born with these conditions. The spectrum of clinical manifestations... Read more
First 4-in-1 Nucleic Acid Test for Arbovirus Screening to Reduce Risk of Transfusion-Transmitted Infections
Arboviruses represent an emerging global health threat, exacerbated by climate change and increased international travel that is facilitating their spread across new regions. Chikungunya, dengue, West... Read more
POC Finger-Prick Blood Test Determines Risk of Neutropenic Sepsis in Patients Undergoing Chemotherapy
Neutropenia, a decrease in neutrophils (a type of white blood cell crucial for fighting infections), is a frequent side effect of certain cancer treatments. This condition elevates the risk of infections,... Read more
First Affordable and Rapid Test for Beta Thalassemia Demonstrates 99% Diagnostic Accuracy
Hemoglobin disorders rank as some of the most prevalent monogenic diseases globally. Among various hemoglobin disorders, beta thalassemia, a hereditary blood disorder, affects about 1.5% of the world's... Read moreImmunology
view channel
Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
Transplanted organs constantly face the risk of being rejected by the recipient's immune system which differentiates self from non-self using T cells and B cells. T cells are commonly associated with acute... Read more
AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
Current strategies for matching cancer patients with specific treatments often depend on bulk sequencing of tumor DNA and RNA, which provides an average profile from all cells within a tumor sample.... Read more
Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
Cancer treatment typically adheres to a standard of care—established, statistically validated regimens that are effective for the majority of patients. However, the disease’s inherent variability means... Read morePathology
view channel.jpg)
Use of DICOM Images for Pathology Diagnostics Marks Significant Step towards Standardization
Digital pathology is rapidly becoming a key aspect of modern healthcare, transforming the practice of pathology as laboratories worldwide adopt this advanced technology. Digital pathology systems allow... Read more
First of Its Kind Universal Tool to Revolutionize Sample Collection for Diagnostic Tests
The COVID pandemic has dramatically reshaped the perception of diagnostics. Post the pandemic, a groundbreaking device that combines sample collection and processing into a single, easy-to-use disposable... Read moreAI-Powered Digital Imaging System to Revolutionize Cancer Diagnosis
The process of biopsy is important for confirming the presence of cancer. In the conventional histopathology technique, tissue is excised, sliced, stained, mounted on slides, and examined under a microscope... Read more
New Mycobacterium Tuberculosis Panel to Support Real-Time Surveillance and Combat Antimicrobial Resistance
Tuberculosis (TB), the leading cause of death from an infectious disease globally, is a contagious bacterial infection that primarily spreads through the coughing of patients with active pulmonary TB.... Read moreTechnology
view channel
New Diagnostic System Achieves PCR Testing Accuracy
While PCR tests are the gold standard of accuracy for virology testing, they come with limitations such as complexity, the need for skilled lab operators, and longer result times. They also require complex... Read more
DNA Biosensor Enables Early Diagnosis of Cervical Cancer
Molybdenum disulfide (MoS2), recognized for its potential to form two-dimensional nanosheets like graphene, is a material that's increasingly catching the eye of the scientific community.... Read more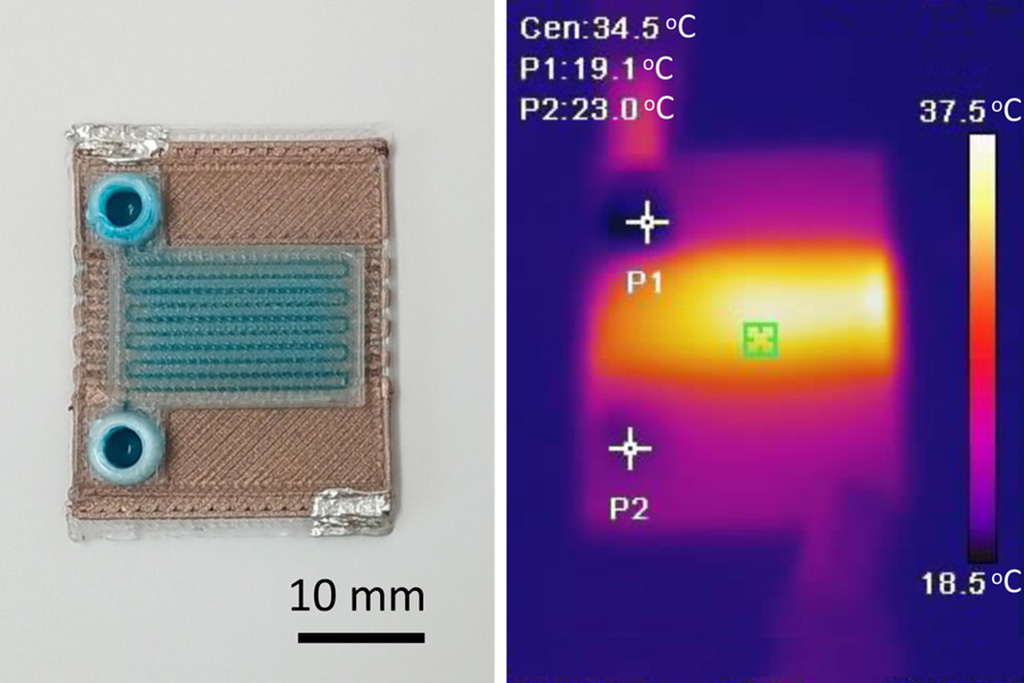
Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
Microfluidics, which are miniature devices that control the flow of liquids and facilitate chemical reactions, play a key role in disease detection from small samples of blood or other fluids.... Read more
Breakthrough in Diagnostic Technology Could Make On-The-Spot Testing Widely Accessible
Home testing gained significant importance during the COVID-19 pandemic, yet the availability of rapid tests is limited, and most of them can only drive one liquid across the strip, leading to continued... Read moreIndustry
view channel_1.jpg)
Thermo Fisher and Bio-Techne Enter Into Strategic Distribution Agreement for Europe
Thermo Fisher Scientific (Waltham, MA USA) has entered into a strategic distribution agreement with Bio-Techne Corporation (Minneapolis, MN, USA), resulting in a significant collaboration between two industry... Read more
ECCMID Congress Name Changes to ESCMID Global
Over the last few years, the European Society of Clinical Microbiology and Infectious Diseases (ESCMID, Basel, Switzerland) has evolved remarkably. The society is now stronger and broader than ever before... Read more