New Blood Tests for TB Could Accelerate Diagnosis
|
By LabMedica International staff writers Posted on 30 Jan 2019 |
Image: The ELISpot TB diagnostic test kit for tuberculosis (Photo courtesy of Oxford Immunotec).
Tuberculosis (TB) is a bacterial infection affecting the lungs, causing cough, weight loss and fevers, and is spread through droplets from coughs and sneezes from infected patients. Diagnosing and treating the condition early is essential for the health of the patient as well as for preventing the spread of TB to others.
There is therefore a need for rapid, convenient tests to rule out a TB diagnosis in suspected cases based on a blood sample. While laboratory cultures of patient samples, such as sputum or invasive biopsies, are used to confirm the presence of the bacterium, the cultures can take several weeks. A negative result does not rule out a diagnosis of TB, as the bacteria cannot be cultured from samples in a large proportion of TB patients.
A team of British scientists working with the Imperial College London (London, UK) carried out a prospective study comparing existing commercially available interferon-gamma release-assays (IGRA) against new generation tests in 845 patients with suspected TB in 10 NHS hospitals in England. Patient blood samples were analyzed using both sets of tests, the results of which benchmarked against a confirmed diagnoses based on positive culture results.
Patients were tested for Mycobacterium tuberculosis infection at baseline with commercially available assays: T-SPOT. TB and QuantiFERON-TB Gold In-Tube and second-generation IGRAs incorporating novel M tuberculosis antigens, and followed up for six to12 months to establish definitive diagnoses. These new antigens are used in the generic ELISpot (enzyme-linked immunospot) platform technology on which the commercial T-SPOT.TB test is based. Sensitivity, specificity, positive and negative likelihood ratios, and predictive values of the tests were determined.
Analysis of the rapid test results revealed that the second-generation test has a diagnostic sensitivity of 94% in patients with confirmed TB - meaning it gives a positive result for 94% of patients with infection, significantly and substantially higher than either of the existing commercially available IGRA tests, which range from 67.3% and 81.4%. The findings indicate the test would be much more accurate at ruling out TB infection in suspected cases of TB, so saving time and resources and enabling patients to receive treatment more rapidly.
The authors concluded that commercially available IGRAs do not have sufficient accuracy for diagnostic evaluation of suspected tuberculosis. Second-generation tests, however, might have sufficiently high sensitivity, low negative likelihood ratio, and correspondingly high negative predictive value in low-incidence settings to facilitate prompt rule-out of tuberculosis.
Ajit Lalvani, DM, a professor of Infectious Diseases and a co-author of the study, said, “Tens of thousands of patients undergo diagnostic assessment for symptoms suggestive of TB, resulting in over 5,000 cases of TB diagnosed each year. Stopping the use of the existing, inadequate tests could save the NHS a lot of money. In contrast, the new, more accurate rapid blood test will improve and accelerate diagnostic assessment of patients with suspected TB.” The study was published on January 14, 2019, in the journal Lancet Infectious Diseases.
There is therefore a need for rapid, convenient tests to rule out a TB diagnosis in suspected cases based on a blood sample. While laboratory cultures of patient samples, such as sputum or invasive biopsies, are used to confirm the presence of the bacterium, the cultures can take several weeks. A negative result does not rule out a diagnosis of TB, as the bacteria cannot be cultured from samples in a large proportion of TB patients.
A team of British scientists working with the Imperial College London (London, UK) carried out a prospective study comparing existing commercially available interferon-gamma release-assays (IGRA) against new generation tests in 845 patients with suspected TB in 10 NHS hospitals in England. Patient blood samples were analyzed using both sets of tests, the results of which benchmarked against a confirmed diagnoses based on positive culture results.
Patients were tested for Mycobacterium tuberculosis infection at baseline with commercially available assays: T-SPOT. TB and QuantiFERON-TB Gold In-Tube and second-generation IGRAs incorporating novel M tuberculosis antigens, and followed up for six to12 months to establish definitive diagnoses. These new antigens are used in the generic ELISpot (enzyme-linked immunospot) platform technology on which the commercial T-SPOT.TB test is based. Sensitivity, specificity, positive and negative likelihood ratios, and predictive values of the tests were determined.
Analysis of the rapid test results revealed that the second-generation test has a diagnostic sensitivity of 94% in patients with confirmed TB - meaning it gives a positive result for 94% of patients with infection, significantly and substantially higher than either of the existing commercially available IGRA tests, which range from 67.3% and 81.4%. The findings indicate the test would be much more accurate at ruling out TB infection in suspected cases of TB, so saving time and resources and enabling patients to receive treatment more rapidly.
The authors concluded that commercially available IGRAs do not have sufficient accuracy for diagnostic evaluation of suspected tuberculosis. Second-generation tests, however, might have sufficiently high sensitivity, low negative likelihood ratio, and correspondingly high negative predictive value in low-incidence settings to facilitate prompt rule-out of tuberculosis.
Ajit Lalvani, DM, a professor of Infectious Diseases and a co-author of the study, said, “Tens of thousands of patients undergo diagnostic assessment for symptoms suggestive of TB, resulting in over 5,000 cases of TB diagnosed each year. Stopping the use of the existing, inadequate tests could save the NHS a lot of money. In contrast, the new, more accurate rapid blood test will improve and accelerate diagnostic assessment of patients with suspected TB.” The study was published on January 14, 2019, in the journal Lancet Infectious Diseases.
Latest Immunology News
- AI Predicts Tumor-Killing Cells with High Accuracy
- Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
- AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
- Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
- Testing Method Could Help More Patients Receive Right Cancer Treatment
- Groundbreaking Test Monitors Radiation Therapy Toxicity in Cancer Patients
- State-Of-The Art Techniques to Investigate Immune Response in Deadly Strep A Infections
- Novel Immunoassays Enable Early Diagnosis of Antiphospholipid Syndrome
- New Test Could Predict Immunotherapy Success for Broader Range Of Cancers
- Simple Blood Protein Tests Predict CAR T Outcomes for Lymphoma Patients
- Cell Sorter Chip Technology to Pave Way for Immune Profiling at POC
- Chip Monitors Cancer Cells in Blood Samples to Assess Treatment Effectiveness
- Automated Immunohematology Approaches Can Resolve Transplant Incompatibility
- AI Leverages Tumor Genetics to Predict Patient Response to Chemotherapy
- World’s First Portable, Non-Invasive WBC Monitoring Device to Eliminate Need for Blood Draw
- Predictive T-Cell Test Detects Immune Response to Viruses Even Before Antibodies Form
Channels
Clinical Chemistry
view channel
3D Printed Point-Of-Care Mass Spectrometer Outperforms State-Of-The-Art Models
Mass spectrometry is a precise technique for identifying the chemical components of a sample and has significant potential for monitoring chronic illness health states, such as measuring hormone levels... Read more.jpg)
POC Biomedical Test Spins Water Droplet Using Sound Waves for Cancer Detection
Exosomes, tiny cellular bioparticles carrying a specific set of proteins, lipids, and genetic materials, play a crucial role in cell communication and hold promise for non-invasive diagnostics.... Read more
Highly Reliable Cell-Based Assay Enables Accurate Diagnosis of Endocrine Diseases
The conventional methods for measuring free cortisol, the body's stress hormone, from blood or saliva are quite demanding and require sample processing. The most common method, therefore, involves collecting... Read moreMolecular Diagnostics
view channel
World’s First One-Minute Hepatitis C Antibody Test Facilitates Quick Triage
Rapid and accurate testing is crucial in the fight against Hepatitis C. Hepatitis C blood testing determines whether someone has been infected with the Hepatitis C virus. Having regular access to testing... Read more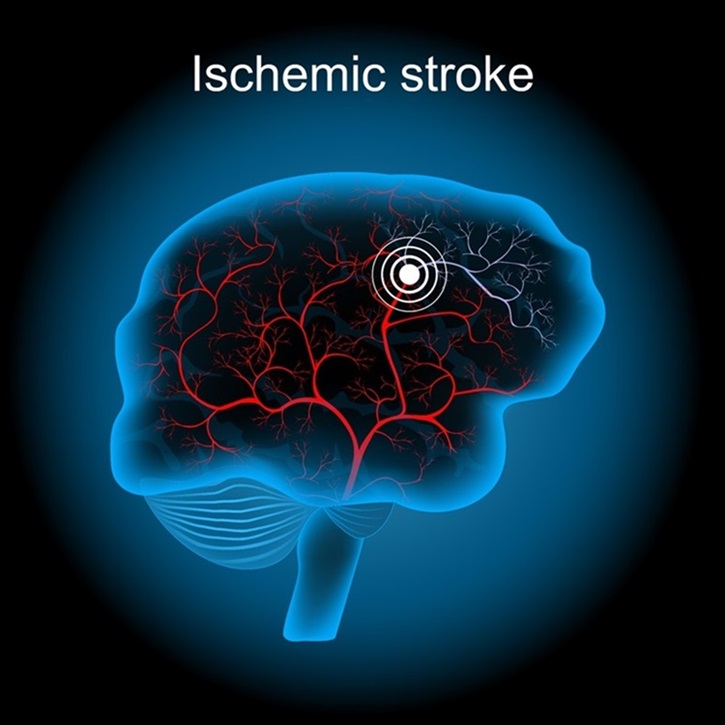
Game-Changing Blood Test for Stroke Detection Could Bring Life-Saving Care to Patients
Stroke is the primary cause of disability globally and ranks as the second leading cause of death. However, timely early intervention can prevent severe outcomes. Most strokes are ischemic, resulting from... Read moreHematology
view channel
Next Generation Instrument Screens for Hemoglobin Disorders in Newborns
Hemoglobinopathies, the most widespread inherited conditions globally, affect about 7% of the population as carriers, with 2.7% of newborns being born with these conditions. The spectrum of clinical manifestations... Read more
First 4-in-1 Nucleic Acid Test for Arbovirus Screening to Reduce Risk of Transfusion-Transmitted Infections
Arboviruses represent an emerging global health threat, exacerbated by climate change and increased international travel that is facilitating their spread across new regions. Chikungunya, dengue, West... Read more
POC Finger-Prick Blood Test Determines Risk of Neutropenic Sepsis in Patients Undergoing Chemotherapy
Neutropenia, a decrease in neutrophils (a type of white blood cell crucial for fighting infections), is a frequent side effect of certain cancer treatments. This condition elevates the risk of infections,... Read more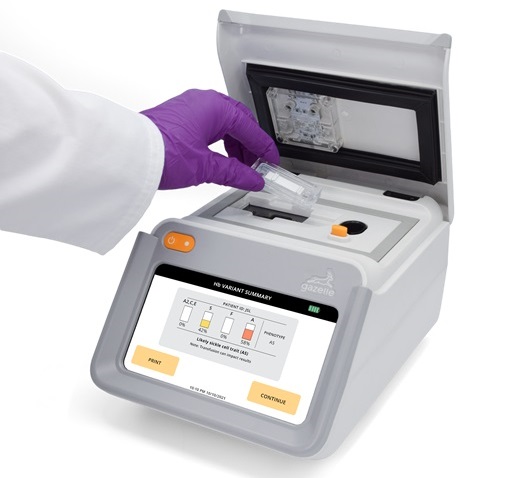
First Affordable and Rapid Test for Beta Thalassemia Demonstrates 99% Diagnostic Accuracy
Hemoglobin disorders rank as some of the most prevalent monogenic diseases globally. Among various hemoglobin disorders, beta thalassemia, a hereditary blood disorder, affects about 1.5% of the world's... Read moreImmunology
view channel.jpg)
AI Predicts Tumor-Killing Cells with High Accuracy
Cellular immunotherapy involves extracting immune cells from a patient's tumor, potentially enhancing their cancer-fighting capabilities through engineering, and then expanding and reintroducing them into the body.... Read more
Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
Transplanted organs constantly face the risk of being rejected by the recipient's immune system which differentiates self from non-self using T cells and B cells. T cells are commonly associated with acute... Read more
AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
Current strategies for matching cancer patients with specific treatments often depend on bulk sequencing of tumor DNA and RNA, which provides an average profile from all cells within a tumor sample.... Read more
Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
Cancer treatment typically adheres to a standard of care—established, statistically validated regimens that are effective for the majority of patients. However, the disease’s inherent variability means... Read morePathology
view channel
Groundbreaking CRISPR Screen Technology Rapidly Determines Disease Mechanism from Tissues
Thanks to over a decade of advancements in human genetics, scientists have compiled extensive lists of genetic variations linked to a wide array of human diseases. However, understanding how a gene contributes... Read more
New AI Tool Classifies Brain Tumors More Quickly and Accurately
Precision in diagnosing and categorizing tumors is essential for delivering effective treatment to patients. Currently, the gold standard for identifying various types of brain tumors involves DNA methylation-based... Read moreTechnology
view channel
New Diagnostic System Achieves PCR Testing Accuracy
While PCR tests are the gold standard of accuracy for virology testing, they come with limitations such as complexity, the need for skilled lab operators, and longer result times. They also require complex... Read more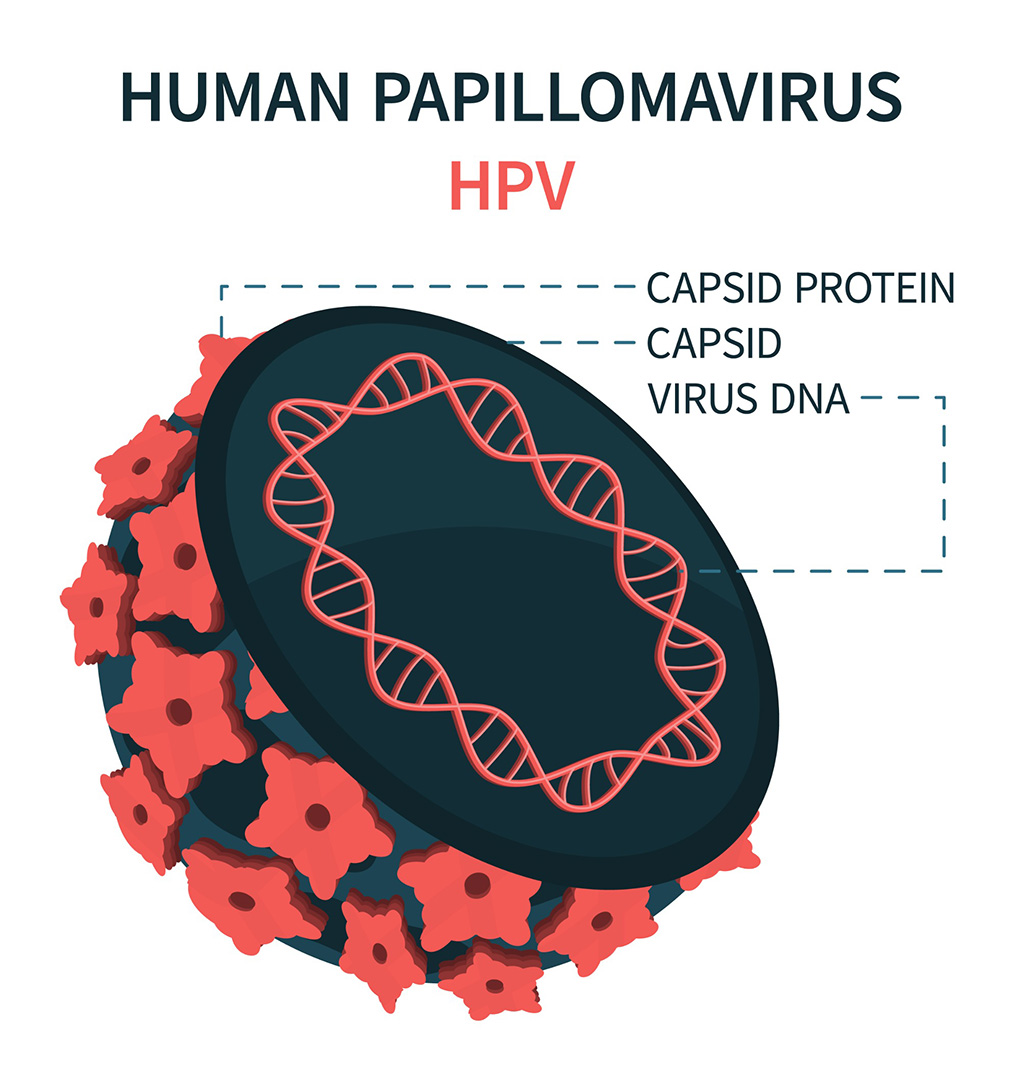
DNA Biosensor Enables Early Diagnosis of Cervical Cancer
Molybdenum disulfide (MoS2), recognized for its potential to form two-dimensional nanosheets like graphene, is a material that's increasingly catching the eye of the scientific community.... Read more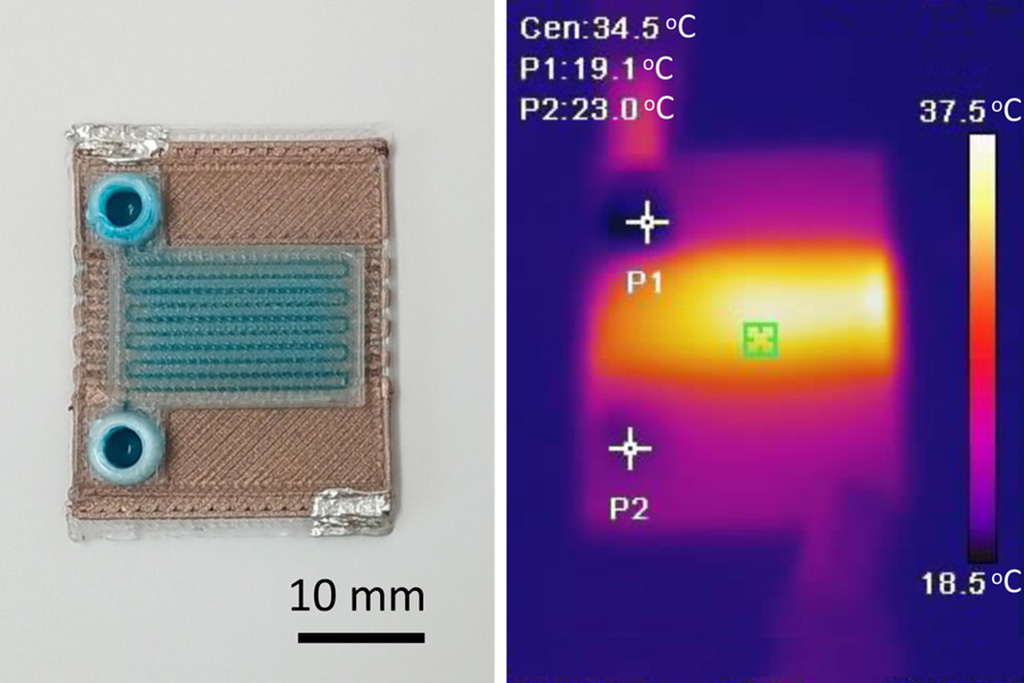
Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
Microfluidics, which are miniature devices that control the flow of liquids and facilitate chemical reactions, play a key role in disease detection from small samples of blood or other fluids.... Read more
Breakthrough in Diagnostic Technology Could Make On-The-Spot Testing Widely Accessible
Home testing gained significant importance during the COVID-19 pandemic, yet the availability of rapid tests is limited, and most of them can only drive one liquid across the strip, leading to continued... Read moreIndustry
view channel
Danaher and Johns Hopkins University Collaborate to Improve Neurological Diagnosis
Unlike severe traumatic brain injury (TBI), mild TBI often does not show clear correlations with abnormalities detected through head computed tomography (CT) scans. Consequently, there is a pressing need... Read more
Beckman Coulter and MeMed Expand Host Immune Response Diagnostics Partnership
Beckman Coulter Diagnostics (Brea, CA, USA) and MeMed BV (Haifa, Israel) have expanded their host immune response diagnostics partnership. Beckman Coulter is now an authorized distributor of the MeMed... Read more_1.jpg)









 Reagent.jpg)